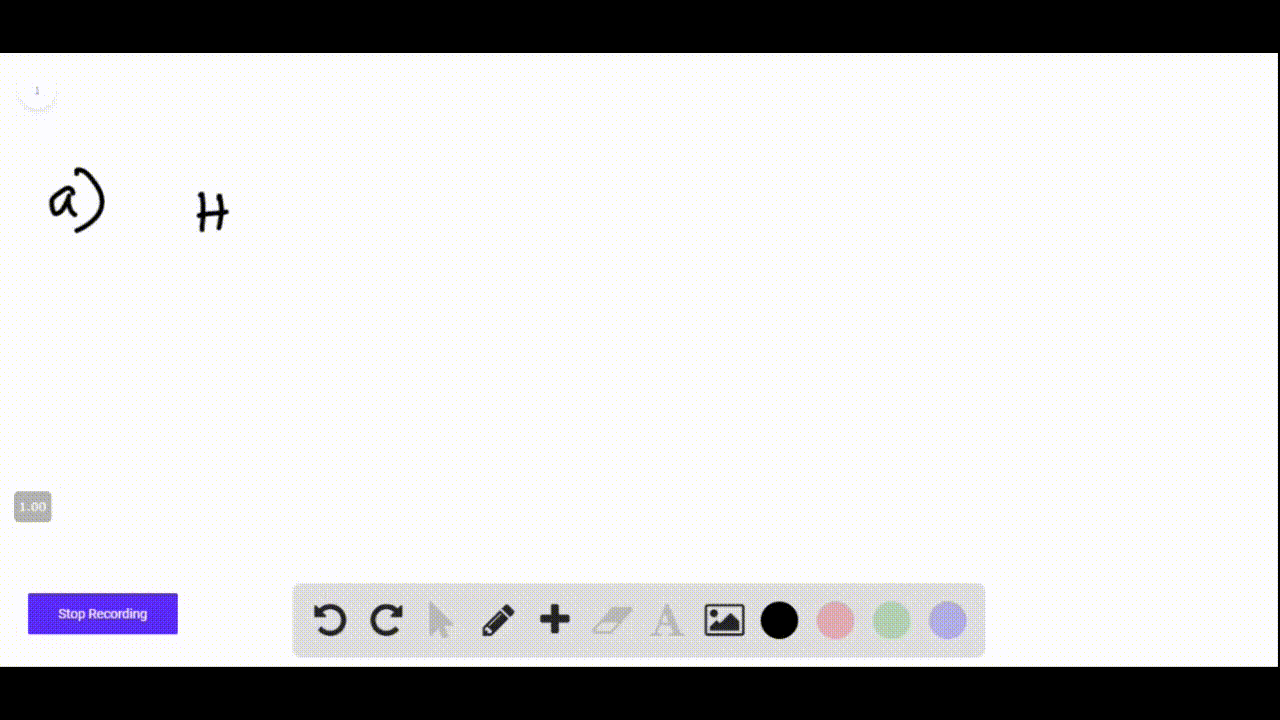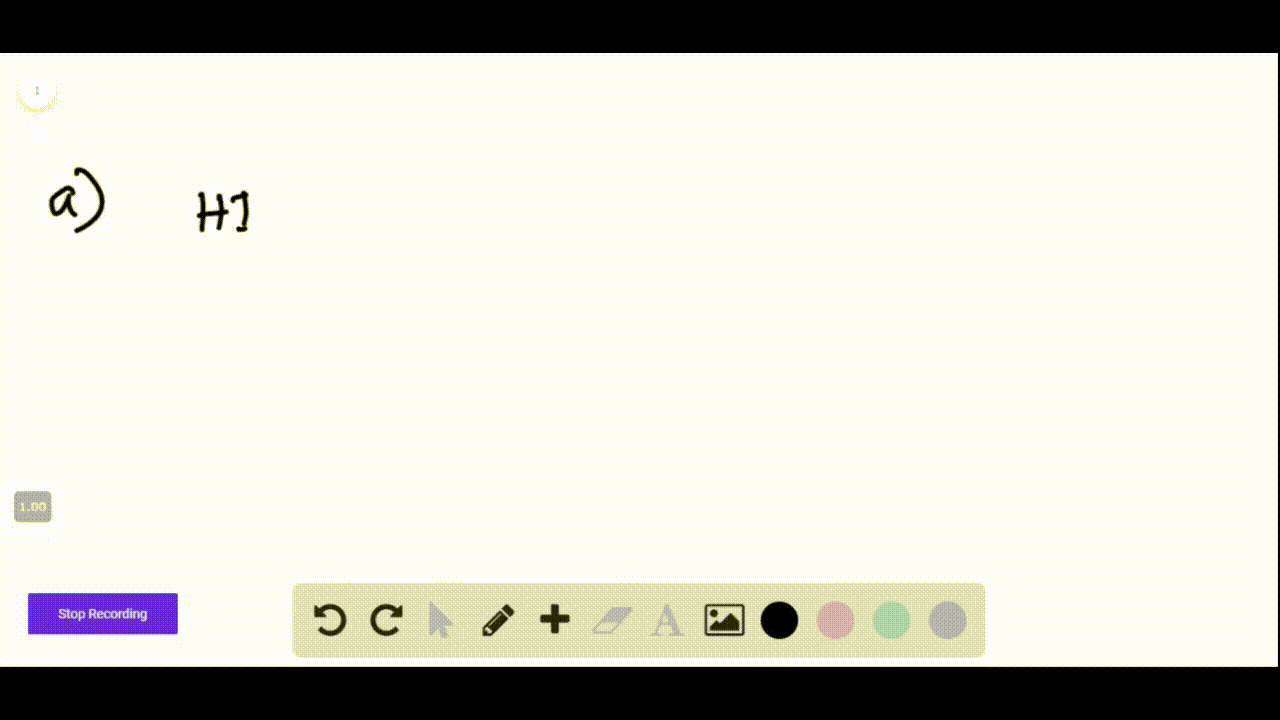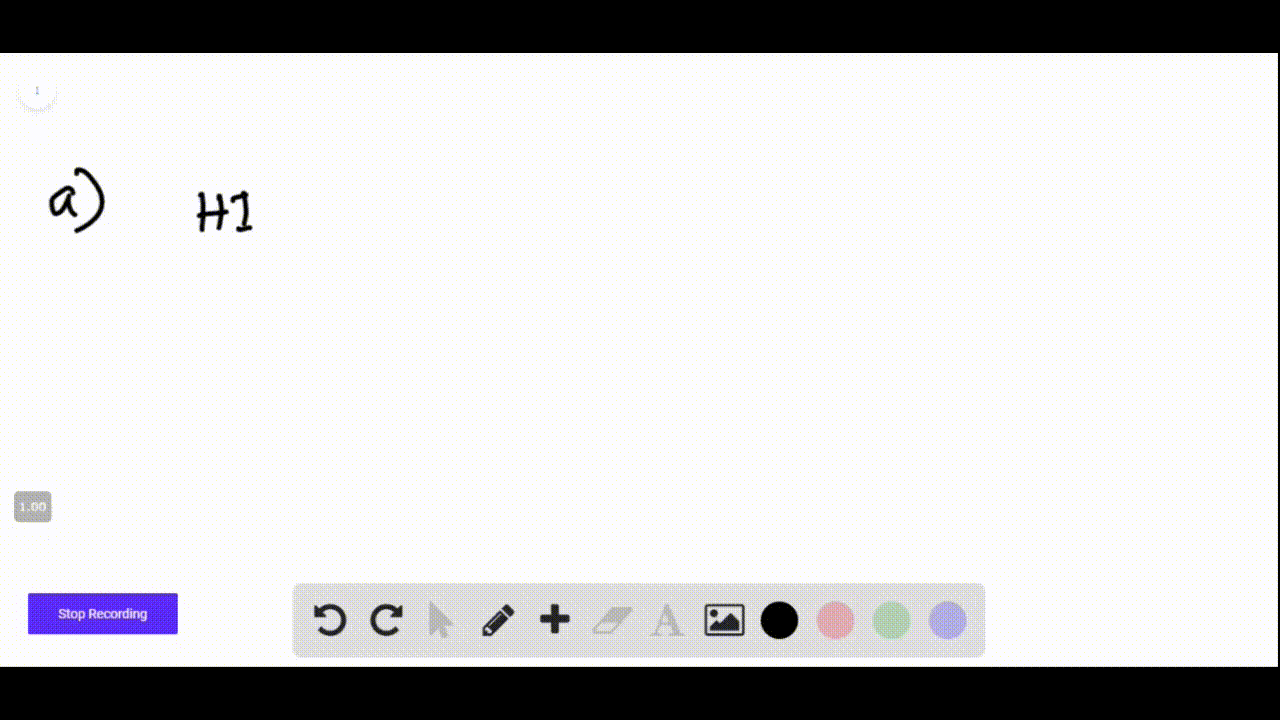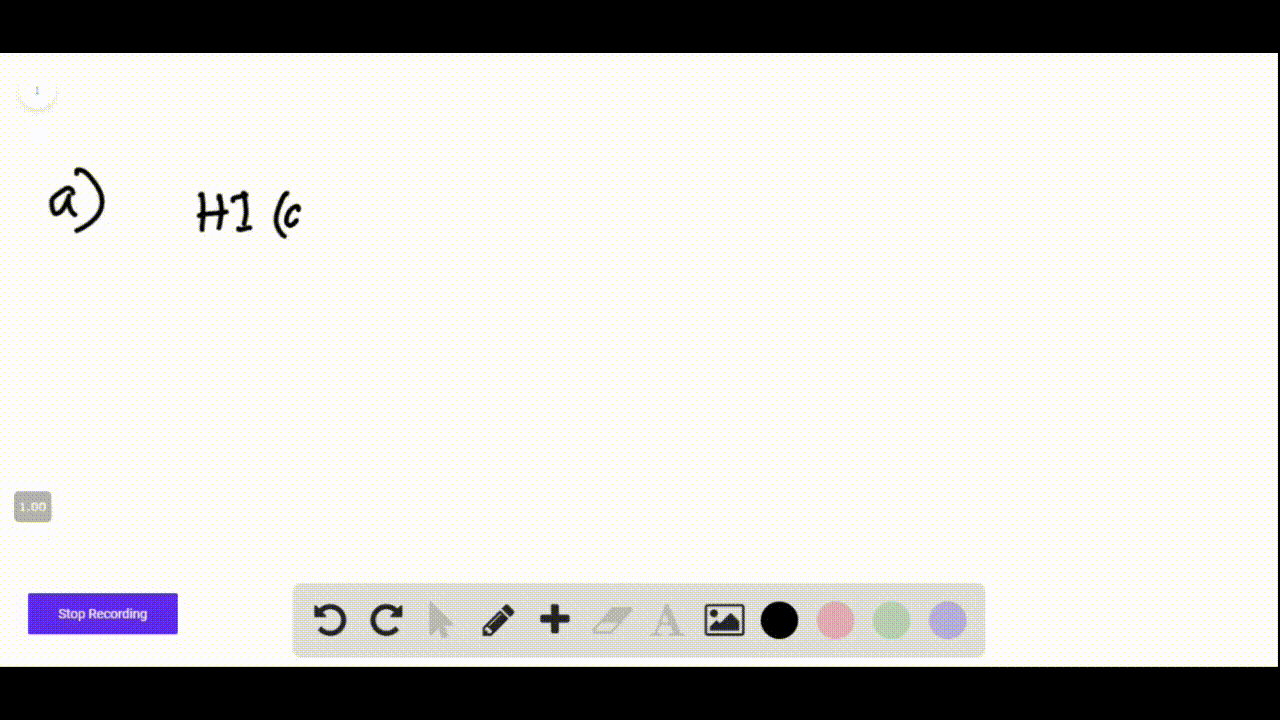
Hi + Naoh Net Ionic
What Is The Ionic Equation For The Reaction Between Calcium Carbonate And Hydrochloric Acid Quora

What Is The Net Ionic Equation For Khc8h4o4 Aq Naoh Aq Knac8h4o4 H2o L Homeworklib

Solved What Is The Net Ionic Equation For The Reaction Th Chegg Com

What Is The Net Ionic Equation Of The Reaction Of Fecl2 With Naoh Express You Answer As A Chemical Brainly In
Volumetric Titrations Analytical Chemistry Video Clutch Prep

How To Balance Naoh Hi Nai H2o Sodium Hydroxide Hydroiodic Acid Youtube
The net ionic equation for the reaction that results from mixing 1 M HCl and 1 M NaOH is H (aq) OH (aq) → H 2 O (l) The Cl and Na ions do not react and are not listed in the net ionic equation How to Write a Net Ionic Equation.
Hi + naoh net ionic. Problem 60 Hard Difficulty Write balanced complete ionic and net ionic equations for each acidbase reaction a HI(aq) RbOH(aq) b HCHO2(aq) NaOH(aq) c HC2H3O2(aq) LiOH(aq). Submit Part C What is the net ionic equation of the reaction of MgSO4 with Pb(NO3)2?. Answer The net ionic equation will be, Explanation Complete ionic equation In complete ionic equation, all the substance that are strong electrolyte and present in an aqueous are represented in the form of ions In the net ionic equations, we are not include the spectator ions in the equations.
(318) seboat@hotmailcom Home;. Net Ionic Equation Definition The net ionic equation is a chemical equation for a reaction that lists only those species participating in the reaction The net ionic equation is commonly used in acidbase neutralization reactions, double displacement reactions, and redox reactions In other words, the net ionic equation applies to reactions. Careful You must realize that H3PO4 is a weak acid and will be only partially dissociated When writing ionic equations involving weak acids, and bases, we do not show them as being completely dissociated as Upamanyu has done Nor can we simply write the reaction of H and OH as CB has done.
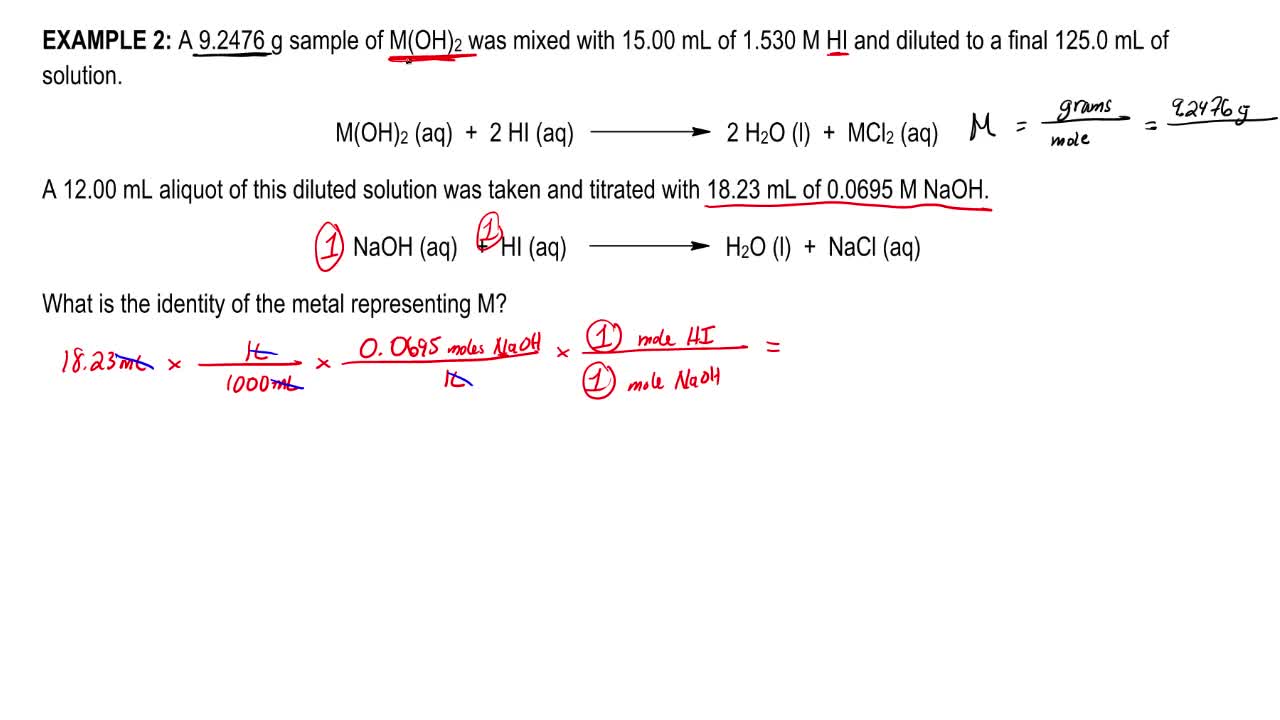
Express your answer as a chemical equation View Available Hint(s) ALP R O ?. HI(aq) 1 NaOH(aq) 1 NaI 1 H 2 O 1 Units molar mass g/mol, weight g Please tell about this free chemistry software to your friends!. Consider the chemical equation HI(aq) NaOH(aq) — Nal(aq) H, O(1) What is the net ionic equation for the reaction?.
Our videos will help you understand concepts, solve your homework, and do great on your exams. Hi what is the net ionic equation NH4Cl NaOH?. MathH_2SO_4 (aq) 2 KOH(aq) \to K_2SO_4(aq) 2H_2O(l)/math That's the overall reaction The net ionic reaction shows only the molecules and ions that react The potassium mathK^/math is already detached from the mathOH^/math and it.
Always use the upper case for the first character in the element name and the lower case for the second character. Question 1 37 pts Write a net ionic equation for the reaction of aqueous hydrocyanic acid and aqueous sodium hydroxide My answer book tells me that in the net ionic equation excludes Na from the equation. My answer book tells me that in the net ionic equation excludes Na from the equation My question is why?.
HI(aq) NaOH(s) → NaI(aq) H 2 O(l) a 0325 L b 516 L c 130 L d 812 L 11 The pH of a M solution of HCl is a 2 b 1118 c 250 d 1150 12. Instructions on balancing chemical equations Enter an equation of a chemical reaction and click 'Balance' The answer will appear below;. There are three main steps for writing the net ionic equation for CuSO4 NaOH = Cu(OH)2 Na2SO4 (Copper (II) sulfate Sodium hydroxide) First, we balance.
Write the ionic and net ionic equation for the following neutralization reactions Label the acid, the base, conjugate base, and conjugate acid Be sure to balance the equations. Chemistry Write the remaining substances as the net ionic equation Use uppercase for the first character in the element and lowercase for the second character Write the remaining substances as. Get an answer for 'What is the ionic equation of NaOH HCl > H2O NaCl' and find homework help for other Science questions at eNotes Follow the link for rules to write net ionic equation.
There are three main steps for writing the net ionic equation for HCN NaOH = NaCN H2O (Hydrogen cyanide Sodium hydroxide) First, we balance the molecu. Answer Save 5 Answers Relevance DrA Lv 7 1 decade ago Favorite Answer NH4Cl NaOH >> NH4OH NaCl NH4 OH >> NH4OH 0 0 hannahmontana 1 decade ago NH4Cl NaOH > NH4OH Na Clso the net ionic equation is NH4 OH > NH4OH and NaCl doesn't form b/c Na and Cl are. Answer Save 5 Answers Relevance DrA Lv 7 1 decade ago Favorite Answer NH4Cl NaOH >> NH4OH NaCl NH4 OH >> NH4OH 0 0 hannahmontana 1 decade ago NH4Cl NaOH > NH4OH Na Clso the net ionic equation is NH4 OH > NH4OH and NaCl doesn't form b/c Na and Cl are.
This reaction is considered a neutralization reaction The base (NaOH) and weak acid (CH 3 COOH) react to produce a salt (NaNO 3 and water (H 2 O) Video NaOH CH3COOH (Net Ionic Equation) How to Write the Net Ionic Equation for NaOH CH3COOH = CH3COONa H2O YouTube MagnetsAndMotors. HI(aq) 1 NaOH(aq) 1 NaI 1 H 2 O 1 Units molar mass g/mol, weight g Please tell about this free chemistry software to your friends!. We know, HCl, NaOH and NaCl are strong electrolytes and H2O is weak electrolyte In aqueous solution, HCl, NaOH and NaCl remain in dissociated form So, we can write the given chemical equation as below H Cl Na OH → Na Cl H2O(l) To.
In order for the reaction to be a full neutralization, twice as many moles of NaOH must react with the H 2 SO 4 The sodium sulfate salt is soluble, and so the net ionic reaction is again the same Different mole ratios occur for other polyprotic acids or bases with multiple hydroxides such as Ca(OH) 2. There are three main steps for writing the net ionic equation for NaOH H3PO4 = Na3PO4 H2O (Sodium hydroxide Phosphoric acid) First, we balance the mol. The net ionic equation will only show the ions that actually take part in the reaction The reaction between the two solutions will result in the formation of the solid, which means that the ions that are not a part of the solid will not take part in the reaction More specifically, they will act as spectator ions.
EXERCISE 1 IONIC BOND Ql An ionic bond g is most likely to be formed when (A) the ionization energy of4 is high and the electron affinity o f 4 List An excess of NaOH was then added and the solution distilled For each Thus, calling NH4Cl and Na2SO4 ionic compounds is somewhat ambiguous. Cnccd@aolcom Request a Quote!. There are three main steps for writing the net ionic equation for NaOH H3PO4 = Na3PO4 H2O (Sodium hydroxide Phosphoric acid).
What is the net ionic equation for the reaction of NaOH(aq) with HCN(aq)?. O H (aq) OH (aq) — H2O(1) O (aq) I. H2so4 naoh net ionic equation h2so4 naoh net ionic equation.
Limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents Therefore, What Is The Net Ionic Equation For Na2CO3 AgNO3?. How to Balance the Net Ionic Equation for HClO 4 NaOH The reaction of Perchloric acid and Sodium hydroxide represents a net ionic equation involving a strong acid and strong base Strong acids and strong bases are considered strong electrolytes and will dissociate completely This means that we will split them apart in the net ionic equation. Problem 60 Hard Difficulty Write balanced complete ionic and net ionic equations for each acidbase reaction a HI(aq) RbOH(aq) b HCHO2(aq) NaOH(aq) c HC2H3O2(aq) LiOH(aq).
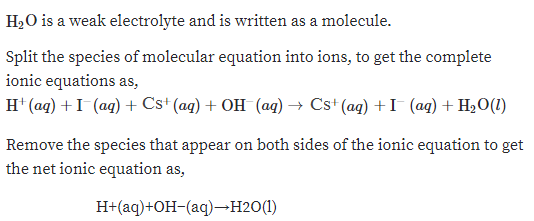
Write the balanced complete ionic equation for the reaction HI(aq)CsOH(aq)→ Write the balanced net ionic equation for the reaction HI(aq)CsOH(aq)→Write the balanced complete ionic equation for the reaction. AgNO3 (aq)NaCl(aq) > NaNO3 (aq)AgCl(s) (List the ions in order of the above equation) Write the net ionic equation for the reaction above AP Chemistry I'm having trouble getting the net ionic equation when HI is added to a buffered solution of HOBr And the net ionic when HI is added to LiOBr. In order for the reaction to be a full neutralization, twice as many moles of NaOH must react with the H 2 SO 4 The sodium sulfate salt is soluble, and so the net ionic reaction is again the same Different mole ratios occur for other polyprotic acids or bases with multiple hydroxides such as Ca(OH) 2.
Here's how you can do that The thing to recognize here is the fact that you're dealing with a neutralization reaction that features sodium hydroxide, "NaOH", a strong base, and hydrochloric acid, "HCl", a strong acid This tells you that the two reactants will dissociate completely in aqueous solution to produce cations and anions More specifically, you will have "NaOH"_ ((aq)) > "Na"_ ((aq. The net ionic equation is commonly used in acidbase neutralization reactions, double displacement reactions, and redox reactions Ionic charges are not yet supported and will be ignored Concept Problem What is the net ionic equation for NaOH(aq) HCl(aq) â NaCl(aq) H2O(l) *Response times vary by subject and question complexity. Direct link to this balanced equation Instructions on balancing chemical equations.
This means that we will split them apart in the net ionic equation HNO 3 NaOH = NaNO 3 H 2 O is a neutralization reaction (also a double displacement reaction) The strong acid (HNO 3 ) and strong base react to produce a salt (NaNO 3 ) and water (H 2 O). Part B What is the net ionic equation of the reaction of FeCl2 with NaOH ?. We can write the overall equation for the reaction first H2SO4 2NaOH → Na2SO4 2H2O The ionic equation omits ions which act as spectators so we can split the reactants and the products into ions to see which are the spectators H2SO4 → 2H (.
Question 1 )The Reaction HI(aq) NaOH(aq) → H2O(l) NaI(aq) Is A(n) _____ Reaction A Precipitation B Decomposition Reaction C Single Replacement D Write the molecular equation, the complete ionic equation, and the net ionic equation for the following reaction Aqueous solutions of copper(II) nitrate and sodium hydroxide are mixed. The reason to write a chemical equation is to express what we believe is actually happening in a chemical reaction One of the most useful applications of the concept of principal species is in writing net ionic equationsThese are equations that focus on the principal substances and ions involved in a reactionthe principal speciesignoring those spectator ions that really don't get involved. My book doesn't offer any explanation as to why this is and everything with the exception of water is aqueous I don't understand why Na is excluded and not CNAnother thing that came.
How to Balance the Net Ionic Equation for HClO 4 NaOH The reaction of Perchloric acid and Sodium hydroxide represents a net ionic equation involving a strong acid and strong base Strong acids and strong bases are considered strong electrolytes and will dissociate completely This means that we will split them apart in the net ionic equation. Direct link to this balanced equation Instructions on balancing chemical equations. Express your answer as a chemical equation.
There are three main steps for writing the net ionic equation for HI NaOH = NaI H2O (Hydroiodic acid Sodium hydroxide) First, we balance the molecular. HCN aq NaOH aq >. HF OH F H 2 O net ionic BronstedLowry Theory – An acid is a proton (H ) donor and a base is a proton acceptor Any species defined as an acid by Arrhenius will also be defined as an acid by BronstedLowry, but BronstedLowry is more general.
To write the net ionic equation for HCl NaOH = NaCl H2O (Hydrochloric acid Sodium hydroxide) we follow main three steps First, we balance the molecula. There are three main steps for writing the net ionic equation for NaOH HF = NaF H2O (Sodium hydroxide Hydrofluoric acid) First, we balance the molecul. H2O l NaCN aq?.
Our videos prepare you to succeed in your college classes Let us help you simplify your studying If you are having trouble with Chemistry, Organic, Physics, Calculus, or Statistics, we got your back!.

Net Ionic Equations

Acids Bases
Http Web2 Utc Edu Johnny Cantrell Notes Netionic Pdf
Http G Web Umkc Edu Gounevt Weblec211 Pdf Lec10 3 5 3 8 Pdf

Aqueous Reactions
Www Angelo Edu Faculty Kboudrea Index Chapter 04 2 Pdf
People Uwec Edu Bhattas Ppt Ch4 Ch5 A Pdf
Http Library Uob Edu Bh En Teachingresources Chem Chemy 101 Test2 July 05 Pdf

Solved Write Balanced Complete Ionic And Net Ionic Equati Chegg Com

Answered Write The Balanced Molecular Equation Bartleby
People Uwec Edu Bhattas Ppt Ch4 Ch5 A Pdf

Part A Write Balanced Molecular Equation For The Reaction Between Nitric Acid Course Hero

Write Balanced Complete Ionic Equation For The Reaction Hcl Aq Naoh Aq Express Your Answer As A Chemical Equation Homeworklib

File
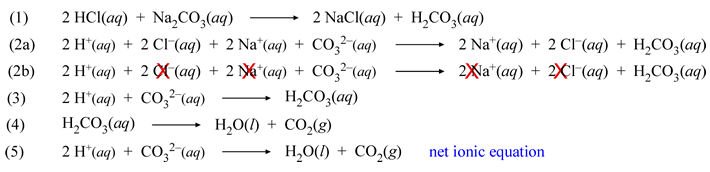
Chem 101 Acids And Bases Introduction

Solved Ionic And Net Ionic Equations 5 Write Balanced Co Chegg Com
Oneclass Suppose A Buffer Solution Is Made From Formic Acid Hcho 2 And Sodium Formate Nacho 2 Wh
Christopherking Name Genchemonelab Manual 09 double displacement reactions F19 Pdf

Ppt Strong Bases Lioh Naoh Koh Rboh Csoh Ca Oh 2 Sr Oh 2 Ba Oh 2 Powerpoint Presentation Id
Http People Chem Umass Edu Cjoseph Chem111 Lecture9 Pdf
Www Angelo Edu Faculty Kboudrea Index Chapter 04 2 Pdf
Resources Finalsite Net Images V Salisbury Uamtacvexfgquniau1fd Apchemistrysummer19 Pdf

Solved Write Balanced Complete Ionic And Net Ionic Equati Chegg Com

Write An Equation For Nh4oh Solution In Water

Notes On Chemical Formula Notes
Christopherking Name Genchemonelab Manual 09 double displacement reactions F19 Pdf
Q Tbn And9gcsqplpzyo0j3jdiklussmkr9yib1jpd8ywwavykgtkpp7urjnug Usqp Cau

Net Ionic Equation For Hno3 Naoh Strong Base And Strong Acid

Ap Chemistry Chapter 4 Quiz Flashcards Quizlet

How To Write The Net Ionic Equation For Hcl Naoh Nacl H2o Youtube

Key Gcc

Question 14 What Is The Net Ionic Equation For Hcl Aq Naoh Aq Nacl Aq H2o 1 Homeworklib
Http Mrosechemistry Weebly Com Uploads 2 7 5 8 Answershw8b Pdf

Write Balanced Complete Ionic And Net Ioni Clutch Prep
Http Www Chemunlimited Com X0111spg18 0104 Version a Pdf
Q Tbn And9gcsqplpzyo0j3jdiklussmkr9yib1jpd8ywwavykgtkpp7urjnug Usqp Cau

6 Reactions In Aqueous Solutions Pdf Free Download

Solved Consider The Chemical Equation Hi Aq Naoh Aq Chegg Com

Write Down The Net Ionic Equation Reaction Between Soldium Hydroxide And Potassium Hydrogen Phthalate Brainly In
Solved Write Balanced Complete Ionic And Net Ioni

Classifying Compounds Compounds Can Be Classified By The Number Of Different Elements They Contain Binary Ternary Or Quaternary Hi Naoh Nh 4 Clo Ppt Download

I Really Need Some Help With Number 3 And Number 4 If You Could Also Explain Homeworklib
Www Wmich Edu Asc Files Ch4ans Pdf
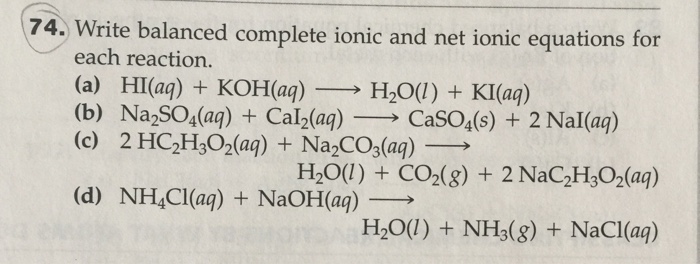
Solved Write Balanced Complete Ionic And Net Ionic Equati Chegg Com
2
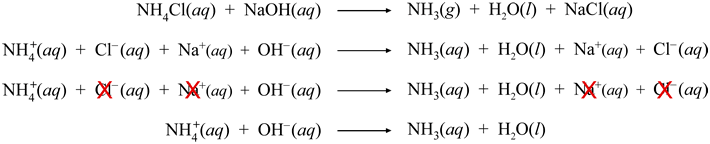
Chem 101 Acids And Bases Introduction

Titration Of A Strong Acid With A Strong Base Chemistry Libretexts

How Can The Net Ionic Equation For Hcl And Naoh Be Determined Quora
Preparatorychemistry Com Bishop Study Guide Atoms 8 Pdf

Review Exam 2 Chem 1411 Ch 4 5 And 6 Studocu

Solved Write Balanced Complete Ionic And Net Ionic Equati Chegg Com

Chapter 4 Notes

Net Ionic Equations Tutorial Ppt Download
Www Unf Edu Michael Lufaso Chem45 Chapter4 Pdf

How Can The Net Ionic Equation For Hcl And Naoh Be Determined Quora

How To Write The Net Ionic Equation For Hcl Naoh Nacl H2o Youtube

How To Balance Naoh Hi Nai H2o Sodium Hydroxide Hydroiodic Acid Youtube
Www Lamar Edu Arts Sciences Files Documents Chemistry Biochemistry Dorris Chapter4 Pdf

Solved Write Balanced Complete Ionic And Net Ioni

Chemical Reactions 1 H2 I2 Hi S 2 Na H2o Naoh H2 Sr 3 Co O2 Ppt Download
Net Ionic Equation For Naoh Ch3cooh Strong Base And Weak Acid
Why Can T Alcohol React With Aqueous Naoh Physics Forums

Unit 4 Chemical Equations And Stoichiometry Ppt Download

Ppt Ionic Equations Powerpoint Presentation Free Download Id
Answered Enter The Balanced Net Ionic Equation Bartleby

8 7 Acid Base Titration
Http Www Conejousd Org Portals 49 Departments Science Cord 15 Ch 11 reactions H worksheet and sg key Pdf Ver 18 01 09 560
Www Strongnet Org Cms Lib6 Oh Centricity Domain 6 Ch4a Pdf
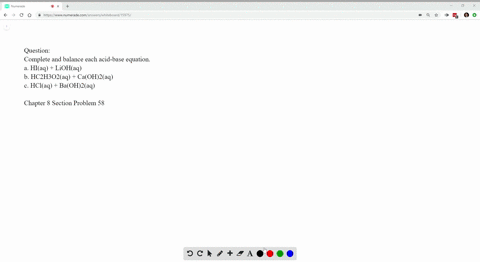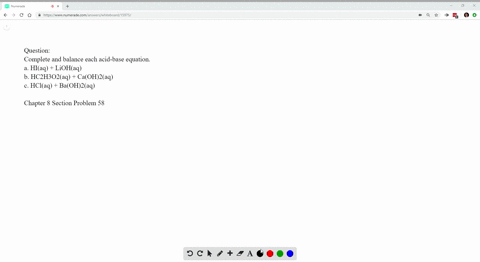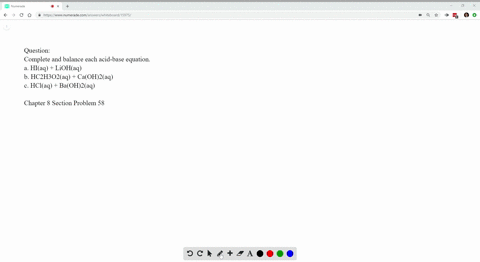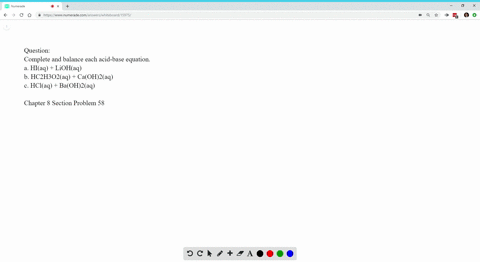
Write Balanced Complete Ionic And Net Ionic Equations For Each Acid Base Reaction A Hi Aq
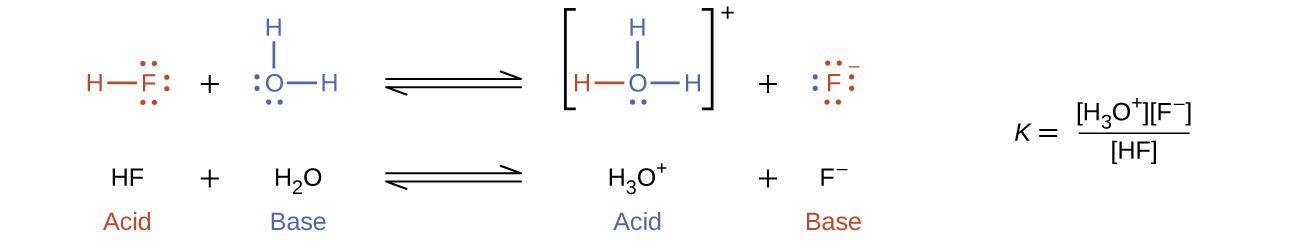
14 1 Bronsted Lowry Acids And Bases Chemistry

Writing And Balancing Chemical Equations Chemistry Atoms First

03 Neutralization Reactions Worksheet Key
3 6 Acid Base Reactions Chemistry Libretexts

Classifying Compounds Compounds Can Be Classified By The Number Of Different Elements They Contain Binary Ternary Or Quaternary Hi Naoh Nh 4 Clo Ppt Download
Ww2 Odu Edu Ppleban Pdf Chem115 Zumdahl4 Pdf
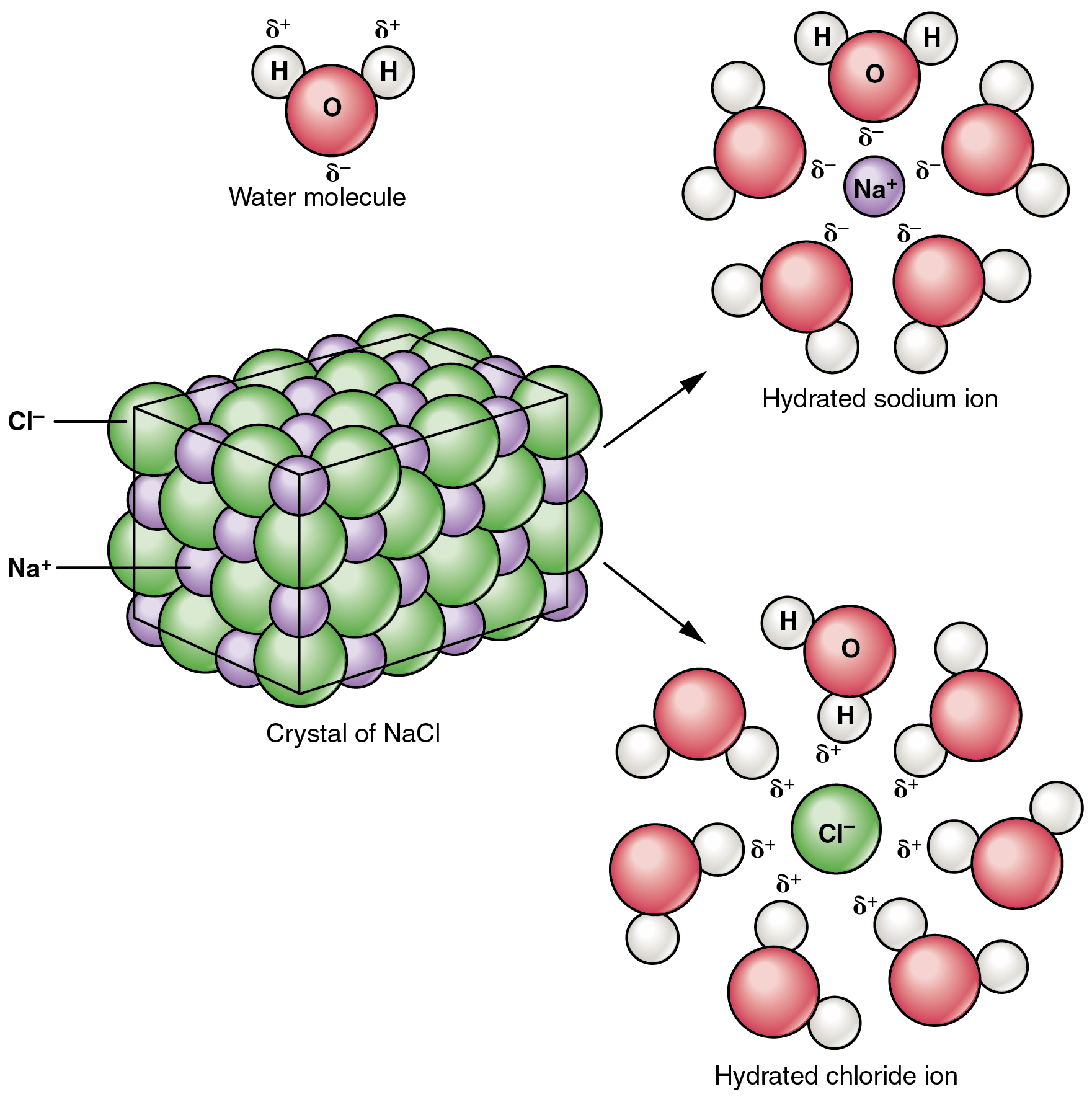
Molecular Complete Ionic And Net Ionic Equations Article Khan Academy

How To Write The Net Ionic Equation For Hi Naoh Nai H2o Youtube

Naohh2so4na2so4h2o Balance Dowload Anime Wallpaper Hd
Http Hartman Science Pbworks Com W File Fetch Ch 4 practice Pdf

Solved Write Balanced Complete Ionic And Net Ionic Equati Chegg Com

Write Balanced Complete Ionic And Net Ionic Equations For Each Reaction A Math Mathrm Hcl A Q Operatorname Lioh A Q Longrightarrow Mathrm H 2 Mathrm O L Mathrm Licl A Q Math B Math Operatorname Mgs A Q Mathrm Cucl 2 A Q
Www Atticacsd Org Site Handlers Filedownload Ashx Moduleinstanceid 444 Dataid 2239 Filename Chapter 4 presentation Pdf

How To Write The Net Ionic Equation For Hi Naoh Nai H2o Youtube
People Wou Edu Postonp Ch222 Pdf Ch08 W15 Pdf

Complete Ionic Equation Net Ionic Equations Slideshow And Powerpoint Viewer Electrolytes From Conductivity Test Electrolytes Are Substances Whose Aqueous Solution Is A Conductor Of Electricity
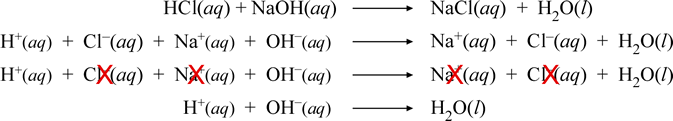
Chem 101 Acids And Bases Introduction

Write The Complete Ionic And Net Ionic Equation For Each Of Clutch Prep

Write A Net Ionic Equation For The Overall Reaction That Occurs When Aqueous Solutions Of Sodium Brainly Com

Chapter 4 Review Sheet
Www Unf Edu Michael Lufaso Chem45 Chapter4 Pdf
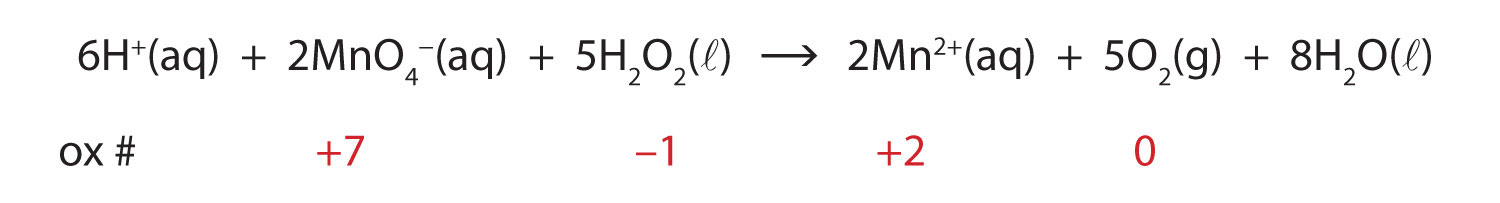
Chemical Reactions And Equations

Answered 4 60 Write Balanced Net Ionic Equations Bartleby
Www2 Chemistry Msu Edu Courses Cem151 Exam2 14 Ans Pdf

Writing And Balancing Chemical Equations Introductory Chemistry Lecture Lab

Net Ionic Equations

Solved Write Balanced Ionic Equations For The Fol



