Modern Periodic Table Of Elements With Electronegativity

Periodic Trends In Electronegativity Ck 12 Foundation
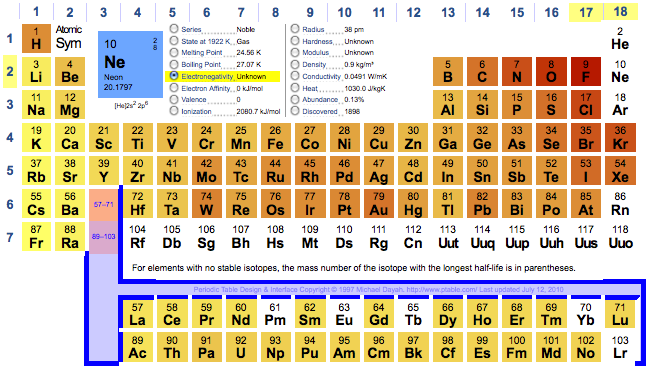
Internet Database Of Periodic Tables Chemogenesis

Electronegativity Definition Oxidation Number Non Polar Bond

The Parts Of The Periodic Table
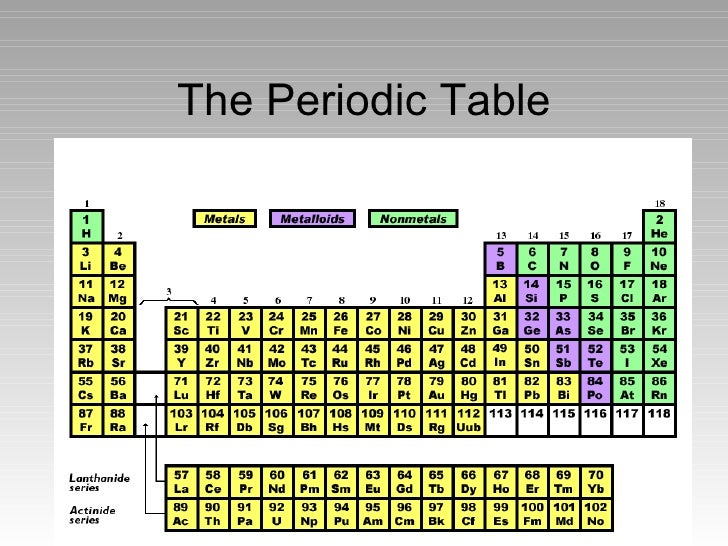
The Periodic Table

Which Element Is Highest In Electronegativity Which Element Is Lowest In Electronegativity Ppt Download
2) Nonmetals are located on the right side of the periodic table 3) Metalloids constitute some border line elements and are shown by zigzag line Metallic Character The metallic character of the elements is due to their electron releasing tendency By losing electrons, metal atoms change to positive ions (cations) These are therefore.

Modern periodic table of elements with electronegativity. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structureThe chemical symbol for Hydrogen is H With a standard atomic weight of circa 1008, hydrogen is the lightest element on the periodic table Its monatomic form (H) is the most abundant chemical substance in the Universe, constituting roughly 75% of all baryonic mass. Electronegativity falls as we go down the periodic table The electronegativity of boron and aluminium are and 15 respectively and the electronegativity of beryllium and boron is 15 and respectively The increase from group 2 to group 3 is formed by the fall as we go down Group 3 from Boron to aluminium. •Elements can be divided into groups, or families ELEMENT FAMILIES ELEMENT FAMILIES • Each column of the periodic table contains one element family • Hydrogen is usually considered separately, so the first element family begins with lithium and sodium in the first column • Just as human family members often have similar looks and traits, members of element families have similar.
Click here to buy a book, photographic periodic table poster, card deck, or 3D print based on the images you see here!. RS Mulliken proposed an electronegativity scale in which the Mulliken electronegativity, Χ M is related to the electron affinity EA v (a measure of the tendency of an atom to form a negative species) and the ionization potential IE v (a measure of the tendency of an atom to form a positive species) by the equation Χ M = (IE v EA v)/2 The subscript v denotes a specific valence state. Explore each Elements Electronegativity in one interactive Periodic Table Explore all 118 elements and their Electronegativity numbers.
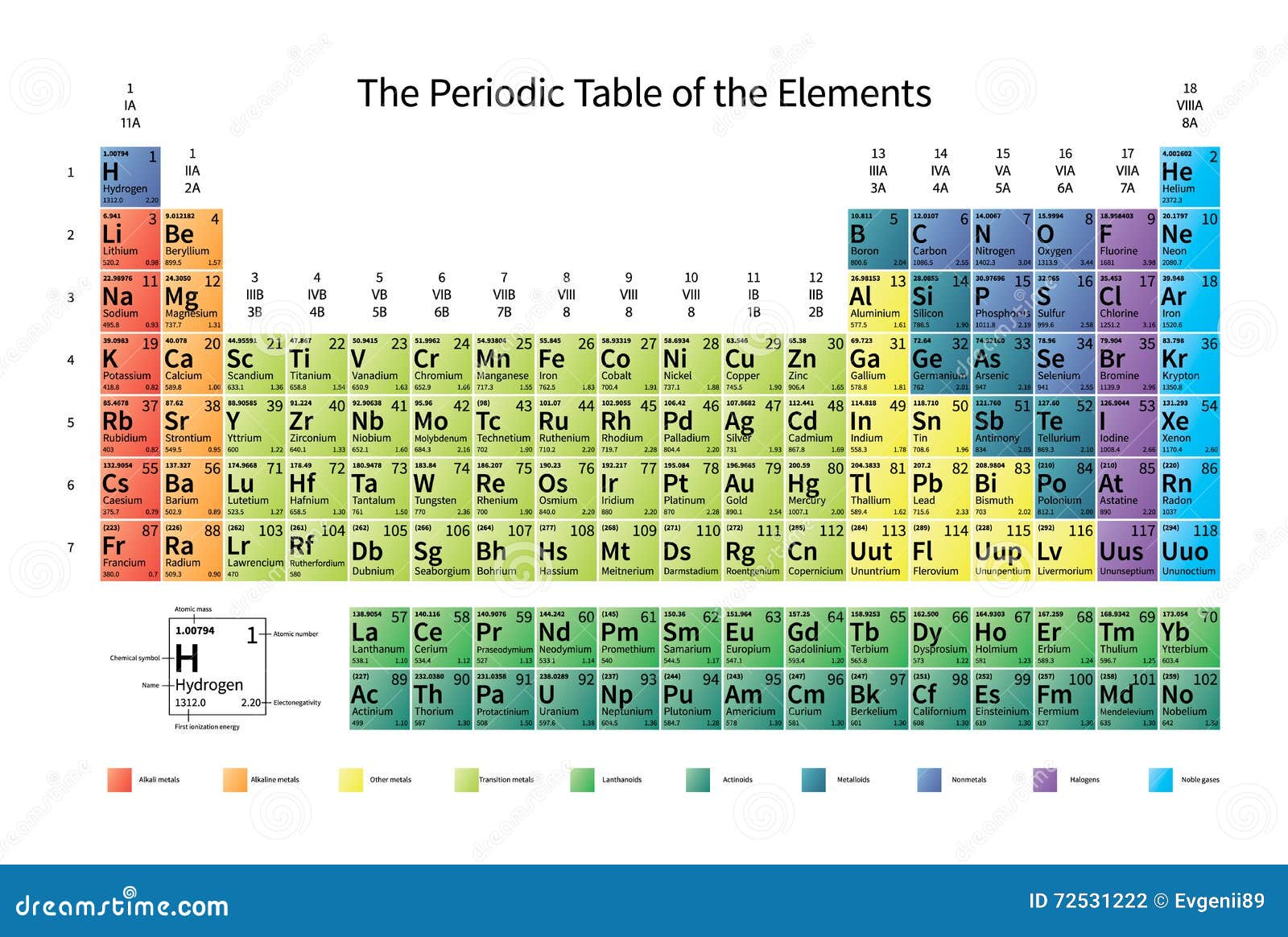
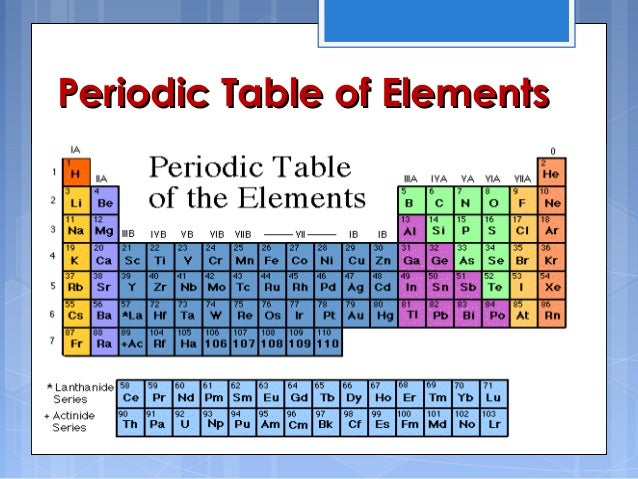
Two major Factors controlling the electronegativity of elements in the periodic table 1) Size of the element in the periodic table 2)Position of element in periodic table means closeness to noble gases Electronegativity trends part 2 video will help you in understanding the variation of electronegativity in periodic table. The position of certain elements in the Modern Periodic Table is shown below Oxygen has a high electronegativity, due to which it has a higher tendency to attract electrons rather than donating them (iii) Element D is a metal with valency 2 Element D is magnesium, due to its low electronegativity it has a very high tendency to donate. Modern Periodic Table Of Elements In 1913, Moseley proved that the atomic number is a very important property of an element After that, Neils Bohr made the modern periodic table using the atomic number According to the modern periodic classification, the properties of elements are the periodic function of their atomic numbers.
Modern Periodic Law states that properties of elements are a periodic function of their atomic number Q How does the electronegativity of elements relate to their metallic or nonmetallic character?. Periodic Table of Elements Printable Periodic Table With Electronegativity Electronegativity is also one of the topics which students must know if he or she is studying chemistry because without knowing the electronegativity one cannot come to know whether the particular element is electron, proton, or neutron. The modern periodic table, grouped by atomic number, or how many protons are in the nucleus of an atom of an element Dimitri Mendeleev’s table original, complete with spaces for missing elements.
2) Nonmetals are located on the right side of the periodic table 3) Metalloids constitute some border line elements and are shown by zigzag line Metallic Character The metallic character of the elements is due to their electron releasing tendency By losing electrons, metal atoms change to positive ions (cations) These are therefore. The Periodic Table of the Elements is useful for revealing patterns and trends in the elements Which statement accurately describes a pattern in the size of atomic radii in the Periodic Table of the Elements?. Seeing chemical elements arranged in the modern periodic table is as familiar as seeing a map of the world, but it was not always so obvious The creator of the periodic table, Dmitri Mendeleev , in 1869 began collecting and sorting known properties of elements, like he was playing a game, while traveling by train.
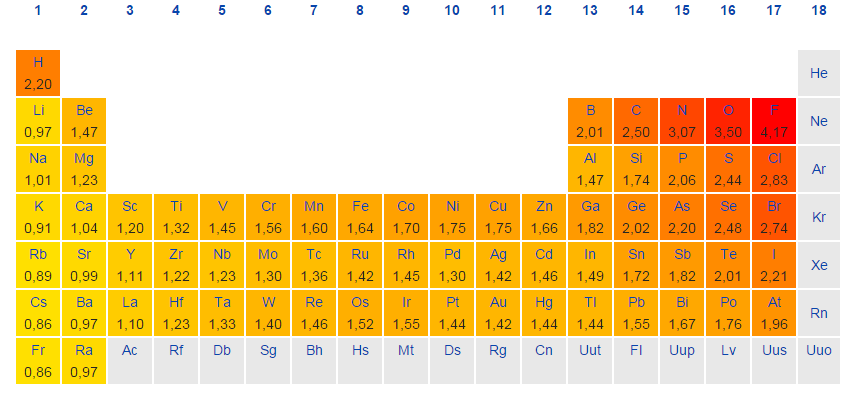
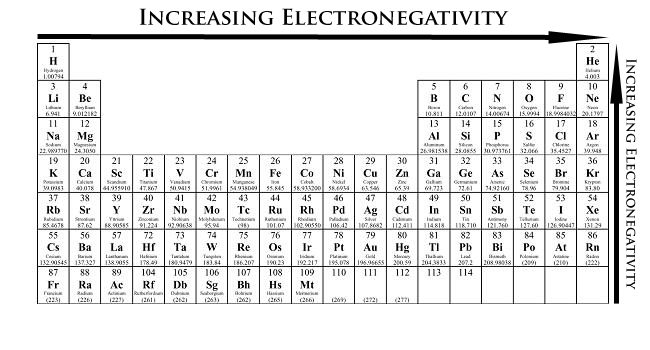
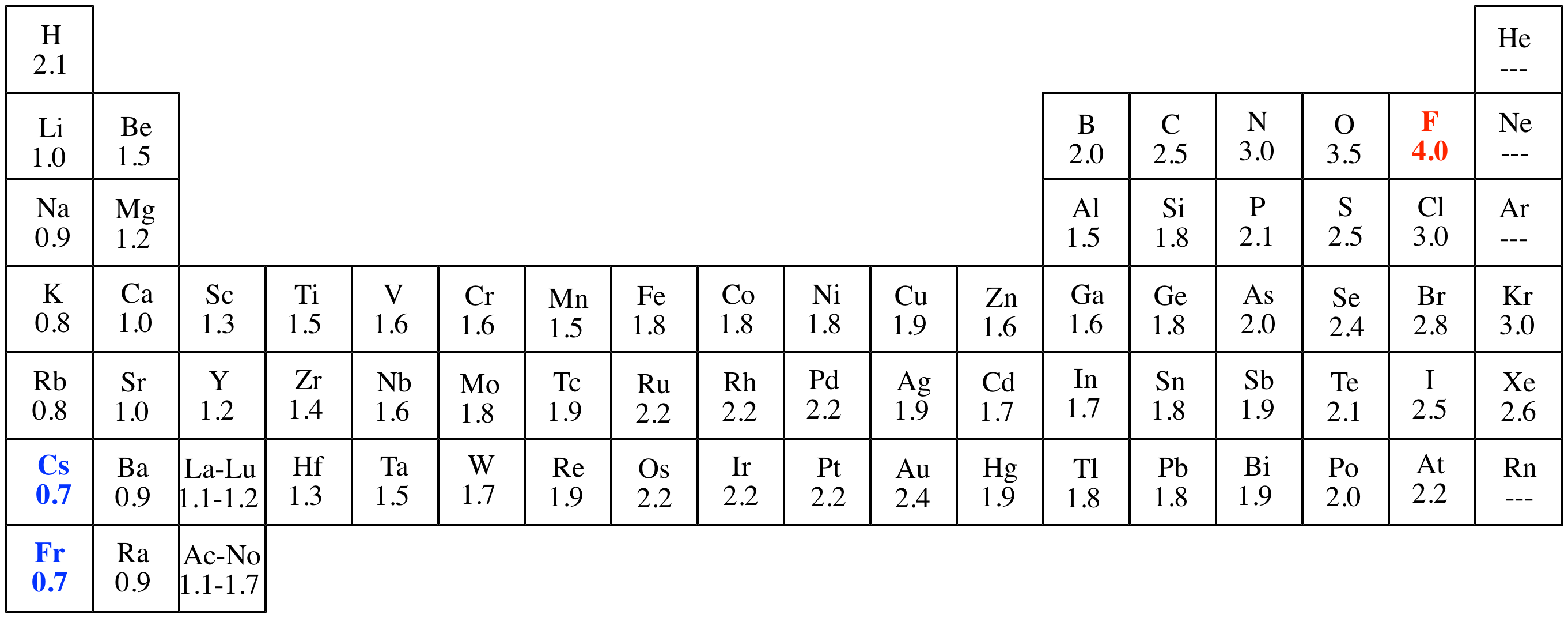
Ch 5 The Periodic Table Video #1 Ch 5 The Periodic Table Video #1 by Ms McAuley 2 years ago 15 minutes 278 views Introduction to, Chapter 5 , Notes, pp 12 The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity by Professor Dave. Values for electronegativity run from 0 to 4 Electronegativity is used to predict whether a bond between atoms will be ionic or covalent It can also be used to predict if the resulting molecule will be polar or nonpolar This table is a list of electronegativity values of the elements. Electronegativity is a measure of the ability of an atom to attract the electrons when the atom is part of a compound Electronegativity values generally increase from left to right across the periodic table Electronegativities generally decrease from top to bottom of a group The highest electronegativity value is for fluorine.
The periodic table supports an understanding of the modern atomic model Several properties of the elements exhibit definite periodic trends, that is, atomic size, ionization energy, and electronegativityThe periodic properties of the elements suggest that elements with similar chemical properties have similar arrangements of electrons. Values for electronegativity run from 0 to 4 Electronegativity is used to predict whether a bond between atoms will be ionic or covalent It can also be used to predict if the resulting molecule will be polar or nonpolar This table is a list of electronegativity values of the elements. Common Properties Abundance in Earth's Crust.
Electronegativity is the power or tendency to attract a pair of electrons that are being shared in a bond The most electronegative element in the periodic table is fluorine followed by nitrogen. Which element has the greatest ionization energy and electronegativity?. In the modern periodic table, the electronegativity of elements increases across a period (row) and decreases down a group (column) Therefore, the bottomleft most element (francium) is predicted to have the lowest electronegativity and the topright most element (fluorine) is predicted to have the highest electronegativity.
According to Pauling scale fluorine has the highest electronegativity in the periodic table and its value is 40, whereas caesium and francium are the least electronegative elements with values on the Pauling scale as low as 07. Each one of the rows of the Modern Periodic Table is called period If you take a look at the elements in one row, they all have the same number of atomic orbitals What Are Groups In The Modern Periodic Table?. The definitive online periodic table reference site including technical data, and photographs and descriptions of thousands of samples of the chemical elements Click any element below to see more Newest samples 28 October, 17 What you'll find on this site My Popular Science Column.
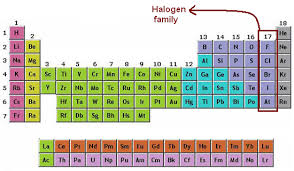
•Elements can be divided into groups, or families ELEMENT FAMILIES ELEMENT FAMILIES • Each column of the periodic table contains one element family • Hydrogen is usually considered separately, so the first element family begins with lithium and sodium in the first column • Just as human family members often have similar looks and traits, members of element families have similar. A tabular arrangement of elements in groups and periods which highlights the regular trends in properties of elements is defined as the periodic table There are eighteen vertical columns known as groups in the modern periodic table which are arranged from left to right and seven horizontal rows which are known as periods. Answer choices fluorine chlorine bromine iodine.
View CEM_periodic tablepdf from CEM 10 at Belmont University The Modern Periodic Table of the Elements 1 0 1 18 Hydrogen 1 Average relative masses are 01 values, rounded to two decimal. Each value is given for the most common and stable oxidation state of the element See also Electronegativities of the elements (data page) ^ The electronegativity of francium was chosen by Pauling as 07, close to that of caesium (also assessed 07 at that point). Electronegativity is the power or tendency to attract a pair of electrons that are being shared in a bond The most electronegative element in the periodic table is fluorine followed by nitrogen.
Elements on the periodic table are arranged based on their electronic configurations Because of this, where an element is on the periodic table can tell you about its chemical properties Elements in the same group (column) have the same number of outer electrons, while Elements in the same period (row) have the same number of electron shells. Each value is given for the most common and stable oxidation state of the element See also Electronegativities of the elements (data page) ^ The electronegativity of francium was chosen by Pauling as 07, close to that of caesium (also assessed 07 at that point). Sometimes it's nice to have a paper version of the periodic table of the elements that you can refer to when working problems or doing experiments in the lab This is a collection of periodic tables that you can print and use Note For 19 values featuring all 118 elements, more free printable periodic tables are also available.
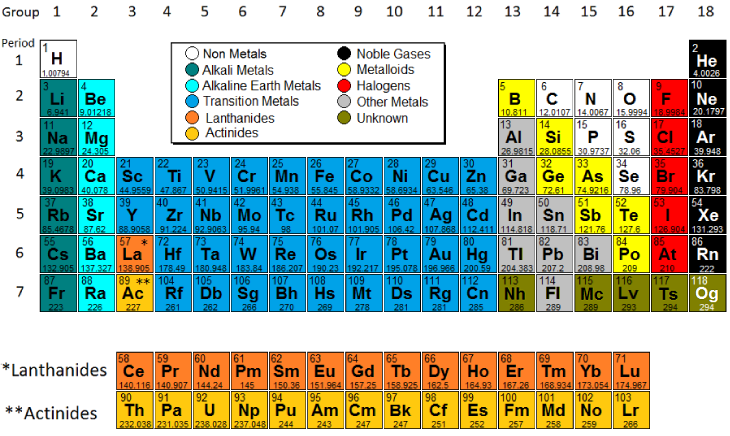
In modern periodic table is based on modern periodic law in which elements are arranged in increasing order of their atomic numbers In the modern periodic table, the elements are arranged in rows and columns These rows and columns are known as periods and groups respectively The table consists of 7 periods and 18 groups. On the periodic table, Fluorine with the value of 40 is the most electronegative element, whereas francium and cesium with the value of 07 is the least electronegative element In this article, we will discuss applications of electronegativity, nonpolar and polar bonds, and oxidation number. Modern Periodic Table Of Elements In 1913, Moseley proved that the atomic number is a very important property of an element After that, Neils Bohr made the modern periodic table using the atomic number According to the modern periodic classification, the properties of elements are the periodic function of their atomic numbers.
Illustration about Periodic table of elements with electronegativity values Illustration of trend, properties, electropositive. Group 14 of the Periodic Table consists of the elements carbon (C), silicon (Si), germanium (Ge), tin (Sn), & lead (Pb) Properties of Group 14 of Periodic Table elements • This group has a mixed type of element with the nonmetal carbon, two metalloids, and two metals The common characteristic is four valence electrons. 1) Elements on the modern Periodic Table are arranged in order of increasing (a) Atomic mass (b) Atomic number (c) Number of neutrons (d) Number of valence electrons 5 As the elements of Group 17 are considered in order of increasing atomic number, there is an increase in (1) Atomic radius (2) Electronegativity (3) First ionization energy (4) Number of electrons in the first shell 7) In the.
Ch 5 The Periodic Table Video #1 Ch 5 The Periodic Table Video #1 by Ms McAuley 2 years ago 15 minutes 278 views Introduction to, Chapter 5 , Notes, pp 12 The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity by Professor Dave. This free periodic table is colorcoded to indicate the electronegativity of an atom of an element Electronegativity is a trend naturally exhibited in the periodic table, but as you can see, it’s not a hardandfast trend Use the table to write the names of compounds and to predict chemical reactions. Mendeleev made an early periodic table In the modern periodic table, elements are in order of atomic number in periods and groups Electronic structures model how electrons are arranged in atoms.
Seeing chemical elements arranged in the modern periodic table is as familiar as seeing a map of the world, but it was not always so obvious The creator of the periodic table, Dmitri Mendeleev , in 1869 began collecting and sorting known properties of elements, like he was playing a game, while traveling by train. PERIODIC TABLE OF ELEMENTS Title Ptablecom Periodic Table Author Michael Dayah Created Date 2/6/17 1235 AM. Nonmetal boxes are located above the diagonal line, and they are included in the red, gold, and green boxes on the Periodic Table Please notice that even though hydrogen is colored with green, it is a metal However, this is the only exception Nonmetals are a very interesting category of elements of the modern periodic table.
On the basis of their positions in the periodic table, arrange Cl, Se, Si, and Sr in order of increasing electronegativity and classify each as a metal, a nonmetal, or a semimetal Given four elements Asked for order by increasing electronegativity and classification Strategy Locate the elements in the periodic table. 1) Elements on the modern Periodic Table are arranged in order of increasing (a) Atomic mass (b) Atomic number (c) Number of neutrons (d) Number of valence electrons 5 As the elements of Group 17 are considered in order of increasing atomic number, there is an increase in (1) Atomic radius (2) Electronegativity (3) First ionization energy (4) Number of electrons in the first shell 7) In the. While a row is called a period, each one of the columns in the Modern Periodic Table is called a Group Just like it happened with the.

Free Printable Periodic Tables Pdf And Png Science Notes And Projects
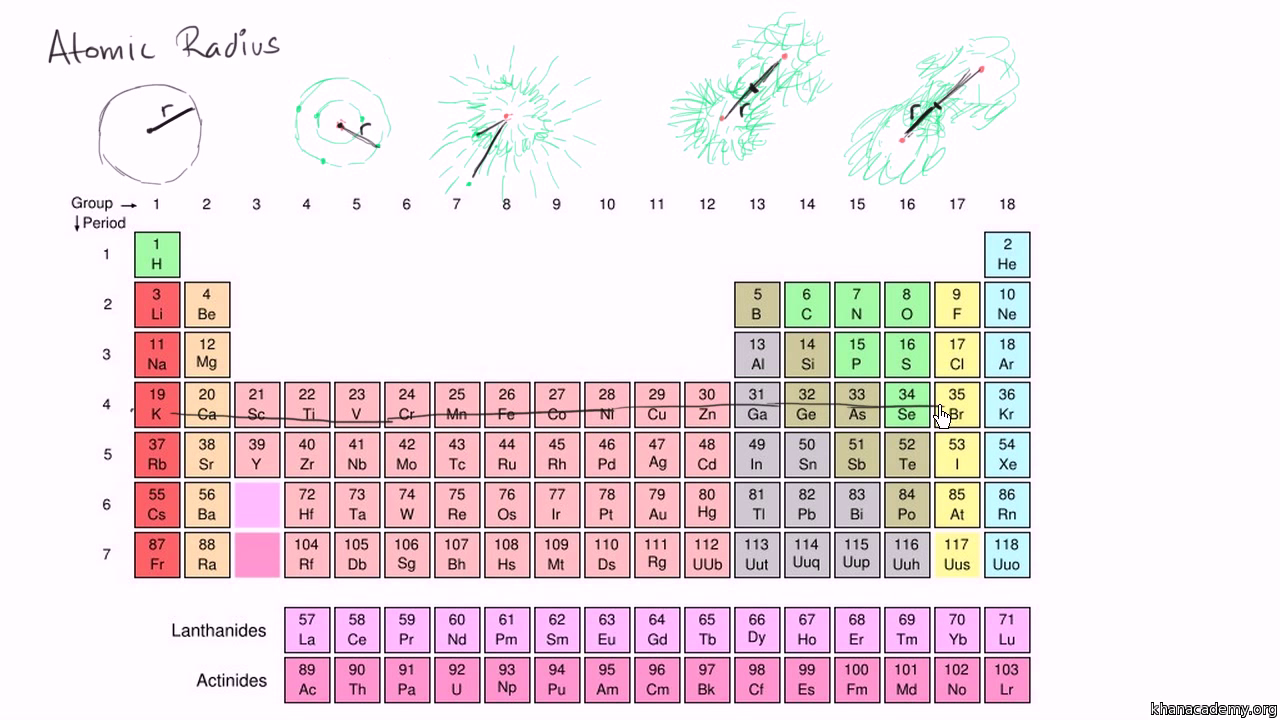
Periodic Table Learn Atomic Structure Periodic Trends Khan Academy

Definition Of Electronegativity Chemistry Dictionary
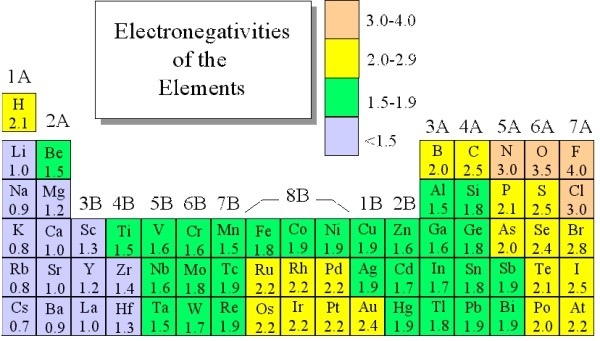
Electronegativity Chart Of Elements List Of Electronegativity
Free Printable Periodic Table Of Elements Download

Free Printable Periodic Tables Pdf And Png Science Notes And Projects

Historical Development Of Periodic Table Mendeleev Doberiener Videos

Chemistry Lesson 1 Instruction Page 1
Bright Colorful Periodic Table Of The Elements With Atomic Mass Electronegativity And 1st Ionization Energy On White Stock Vector Illustration Of Chemistry Physics
Periodic Table Model Science Software

Periodic Table Of Elements Electronegativity Chart

Which Is The Element In Periodic Table Having Highest Electronegativity Quora

Periodic Trends In Electronegativity Valency Metallic Character Chemistry Iitjee Main Askiitians Youtube

Electronegativity Of The Elements

Periodic Table Accessscience From Mcgraw Hill Education

Webelements Periodic Table Periodicity Electronegativity Allen Periodic Table Gallery
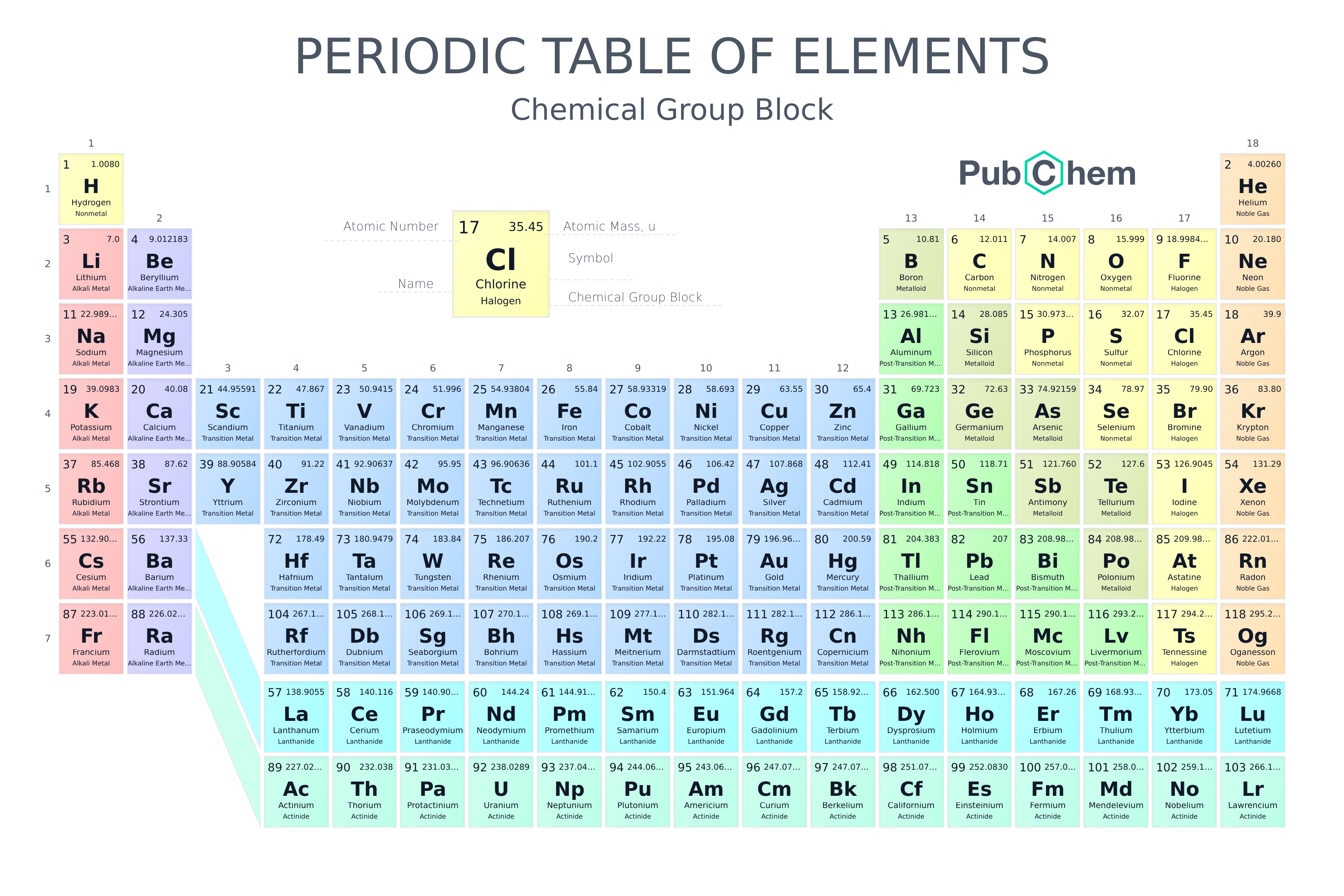
Periodic Table Of Elements Pubchem
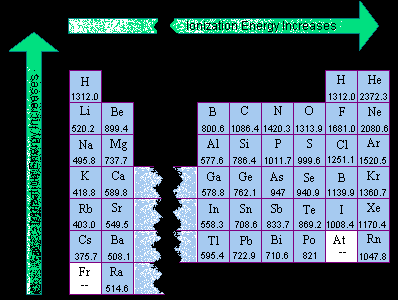
Periodic Table Electronegativity Noble Gases 12

Periodic Table Blocks Of Elements

Electronegativity Periodic Table Webelements Periodic Table Of The Elements Periodic Table Science Symbols

Graduation Of Elements In The Modern Periodic Table Ppt Download

The Parts Of The Periodic Table
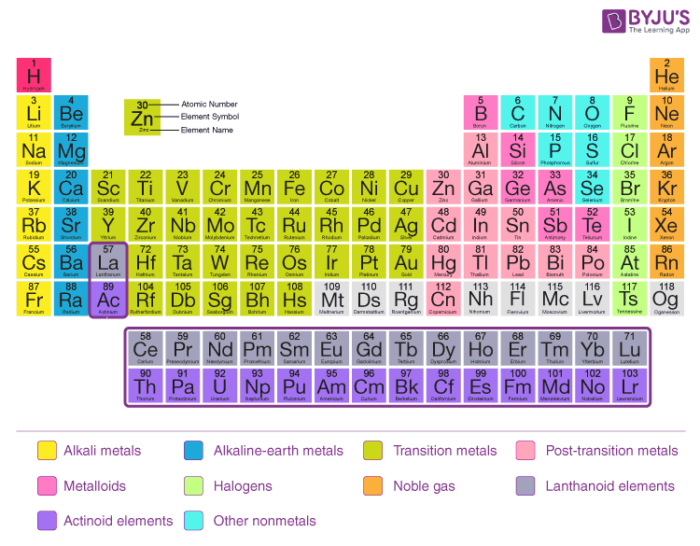
Modern Periodic Law With Detailed Periodic Classification Of Elements

List Of Electronegativity Values Of The Elements
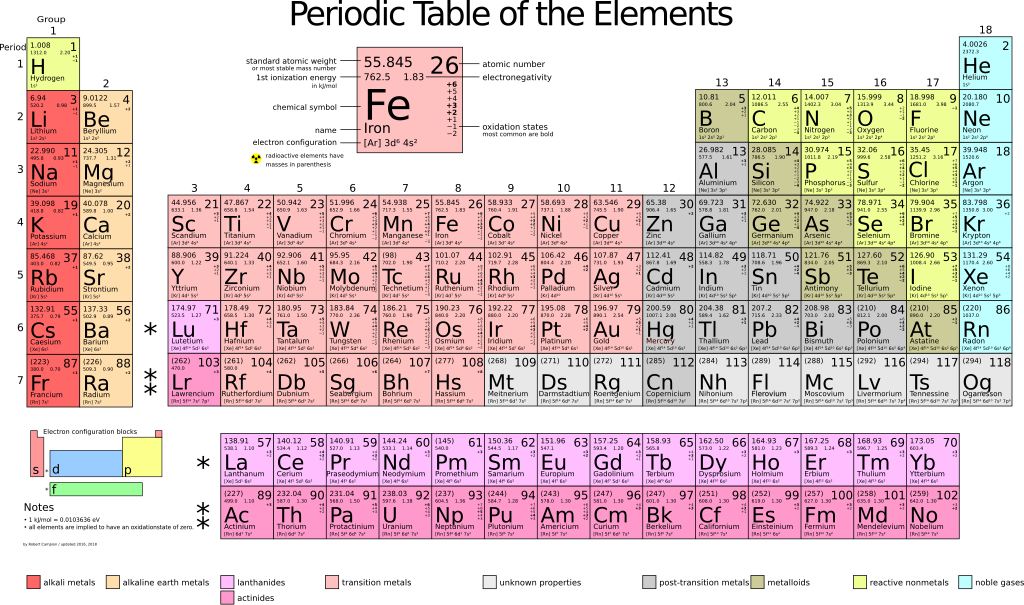
File Periodic Table Large Svg Wikimedia Commons

Table 2 Electronegativity Pauling Scale Periodic Table Of The Elements Periodic Table Projects To Try
Trends In The Periodic Table

العلوم Lesson 2 Graduation Of Properties Of Elements In The Modern Periodic Table First Part

Periodic Table Of The Elements Updated 21 Definition Groups
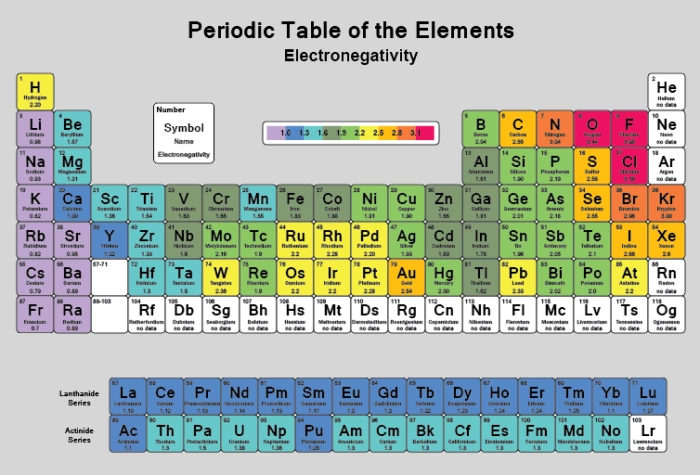
Electronegativity

Periodic Table Wikipedia
3
Periodic Table Development And Trends

The Periodic Table Of The Elements Trends In Atomic Radius Electronegativity Ionization Energy Density And Specific Heat

Periodic Table Electronegativity High Res Stock Images Shutterstock
Periodic Trends Chemistry Libretexts
Halogens Fluorine Chlorine Bromine Iodine Astatine
Illustrated Glossary Of Organic Chemistry Electropositive

Trends The Periodic Table Of Elements
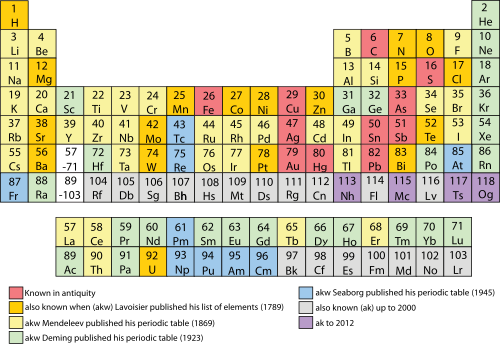
Periodic Table Wikipedia

Periodic Table Z Element Copy Elements Electronegativity Periodic Table Stable Electron Configuration Hd Png Download Transparent Png Image Pngitem
1
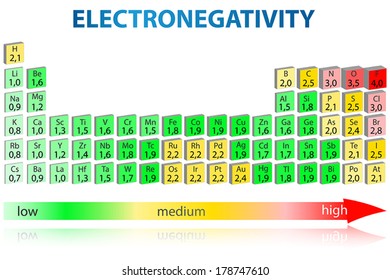
Periodic Table Electronegativity High Res Stock Images Shutterstock
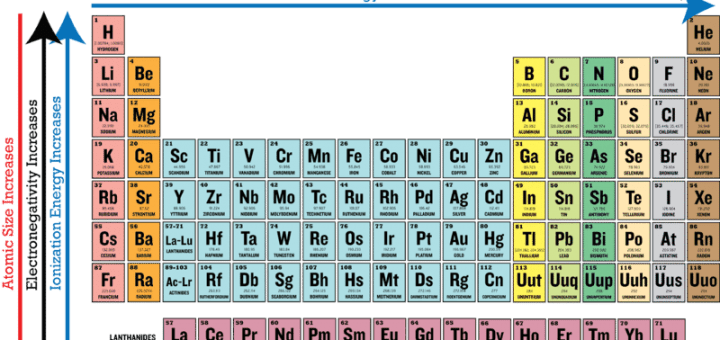
Difference In Electronegativity Science Online

10 New Periodic Table Hd Wallpaper Full Hd 1080p For Pc Desktop Periodic Table Of The Elements Periodic Table Printable Chemistry Periodic Table
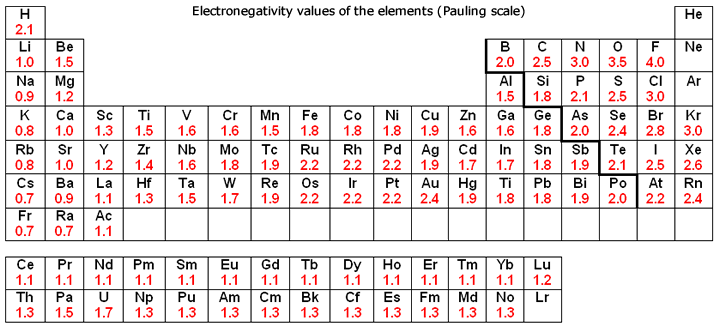
Which Elements Have The Highest Electronegativities On The Periodic Table Socratic

Alkali Metal Definition Properties Facts Britannica
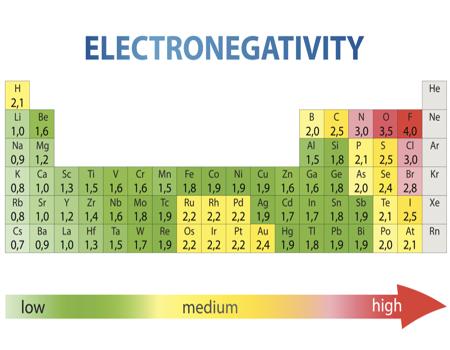
Electronegativity Chart Of Elements List Of Electronegativity
/PeriodicTableElectronegativity-56a12a045f9b58b7d0bca77c.jpg)
What Is Electronegativity And How Does It Work
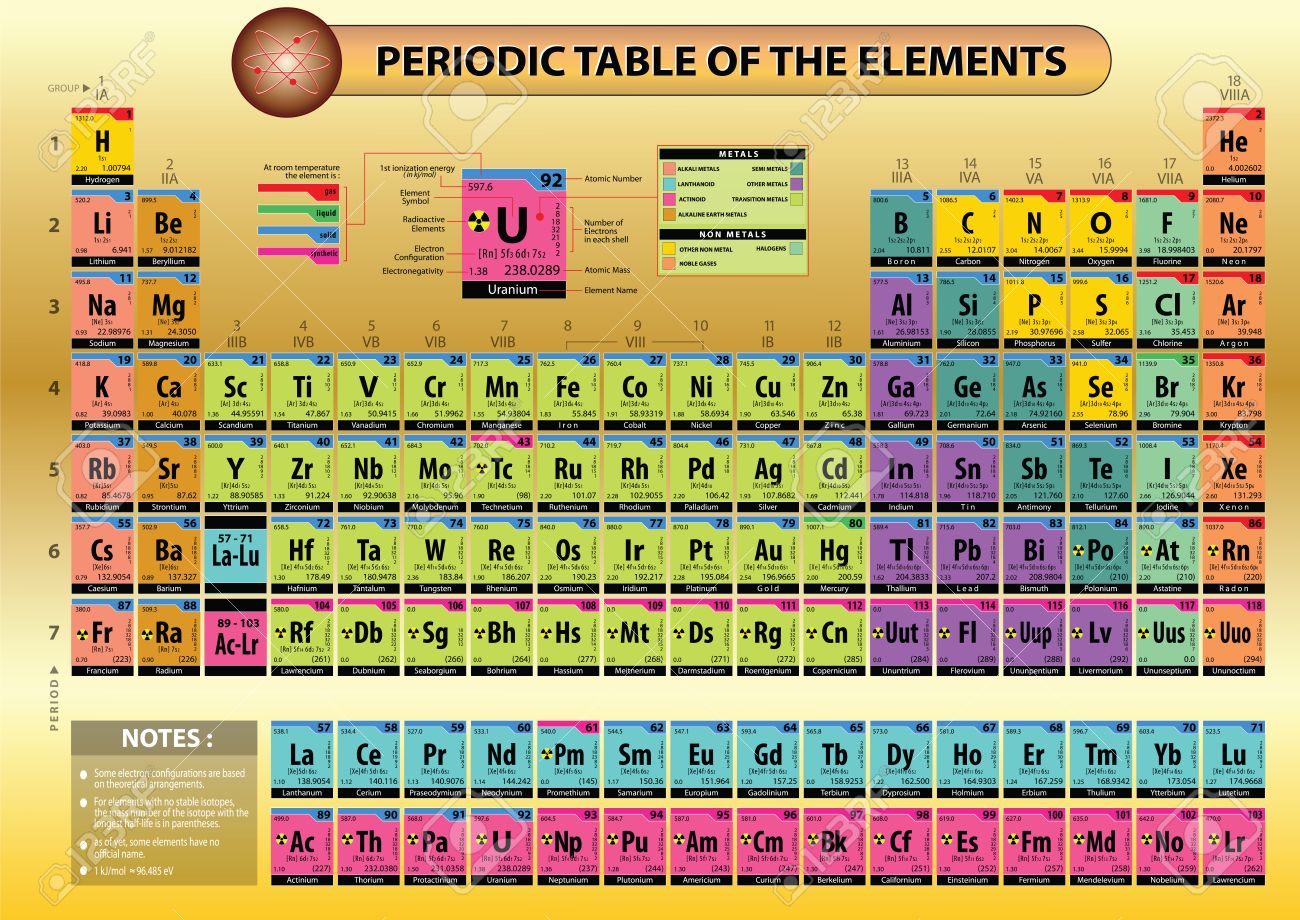
Modern Periodic Table Of Elements With Names And Symbols

Spring Bis2a Facciotti Lecture 02 Biology Libretexts
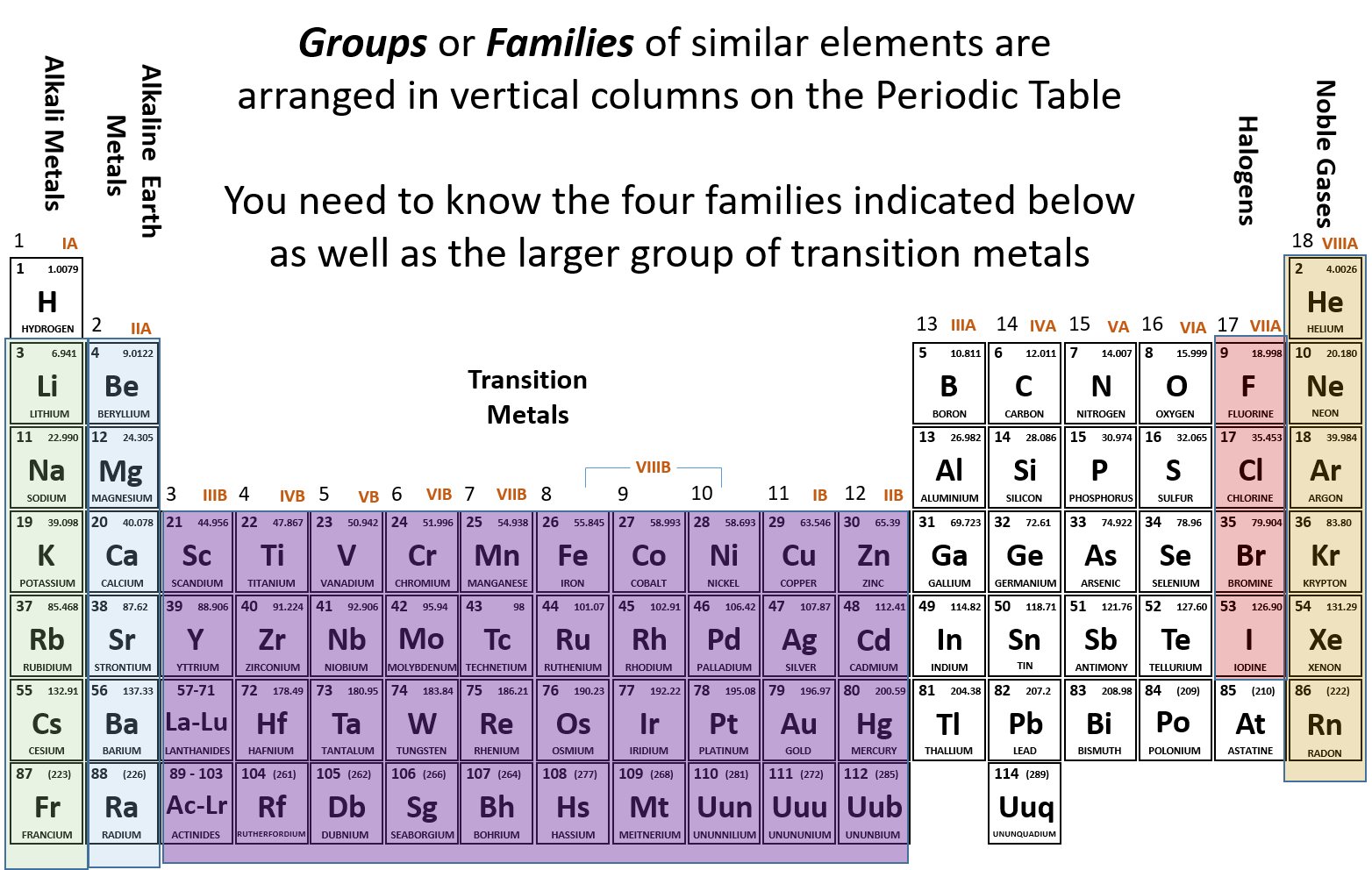
Ch150 Chapter 2 Atoms And Periodic Table Chemistry
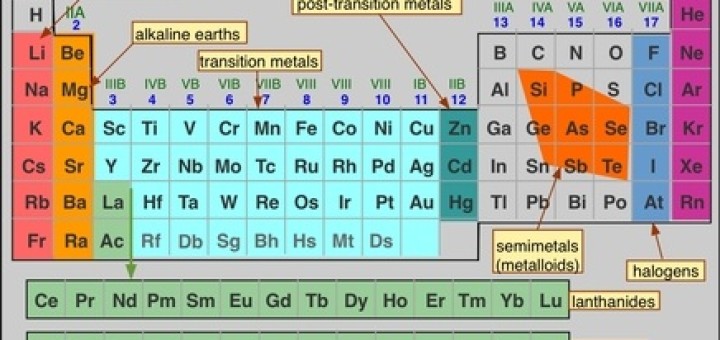
The Graduation Of The Metallic And Nonmetallic Property In The Modern Periodic Table Science Online

Periodic Trends Electronegativity Chemistry For Non Majors

Electronegativity Chart Of Elements List Of Electronegativity
Q Tbn And9gctm94tw5zrgv8npf5ukoynwstqdkx0tocouzbqb0lw3j L5 Sao Usqp Cau

Classification Of Elements Periodicity In Properties Askiitians

Periodic Table Of The Elements Sigma Aldrich
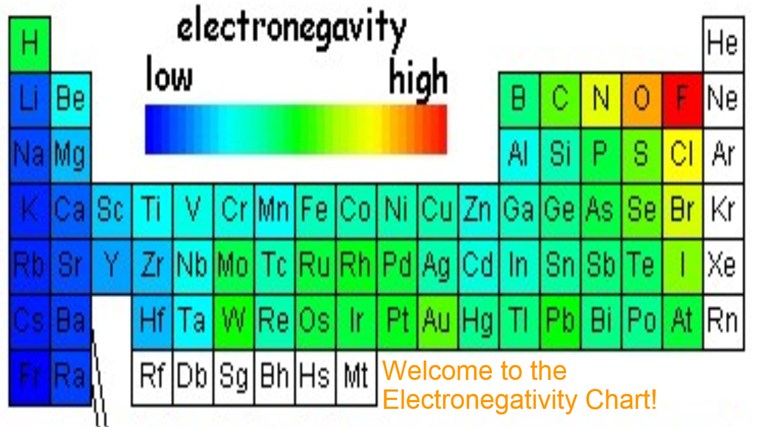
Electronegativity Chart Of Elements List Of Electronegativity
Which Group On The Periodic Table Is The Most Electronegative Quora
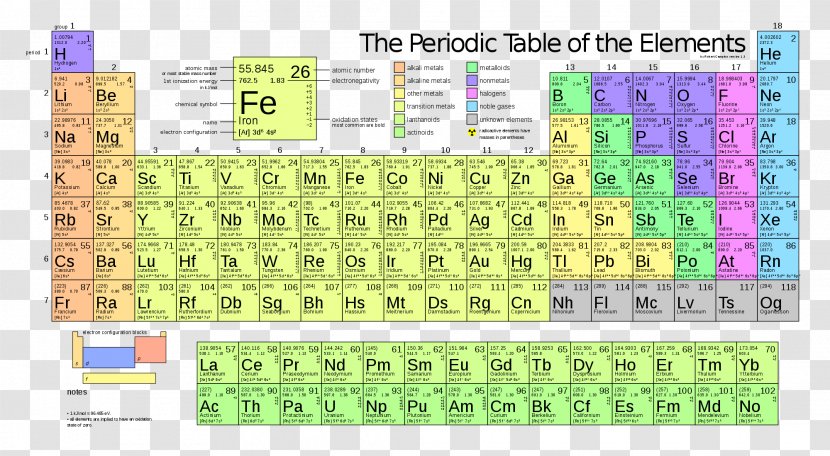
Periodic Table Chemical Element Ionization Energy Electron Configuration Atom Dynamic Elements Transparent Png

Periodic Trends Electronegativity
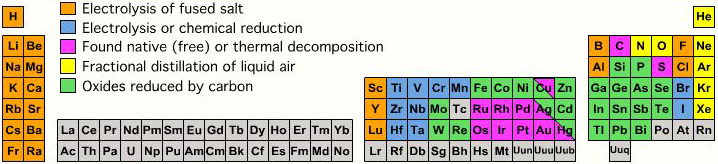
Internet Database Of Periodic Tables Chemogenesis

The Arrangement Of The Elements The Periodic Table Siyavula

Periodic Trends Wikipedia

Cir Room 9 October 10
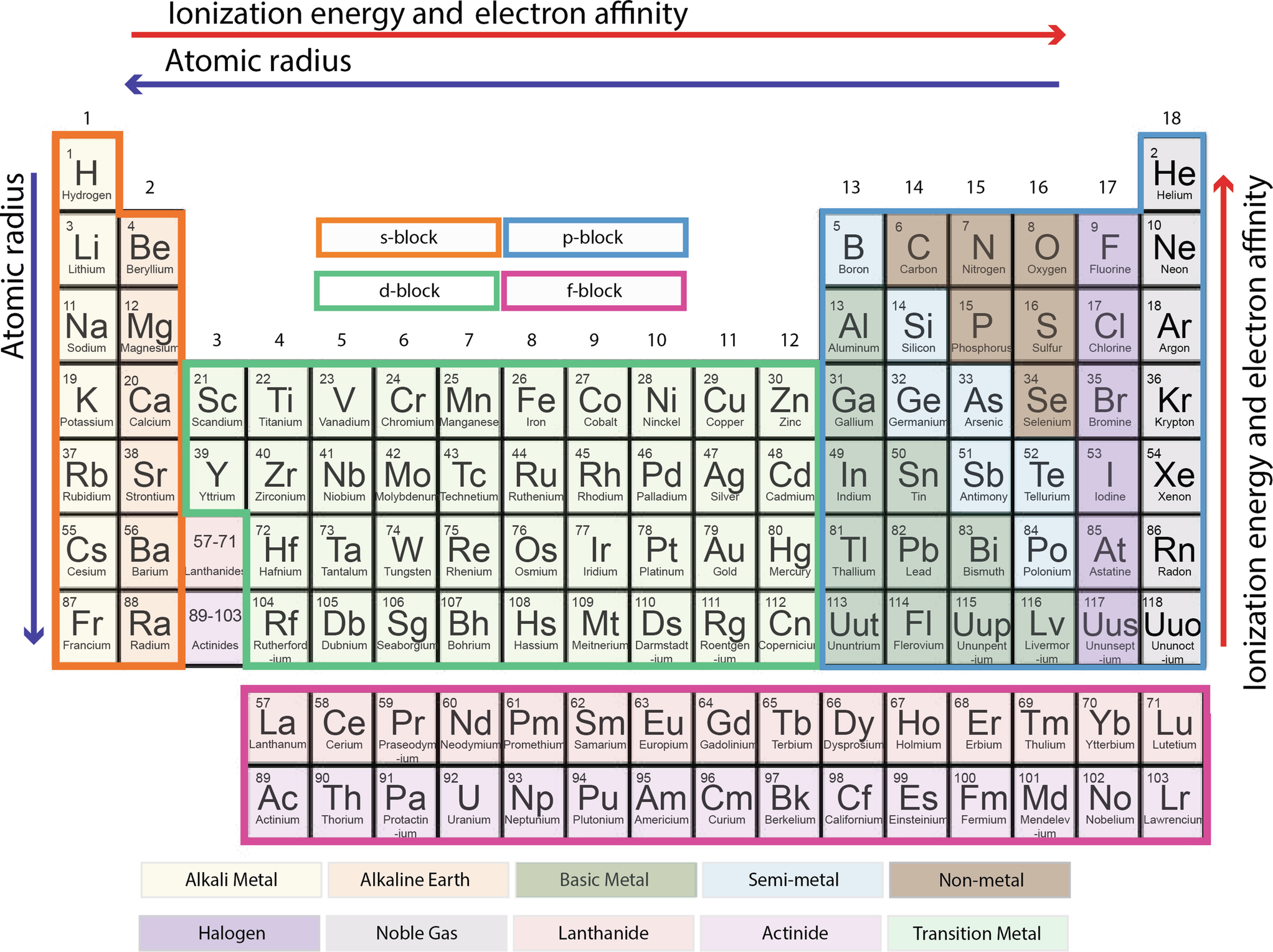
Periodic Table Springerlink

Electronegativity Chart Of Elements List Of Electronegativity

Periodic Table Of Elements Electronegativity Chart

Periodic Table Of Elements Hd Pdf

Modern Periodic Table Read User Generated Content Ck 12 Foundation
Http Www Sciencegeek Net Tables Electronegativity Pdf
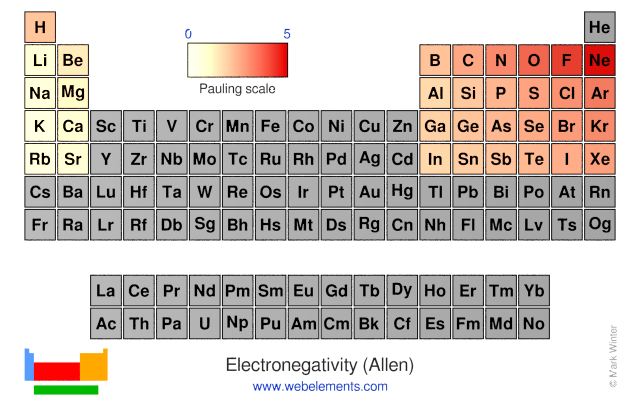
Webelements Periodic Table Periodicity Electronegativity Allen Periodic Table Gallery

Periodic Table Of The Elements Brilliant Math Science Wiki

Knowledgedoor
Free Printable Periodic Table Of Elements Download
/periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
Periodic Law Definition In Chemistry

Free Printable Periodic Tables Pdf And Png Science Notes And Projects

Modern Periodic Table Read User Generated Content Ck 12 Foundation
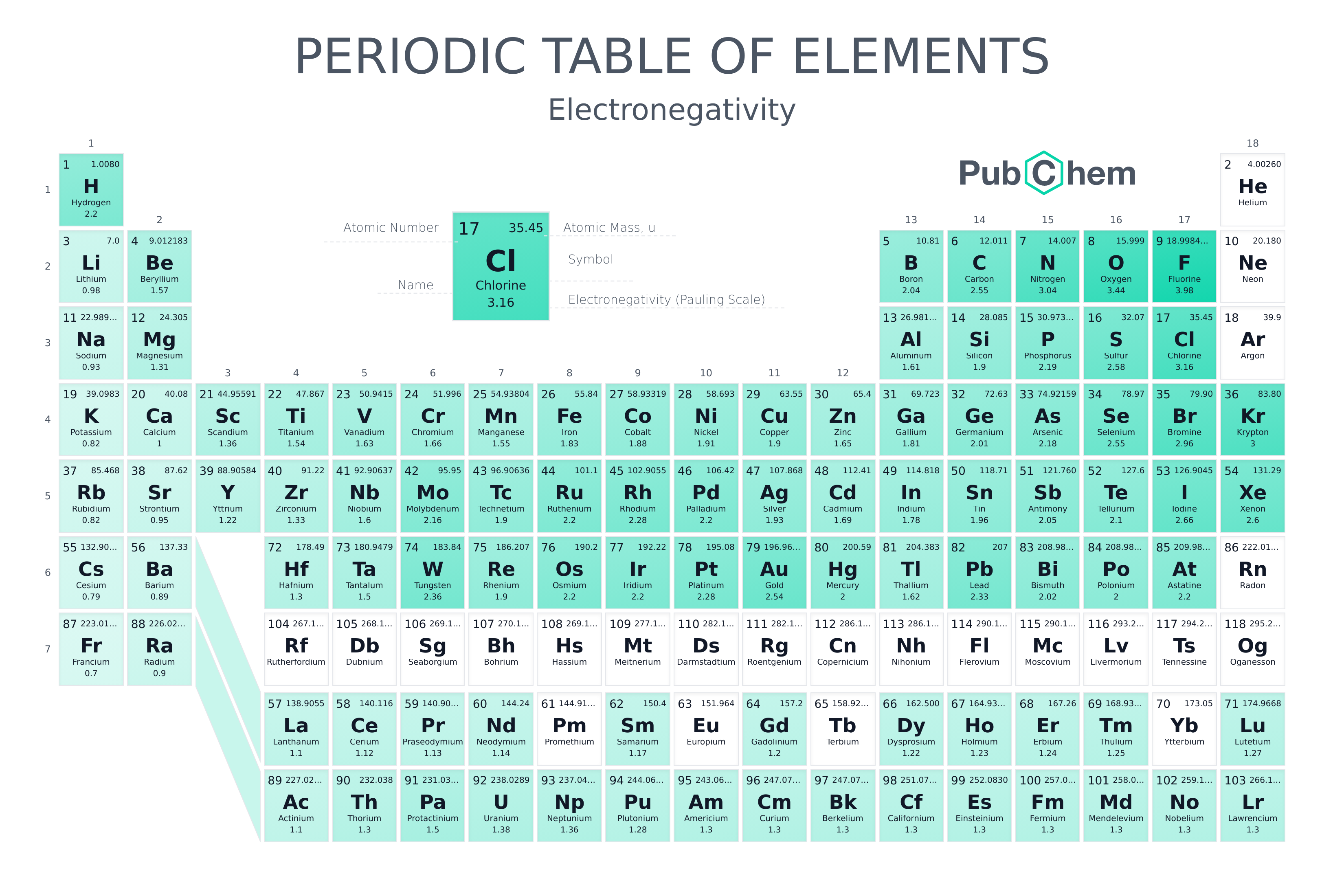
2 4 The Periodic Table Chemistry Libretexts
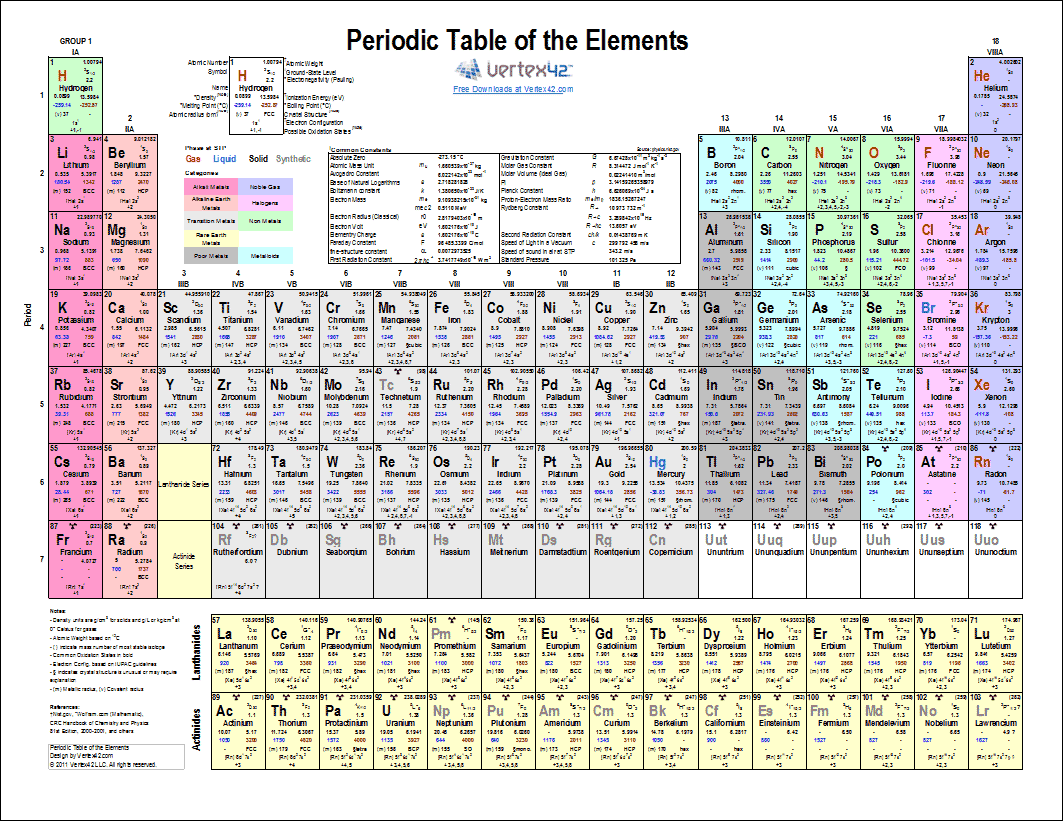
Printable Periodic Table Of Elements Chart And Data
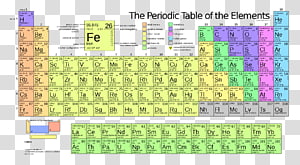
Periodic Table Chemical Element Electronegativity Table Transparent Background Png Clipart Hiclipart
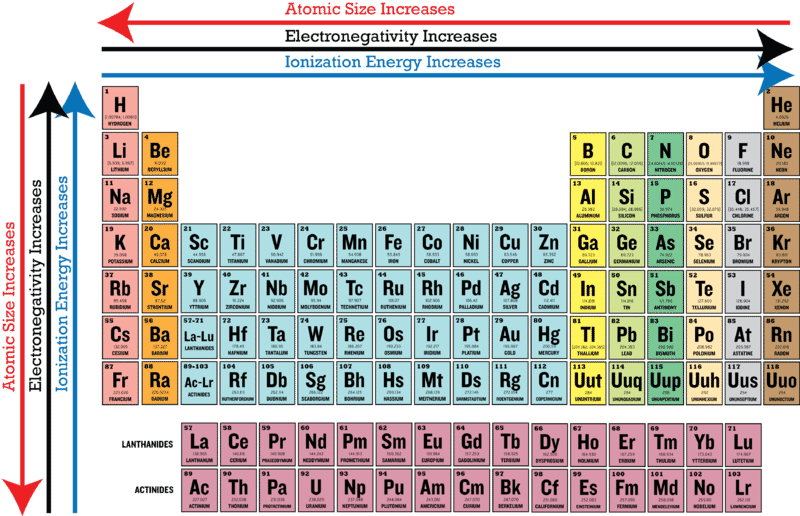
Periodic Trends In Electronegativity Ck 12 Foundation

The Parts Of The Periodic Table

Which Group On The Periodic Table Is The Most Electronegative Quora
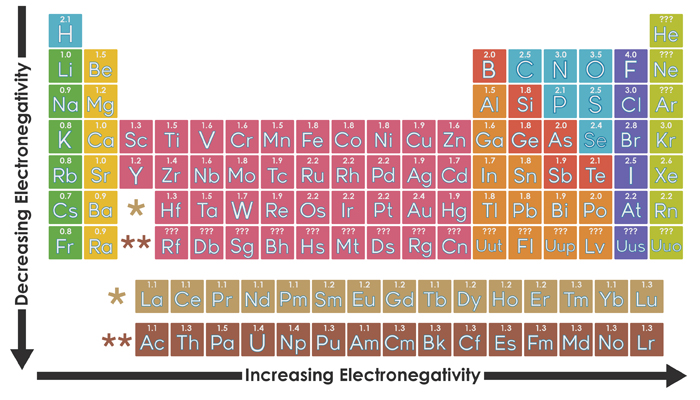
Organizing Atoms And Electrons The Periodic Table Annenberg Learner

What Is Electronegativity Green Planet Solar Energy For All

Electronegativity Definition Periodic Trends Effect On Bonding Faqs
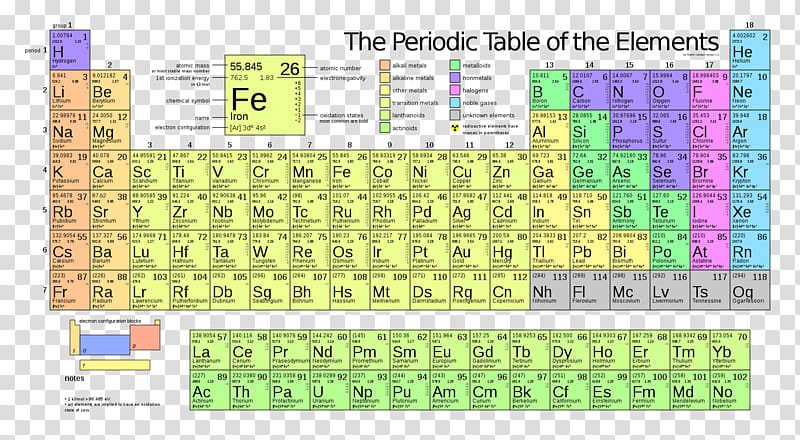
Periodic Table Chemical Element Electronegativity Table Transparent Background Png Clipart Hiclipart
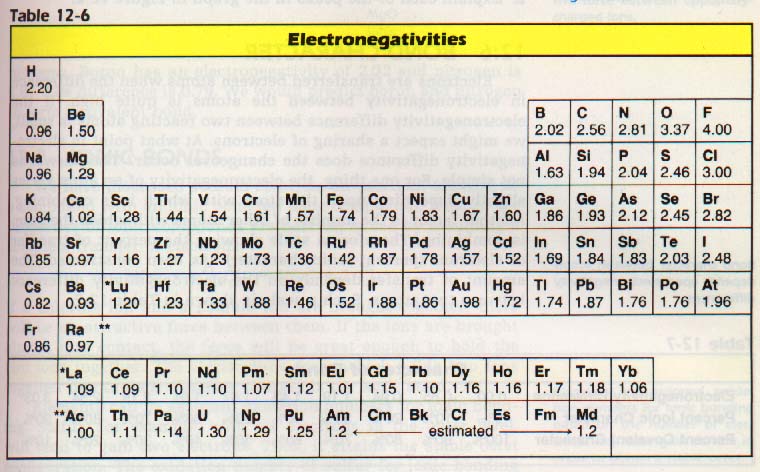
Ch 12
/PeriodicTableElectronegativity-56a12a045f9b58b7d0bca77c.jpg)
What Is Electronegativity And How Does It Work

Electronegativity
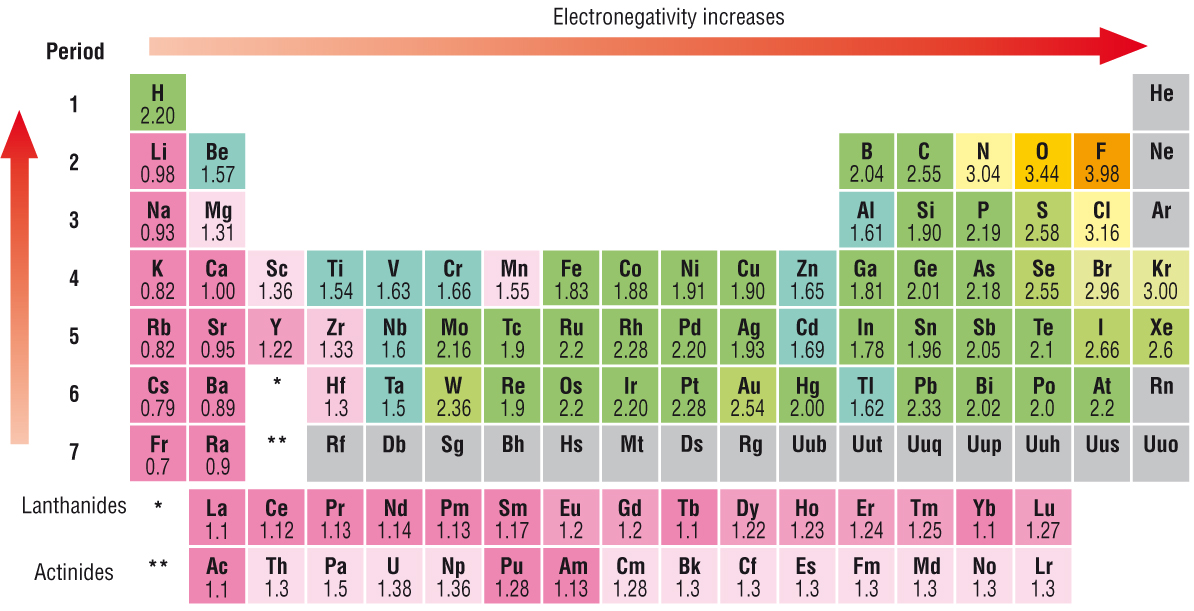
Electronegativity Chart Of Elements List Of Electronegativity

Periodic Table Of Elements Electronegativity Chart

23 Chemistry Ideas Chemistry Chemistry Lessons Teaching Chemistry
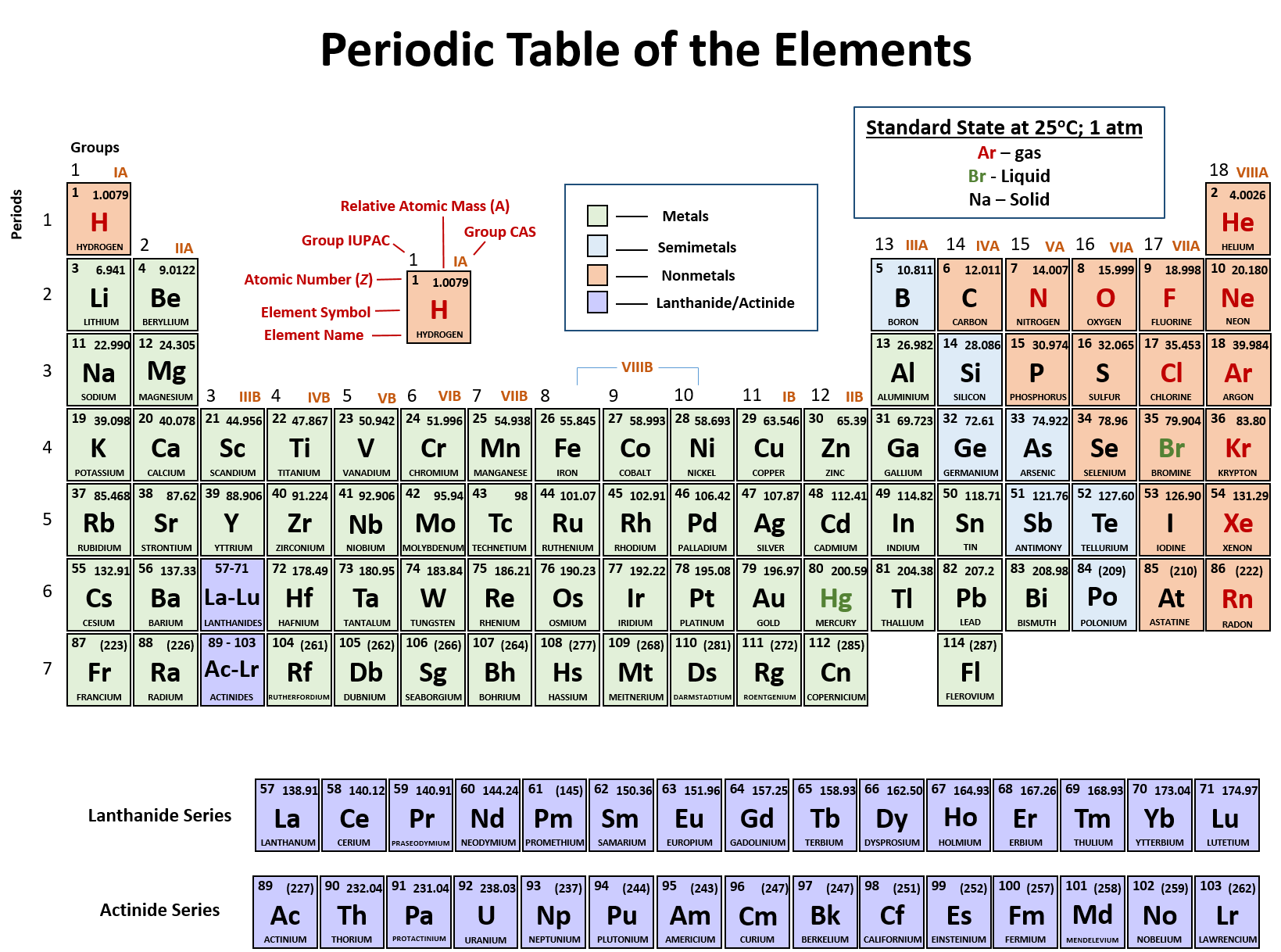
Ch150 Chapter 2 Atoms And Periodic Table Chemistry

Electronegativity Definition Trends Video Lesson Transcript Study Com



