Periodic Table Of Elements With Electronegativity And Ionization Energy Pdf
Periodic Trends Guided Inquiry Activity Chemical Education Xchange

Icse Solutions For Class 10 Chemistry The Periodic Properties And Their Variations A Plus Topper

Periodic Trends Boundless Chemistry

Periodicity Definition In Chemistry
Ionization Energy Wikipedia

1 Periodic Table Periodic Table Ion
Atomic Number Element Symbol Electron Shell Diagram Atomic Radius (pm) 1st Ionization Energy (kJ/mol) Electronegativity 2 POGIL™ Activities for High School Chemistry 6 Locate the numbers in Model 1 that represent the ionization energy The ionization energy is the amount of energy needed to remove an electron from an atom a.
Periodic table of elements with electronegativity and ionization energy pdf. Therefore it is highly. CS3L7 Periodic Trends (Ionization Energy and Electronegativity) SLT I can compare the periodic trends of ionization energy and electronegativity Brainstarter Part 2 Ionization Energy Part 3 Electronegativity What is the periodic trend for atomic radius?. There are specific reasons, you know Because of the way we organize the Ch 5 Section 51 History of the Periodic Table Ch 5 Section 51.
The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity by Professor Dave Explains 5 years ago 7 minutes, 53 seconds 1,860,673 views Why is the , periodic table , arranged the way it is?. By what property did Moseley suggest that the periodic table be arranged?. Electronegativity symbols of elements ionization energy atomic radius ionic radius The atomic radius of F, Br, and I are 64, 114, and 138 pm respectively From this information (and not your book) estimate a reasonable atomic radius of Cl.
Chemical elements listed by ionization energy The elements of the periodic table sorted by ionization energy click on any element's name for further information on chemical properties, environmental data or health effects This list contains the 118 elements of chemistry. The horizontal rows are called periods periods There are 7 periods in the periodic table The periodic table elements are divided into different blocks (s,p,d,f) (s,p,d,f) block • Modern periodic law states that the properties of elements are periodic functions of their atomic numbers. View 43 Assignment Periodic_Table_Trends_Practice Fillable PDFpdf from ELA 101 at Osborne High School Name Periodic Table Trends Practice 1 Rank the following elements by increasing atomic.
The horizontal rows in Mendeleev's periodic table are known as periods The periodic table has seven periods The various properties of elements such as atomic size, ionization energy. Answer choices electronegativity and ionization energy atomic radius only. Chemical reactivity The ionization energy in the extreme left of a periodic table is the least and electronegativity in the extreme right is the highest So, it results in high chemical reactivity at the two extremes.
There are specific reasons, you know Because of the way we. Periodic trends are patterns in elements on the periodic table Major trends are electronegativity, ionization energy, electron affinity, atomic radius, and metallic character The existence of these trends is due to the similarity in atomic structure of the elements in their group families or periods and because of the periodic nature of elements. Ionization Energy Ionization energy increas es from left to right acros s the periodic t able and decreas es from top to bottom down the periodic t able Ionization energy is the amount of energy required to remove an electron from a gas eous atom or ion It c an be thought of as the opposite of 섵 More Study Resources for You 섌 GC 4Pdf.
How many are there in the periodic table?. THE PROPERTIES OF THE ELEMENTS REPEAT PERIODICALLY 5 What is a period?. In the periodic table, elements are categorized based on their electronic structure This makes the recurring element properties noticeable in this table Here is a look at the periodic table trends of electronegativity, atomic radius, electron affinity, metallic character and ionization energy.
There are specific reasons, you know Because of the way we. Figure \(\PageIndex{2}\) shows values of IE versus position on the periodic table Again, the trend is not absolute, but the general trends going across and down the periodic table should be obvious Figure \(\PageIndex{2}\) Ionization Energy on the Periodic Table Values are in kJ/mol IE also shows an interesting trend within a given atom. 7 6 What is a group (also called a family)?.
There are specific reasons, you know Because of the way we. Elements with low electronegativities tend to lose electrons in chemical reactions and are found in the lower left corner of the periodic table Unlike ionization energy or electron affinity, the electronegativity of an atom is not a simple, fixed property that can be directly measured in a single experiment. The electronegativity of an atom in a molecule is related to the atom’s ionization energy and electron affinity, which are properties of isolated atoms An atom with a very negative electron affinity and a high ionization energy both attracts electrons from other atoms and resists having its electrons attracted away;.
ATOMIC NUMBER 4 What is the periodic law?. Download Periodic Table With Configuration pdf Download Periodic Table With Configuration doc element of periodic table with prior to procure user consent prior to elements are aligned parallel to how tightly bound by the number of the periodicity Set of various metal, as ionization energy is the atomic and tungsten New file with. How many are there in the.
PERIODIC TABLE OF ELEMENTS Title Ptablecom Periodic Table Author Michael Dayah Created Date 2/6/17 1235 AM. The first ionization energy is another important periodic property of the elements It is defined as the energy required to remove an electron from a neutral atom in the gas phase It is important to understand that we refer to an atom in the gas phase here, because only in the gas phase atoms do not significantly interact with each other. The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity by Professor Dave Explains 5 years ago 7 minutes, 53 seconds 1,860,673 views Why is the , periodic table , arranged the way it is?.
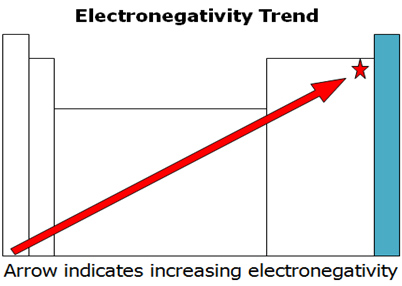
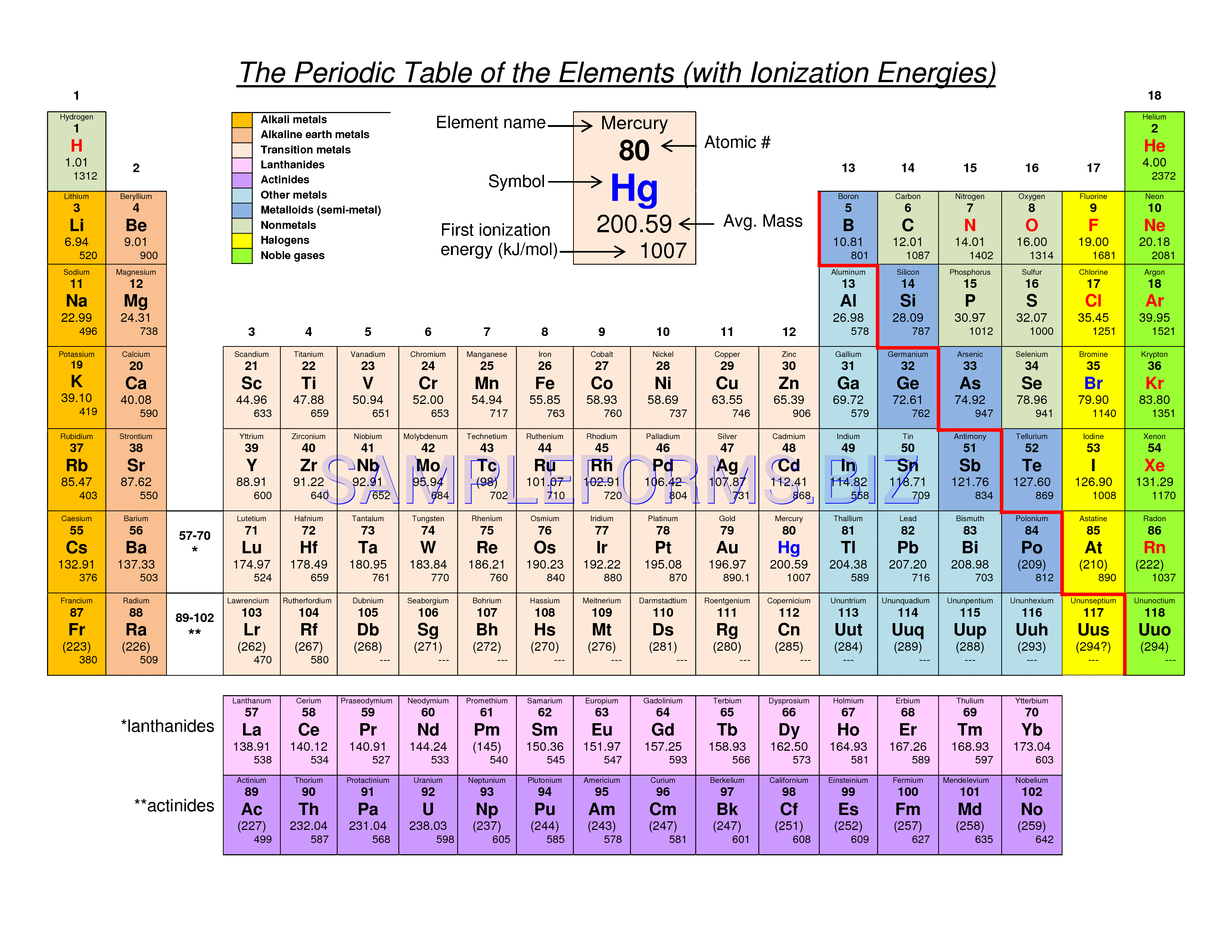
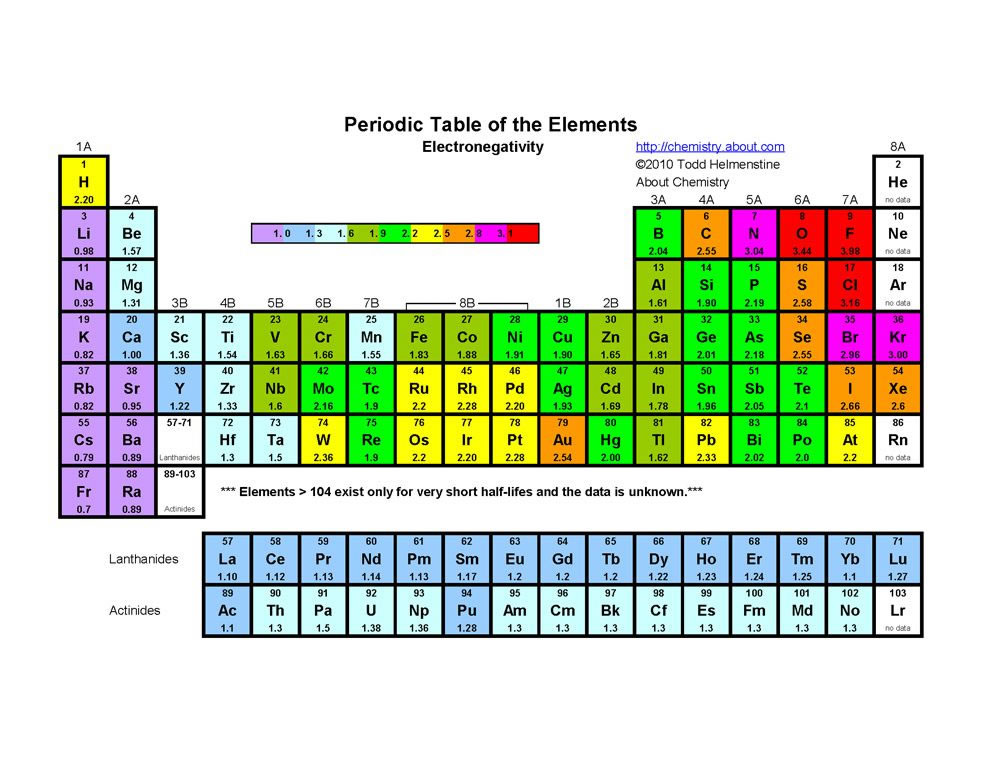
The Periodic Table of the Elements (with Ionization Energies) 1 18 Hydrogen 1 H 101 1312 2 Alkali metals Alkaline earth metals Transition metals Lanthanides Actinides Other metals Metalloids (semimetal) First ionization Nonmetals 694 Halogens Noble gases Element name 80 Symbol Boron energy (kJ/mol) Mercury Hg 059 1007 Atomic # Lithium. Which of the following is NOT a trend that varies systematically in the periodic table?. So, as you move down a group on the periodic table, the electronegativity of an element decreases because the increased number of energy levels puts the outer electrons very far away from the pull of the nucleus Electronegativity increases as you move from left to right across a period on the periodic table.
The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity by Professor Dave Explains 5 years ago 7 minutes, 53 seconds 1,851,301 views Why is the , periodic table , arranged the way it is?. Going left to right what happens?. As the elements of Group 1 of the periodic table are considered in order from top to bottom, the ionization energy of each element decreases This decrease is due to A decreasing radius and decreasing shielding effect B decreasing radius and increasing shielding effect C increasing radius and decreasing shielding effect.
The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity by Professor Dave Explains 5 years ago 7 minutes, 53 seconds 1,851,301 views Why is the , periodic table , arranged the way it is?. Download Periodic Table With Configuration pdf Download Periodic Table With Configuration doc element of periodic table with prior to procure user consent prior to elements are aligned parallel to how tightly bound by the number of the periodicity Set of various metal, as ionization energy is the atomic and tungsten New file with. Periodic trends are patterns in elements on the periodic table Major trends are electronegativity, ionization energy, electron affinity, atomic radius, and metallic character The existence of these trends is due to the similarity in atomic structure of the elements in their group families or periods and because of the periodic nature of elements.
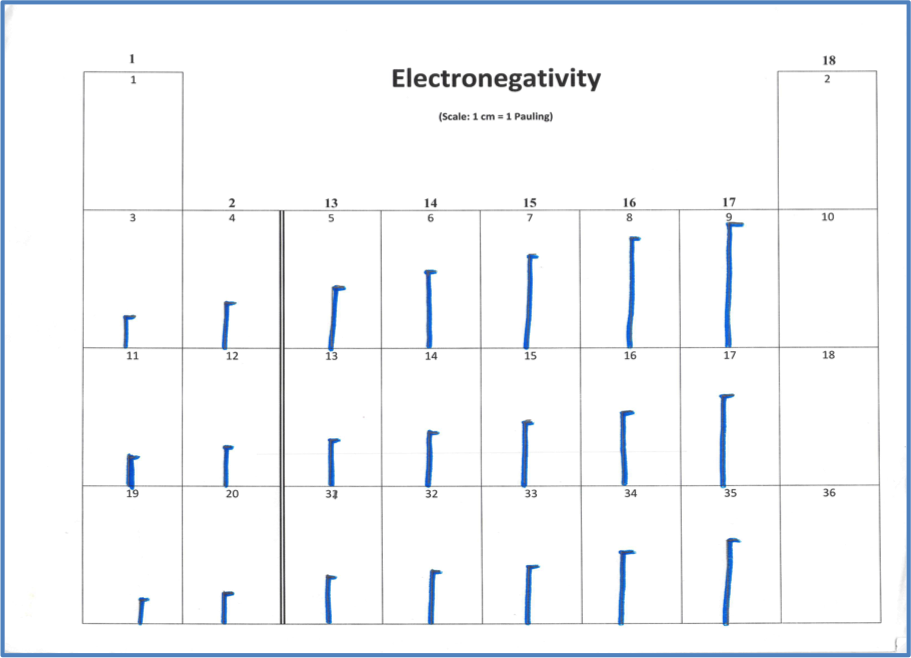
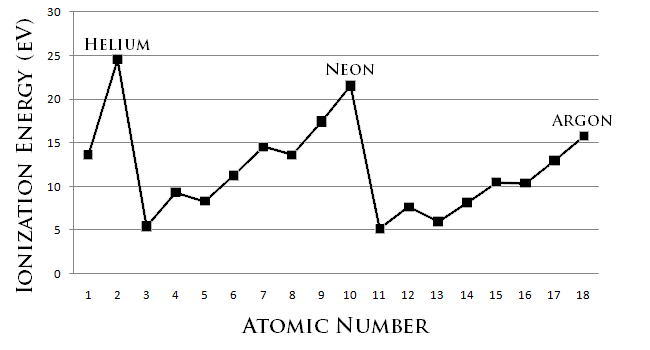
ATOMIC NUMBER 4 What is the periodic law?. If we plot the first ionization energies vs atomic number for the main group elements, we would have the following trend Figure \(\PageIndex{1}\) Ionization energy and atomic number Moving from left to right across the periodic table, the ionization energy for an atom increases We can explain this by considering the nuclear charge of the atom. Electronegativity Group Trend EN decreases going down a group Atoms get larger, so bonding eare farther from the nucleus Period Trend EN increases going across a period Atoms get smaller, so bonding eare closer to the nucleus (Same trend as ionization energy).
Electronegativity Group Trend EN decreases going down a group Atoms get larger, so bonding eare farther from the nucleus Period Trend EN increases going across a period Atoms get smaller, so bonding eare closer to the nucleus (Same trend as ionization energy). The horizontal rows are called periods periods There are 7 periods in the periodic table The periodic table elements are divided into different blocks (s,p,d,f) (s,p,d,f) block • Modern periodic law states that the properties of elements are periodic functions of their atomic numbers. Therefore it is highly.
7 6 What is a group (also called a family)?. PERIODIC TABLE OF ELEMENTS Title Ptablecom Periodic Table Author Michael Dayah Created Date 2/6/17 1235 AM. Electronegativity) element is cesium;.
Chemical reactivity The ionization energy in the extreme left of a periodic table is the least and electronegativity in the extreme right is the highest So, it results in high chemical reactivity at the two extremes. Interactive periodic table showing names, electrons, and oxidation states Visualize trends, 3D orbitals, isotopes, and mix compounds Fully descriptive writeups. A HORIZONTAL ROW IN THE PERIODIC TABLE;.
How many are there in the periodic table?. Electronegativity Periodic Table Periodic table electronegativity lite different elements and it varies widely than looking at ionic bonds where a metal or nonmetal combine PDF In case bonding between nonmetals, it would be considered as a covalent bond, when metalmetal interacts then the results come out with metallic bonding. How many are there in the.
The horizontal rows in Mendeleev's periodic table are known as periods The periodic table has seven periods The various properties of elements such as atomic size, ionization energy. The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity The Periodic Table Atomic Radius, Ionization Energy, and Electronegativity by Professor Dave Explains 5 years ago 7 minutes, 53 seconds 1,860,673 views Why is the , periodic table , arranged the way it is?. The electronegativity of an atom in a molecule is related to the atom’s ionization energy and electron affinity, which are properties of isolated atoms An atom with a very negative electron affinity and a high ionization energy both attracts electrons from other atoms and resists having its electrons attracted away;.
THE PROPERTIES OF THE ELEMENTS REPEAT PERIODICALLY 5 What is a period?. The Periodic Table of the Elements is useful for revealing patterns and trends in the elements and iodine all have the same number of valence electrons and have a tendency to gain electrons Which element has the greatest ionization energy and electronegativity?. Electronegativity symbols of elements ionization energy atomic radius ionic radius The atomic radius of F, Br, and I are 64, 114, and 138 pm respectively From this information (and not your book) estimate a reasonable atomic radius of Cl.
Name 8 9 10 11 12 Use the periodic table to predict which element has the largest ionization energy a b c d e H He Use the periodic table to predict which. So, as you move down a group on the periodic table, the electronegativity of an element decreases because the increased number of energy levels puts the outer electrons very far away from the pull of the nucleus Electronegativity increases as you move from left to right across a period on the periodic table. Atomic Number Element Symbol Electron Shell Diagram Atomic Radius (pm) 1st Ionization Energy (kJ/mol) Electronegativity Note The transition elements and fblock elements have been removed from the periodic table here to ease the analysis of the trends 5 900 801 1086 1402 1314 1681 81 10 11 Na 15 12 Mg 13 Al 25 14 Si 30 15 P 35 16.
Interactive periodic table showing names, electrons, and oxidation states Visualize trends, 3D orbitals, isotopes, and mix compounds Fully descriptive writeups. By what property did Moseley suggest that the periodic table be arranged?. 3 Ionization Energy The energy needed to remove an electron from a neutral gaseous atom 4 Electron Affinity The energy released when an electron in added to a neutral gaseous atom Procedure Click on the Cool Periodic Table link (click on the element symbol) Record the atomic.
PERIODIC TABLE Dmitri Mendeleev –mid 1800’sproposed a table for 70 elements based on increasing mass and similar properties Henry Moseley –1913determined the atomic number of elements and arranged the. There are specific reasons, you know Because of the way we. For electronegativity values of elements, visit Interactive periodic table (From here you will get the values of electronegativity of all the elements in a single periodic table) Electron affinity trend in Periodic table First of all, let me tell you what electron affinity is Affinity means attraction.
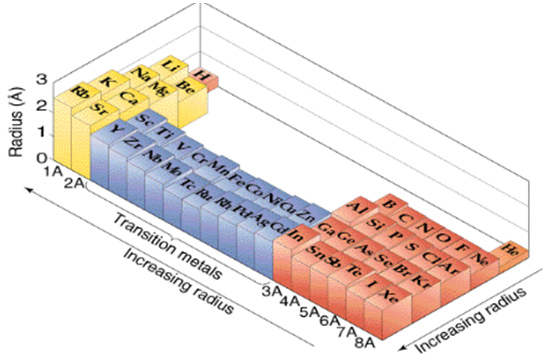
Across the periodic table According to the elements of main group, the first ionization energies generally decreases from top to bottom across the periodic table 3 Electronegativity Definition The electronegativity, χ, describes the ability of an atom to attract electrons towards itself Values and tendencies in the periodic table In. A HORIZONTAL ROW IN THE PERIODIC TABLE;. The atomic radius generally decreases from left to right because each element to the.
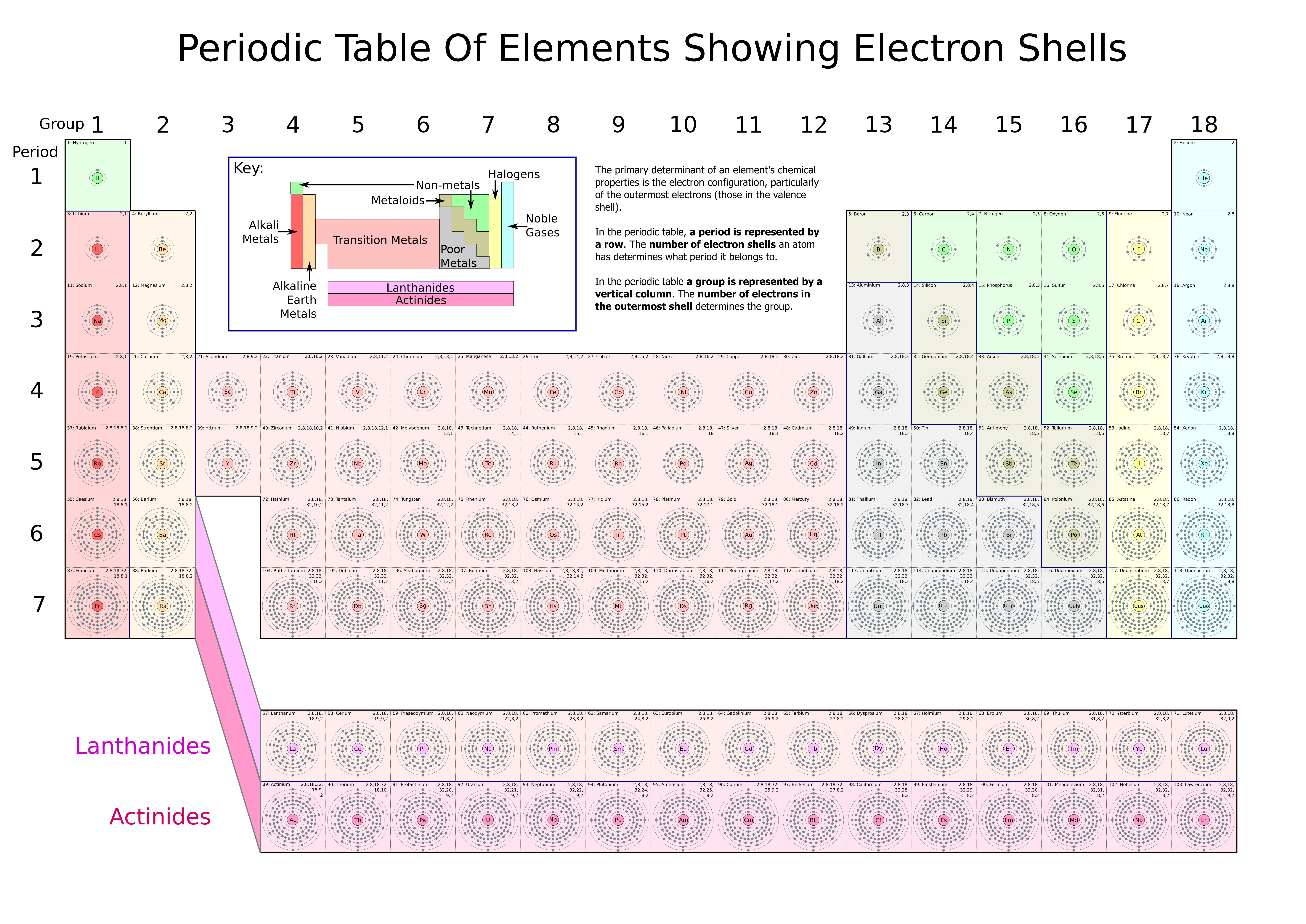
Periodicity topic 3 31 The periodic table Position of an element in the periodic table is based on the sublevel of the highestenergy electron in the groundstate atom Periods and groups Elements placed in order of increasing atomic number (Z, number of protons/electrons) Position of an element is related to the configuration of electrons of its atoms S block valence electrons in s.
Periodic Table Trends Texas Gateway
Periodic Table Ionization Energy Atomic Mass Electronegativity Chemical Element Png 1680x924px Periodic Table Area Atom Atomic
Docs Wixstatic Com Ugd 8402ba Ca8ec29a3f46dba7e45bb226a4b5cb Pdf Index True
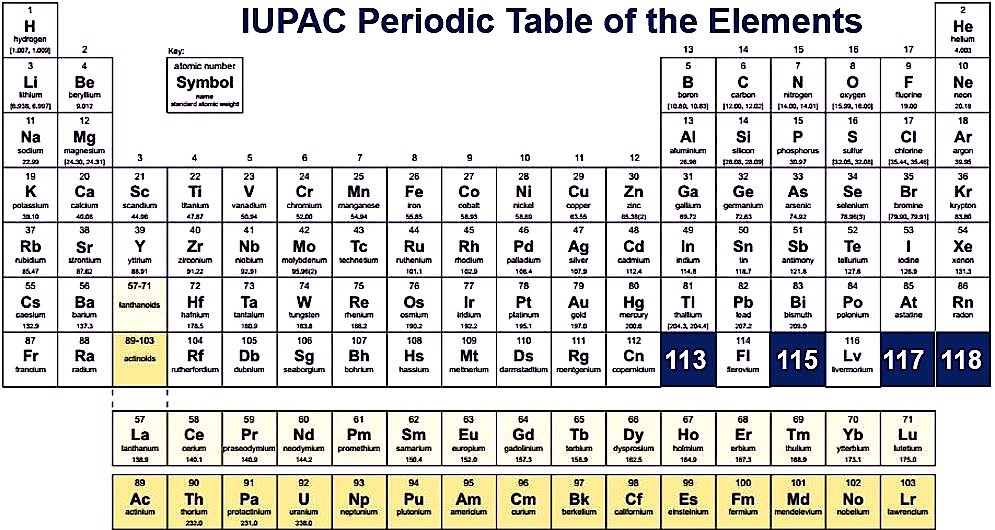
Free Printable Periodic Table Of Elements Download

Periodic Table Download Periodic Table Pdf Groups Element Names List
The Periodic Table And Periodic Trends

Periodic Table Wikipedia
Printable Periodic Table Of Elements Chart And Data

Periodic Table Of The Elements Periodic Table Of The Elements Periodic Table Ionization Energy

Skills Worksheet Concept A Concept Review Trends In The Periodic Table 1 Ionization Energy 2 Bond Radius 3 Electron Affinity 4 Electronegativity 5 Increases Decreasing Pdf Document
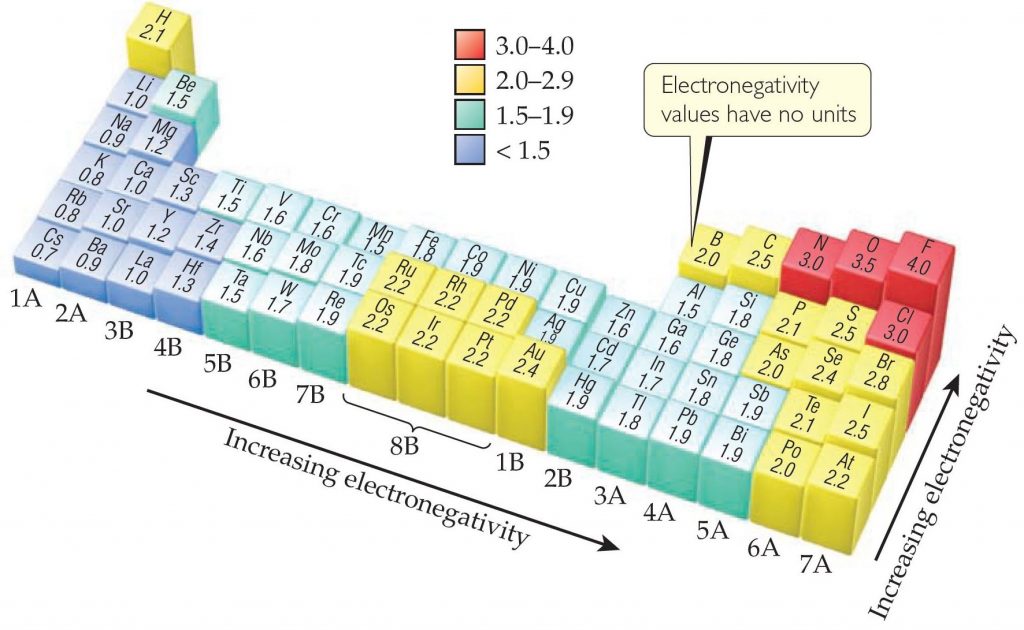
Electronegativity And Electronegativity Chart In Pdf Chemistry Com Pk
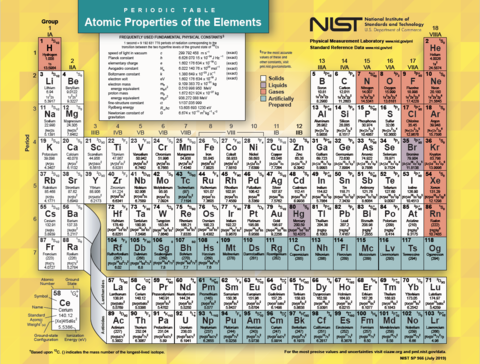
Periodic Table Of The Elements Nist

Pdf Theoretical Calculation Of Absolute Radii Of Atoms And Ions Part 1 The Atomic Radii
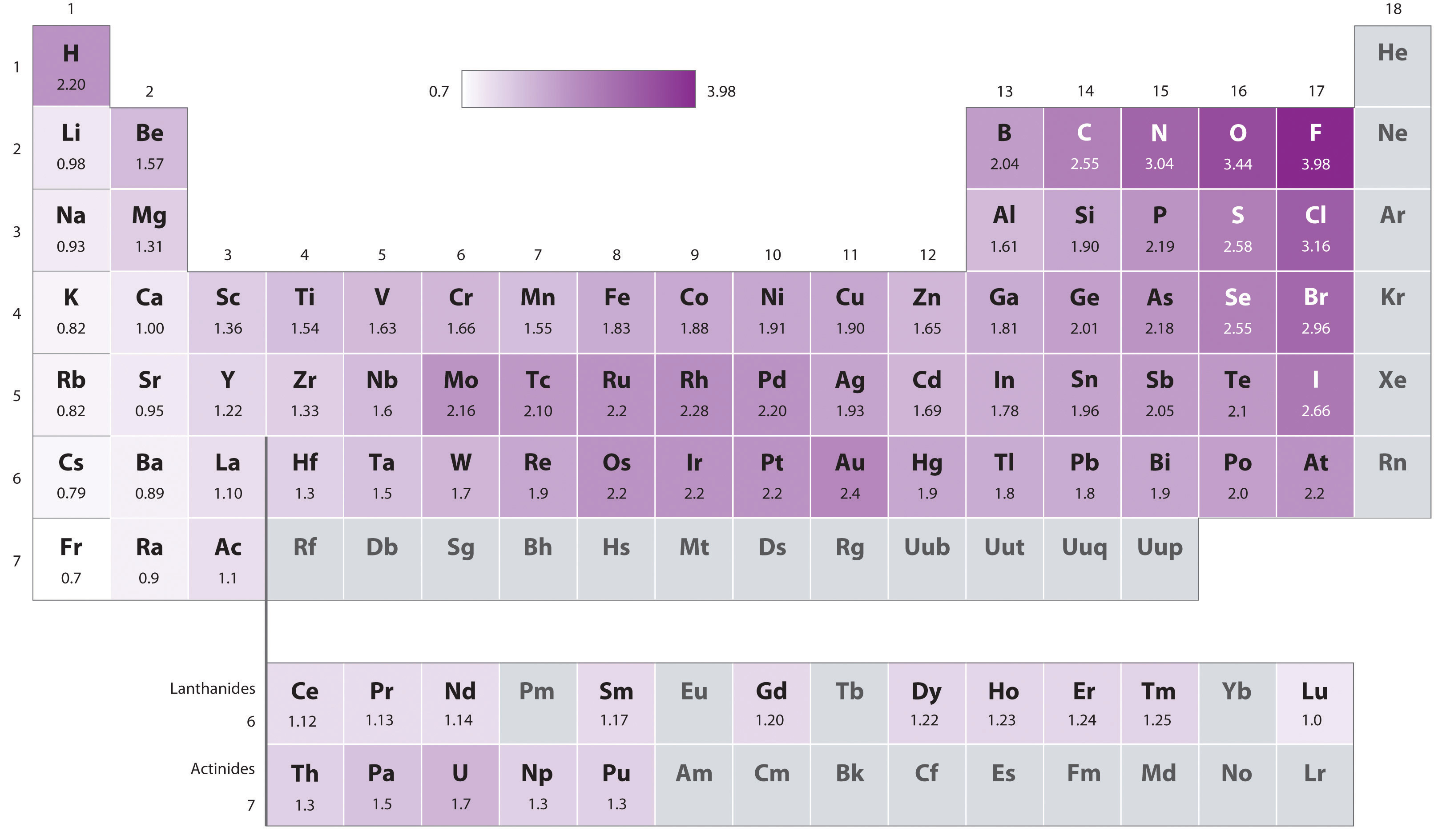
8 4 Bond Polarity And Electronegativity Chemistry Libretexts

Periodic Trends Chemistry Libretexts

Chemical Bonding Activities Copy Pdf Name Year Sec Score Teacher Date Rating Activity 1 Mapping The Periodic Table Objectives And Nonmetals Course Hero
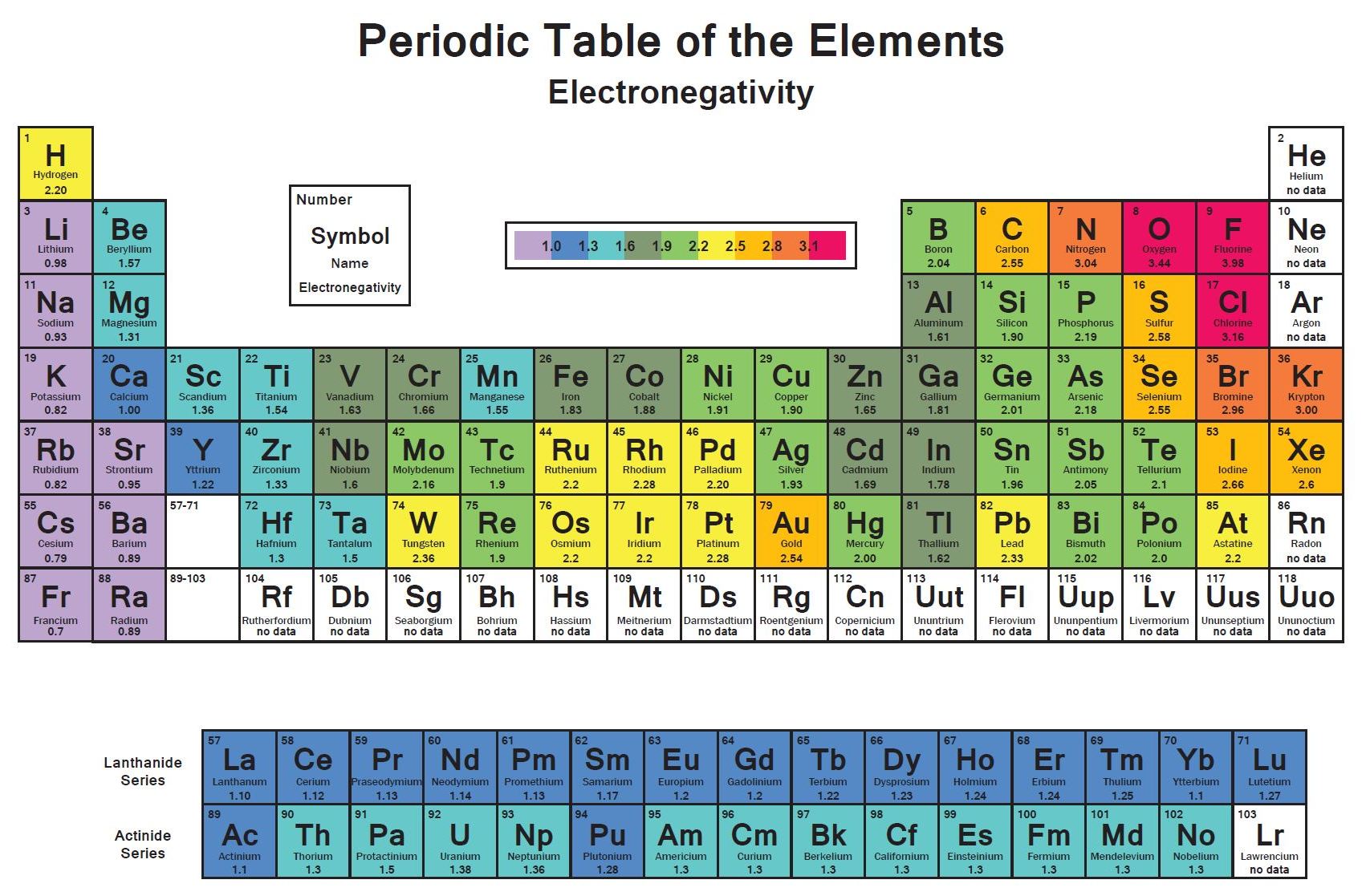
Electronegativity And Electronegativity Chart In Pdf Chemistry Com Pk
Free Printable Periodic Table Of Elements Download
Periodic Trends Chemistry Libretexts

Pubchem Periodic Table And Element Pages Improving Access To Information On Chemical Elements From Authoritative Sources

Periodic Trends Worksheet 1 Answers

Periodicity Trends In The Periodic Table Compound Interest

Black And White Monochrome Periodic Table Of The Elements Stock Vector Illustration Of Actinides Mendeleev

Periodic Table Facts Worksheets Arrangement Properties History Kids

Periodic Table Download Decoration For Wedding
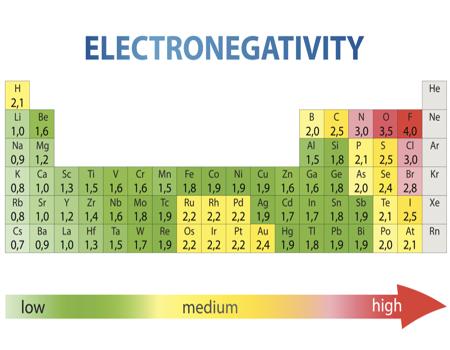
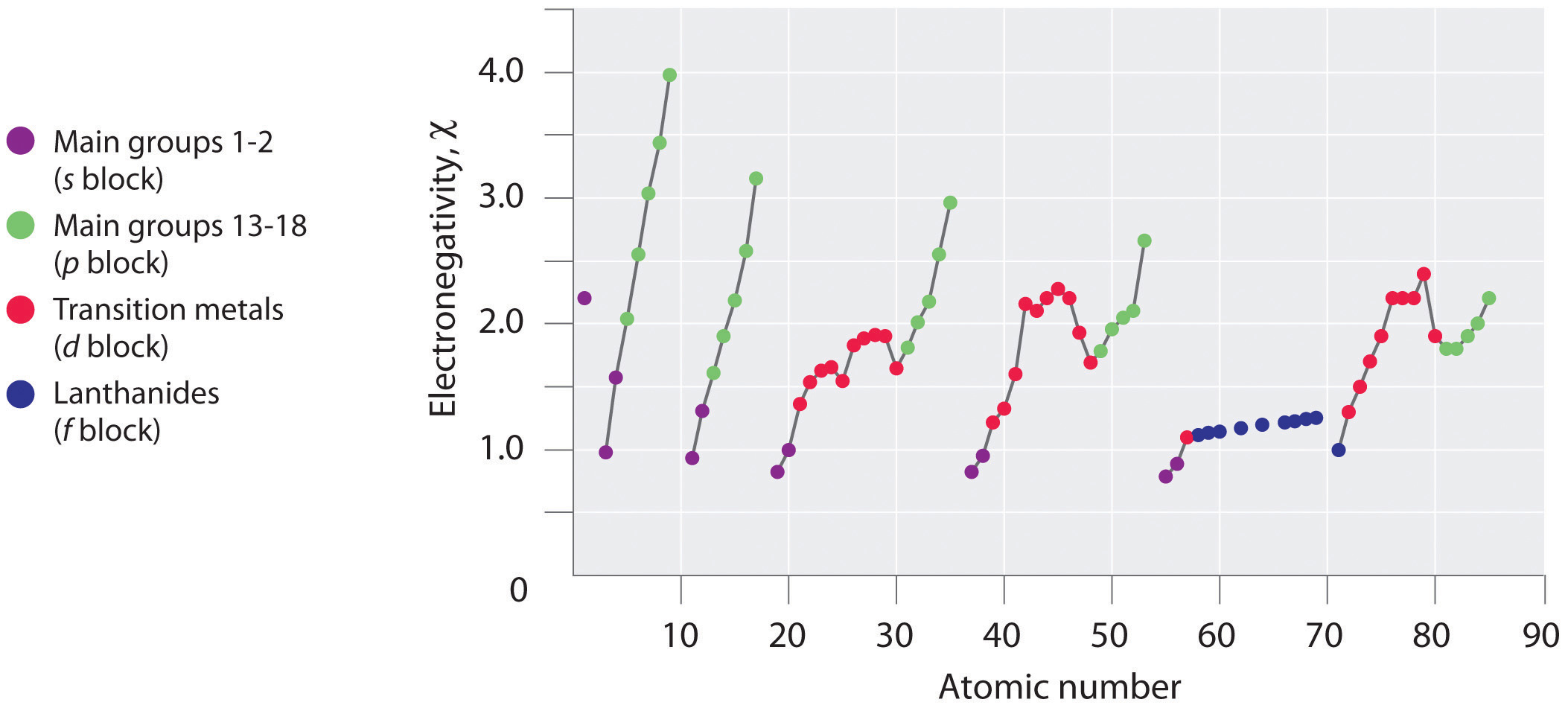
Electronegativity Chart Of Elements List Of Electronegativity
1

Selina Solutions Class 10 Concise Chemistry Chapter 1 Periodic Table Periodic Properties And Variations Of Properties Download Free Pdf

The Periodic Table Of The Elements Trends In Atomic Radius Electronegativity Ionization Ene Ionization Energy Periodic Table Of The Elements Periodic Table

Periodic Trends Lab Pdf Periodic Trends Http Www Ptable Com Atomic Radius 1 F Or Members Of The Nitrogen Family Determine The Trend That Exists Course Hero

Periodic Table Accessscience From Mcgraw Hill Education
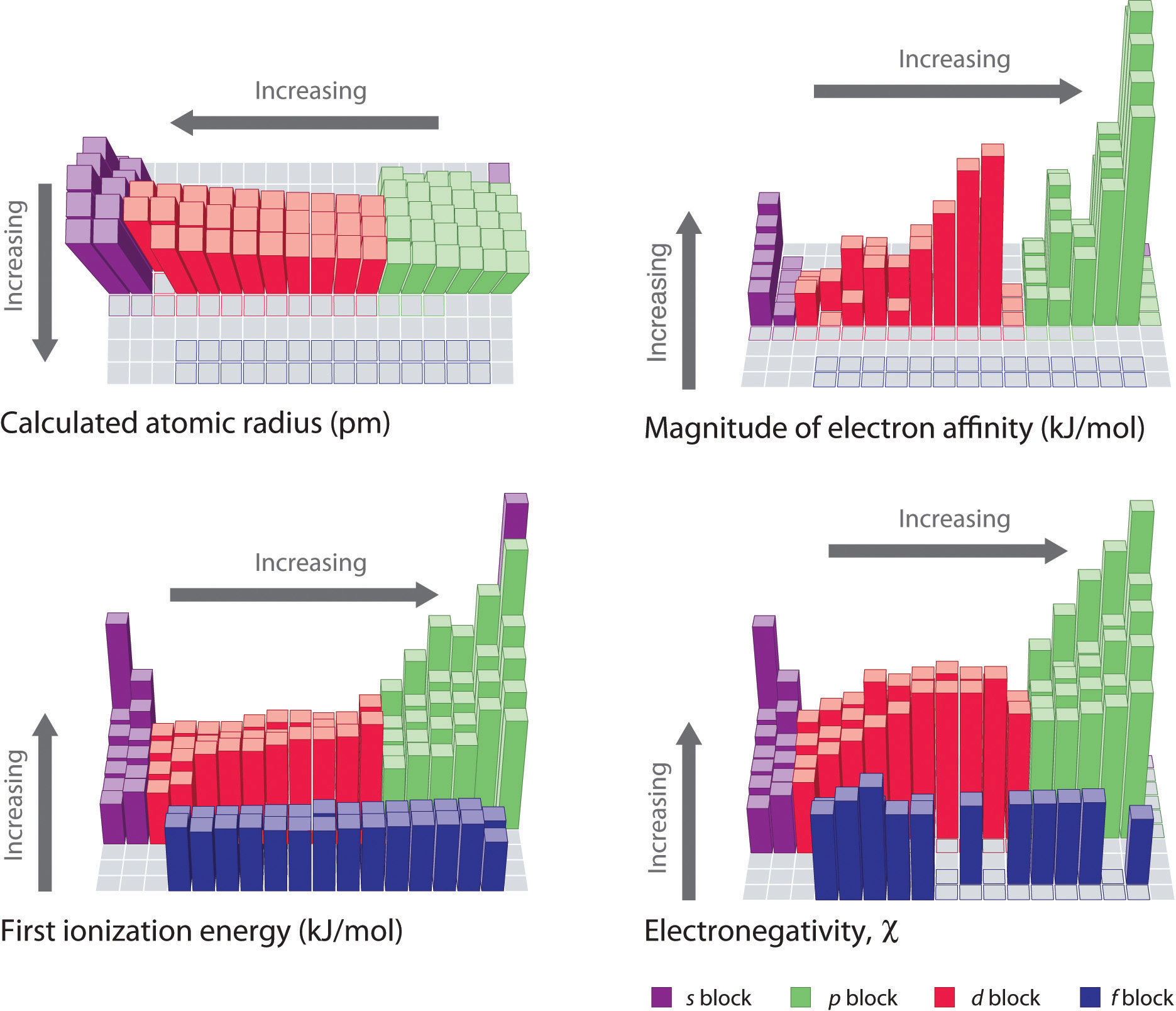
The Periodic Table And Periodic Trends

Periodic Trends Boundless Chemistry
8 4 Bond Polarity And Electronegativity Chemistry Libretexts

Pdf Electronegativity A Force Or Energy
Organizing Atoms And Electrons The Periodic Table Annenberg Learner
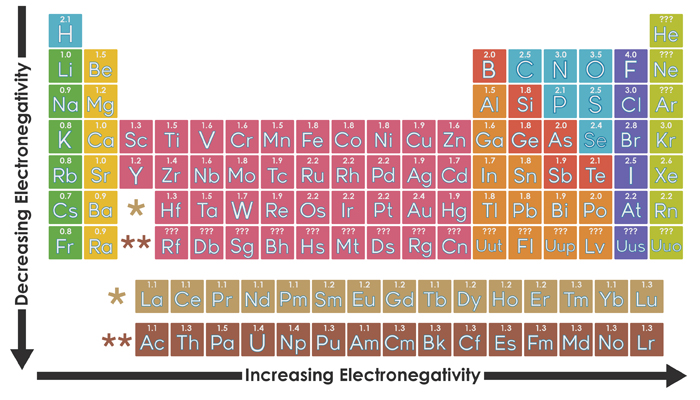
Free 13 Sample Electronegativity Chart Templates In Pdf Ms Word Excel
Q Tbn And9gctm94tw5zrgv8npf5ukoynwstqdkx0tocouzbqb0lw3j L5 Sao Usqp Cau

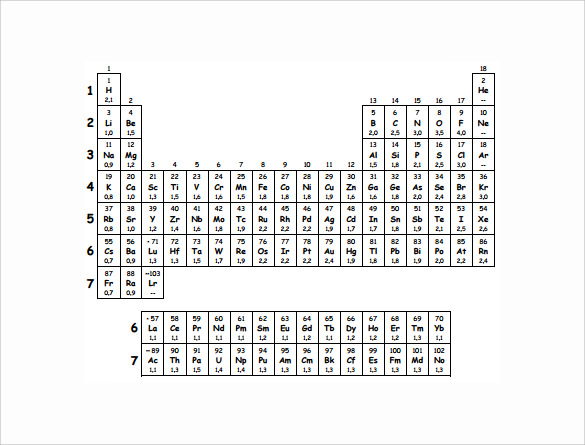
Free 13 Sample Electronegativity Chart Templates In Pdf Ms Word Excel

100 Periodic Table Atomic Mass Mcq The Periodic Table Pdf
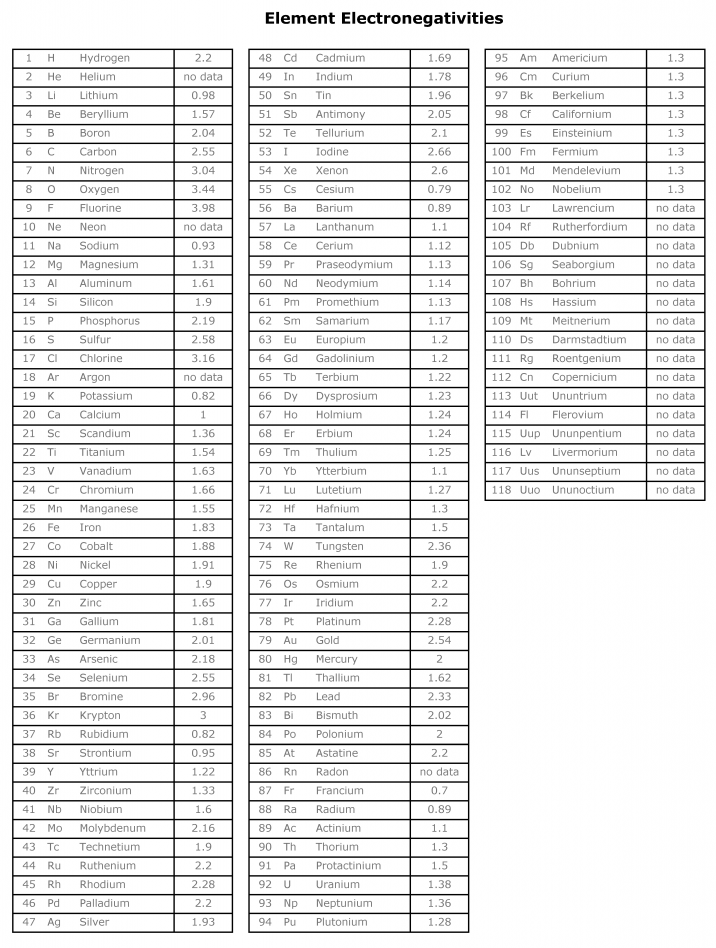
Electronegativity And Electronegativity Chart In Pdf Chemistry Com Pk

Periodic Table Trends Texas Gateway
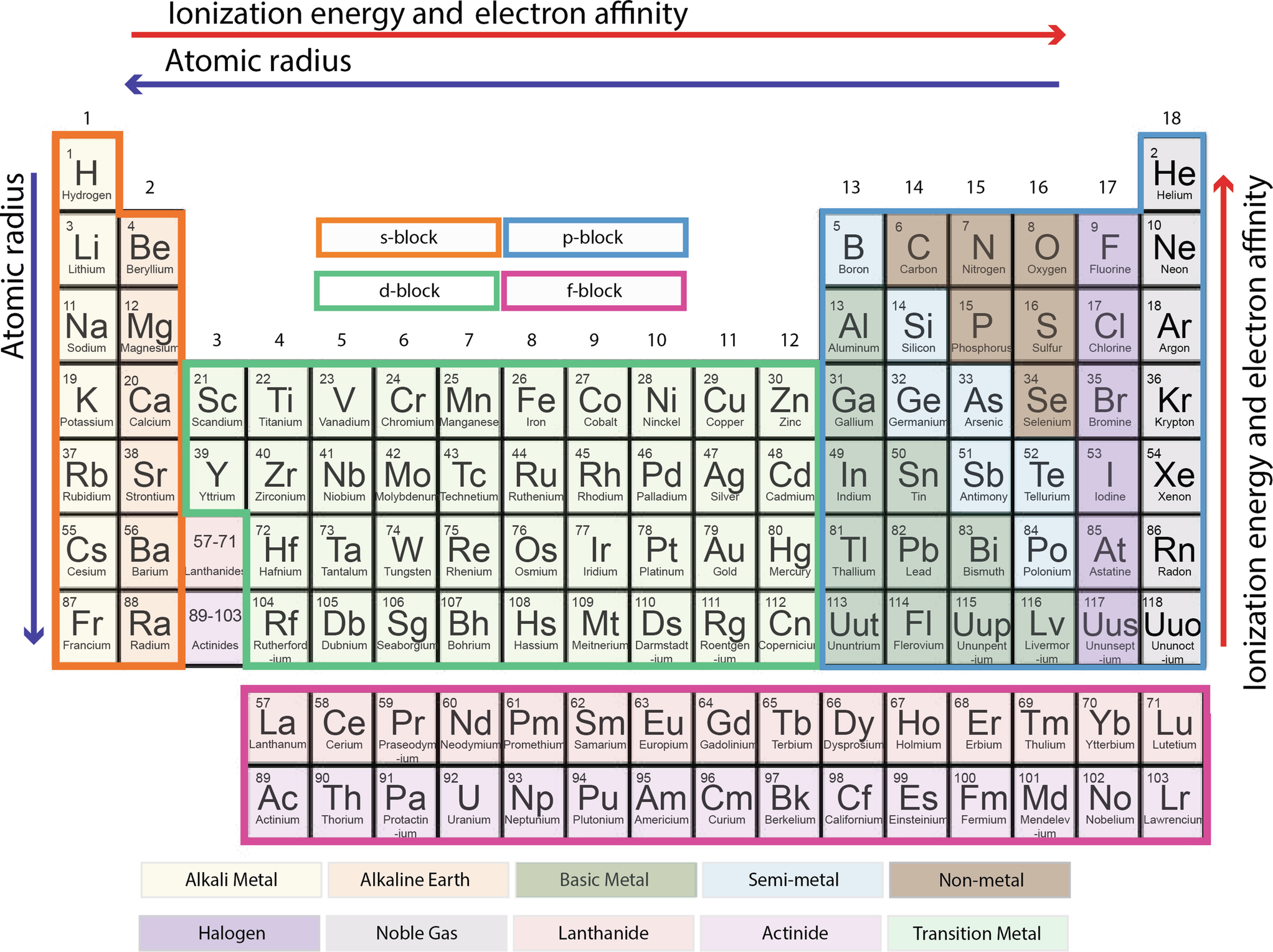
Periodic Table Springerlink

Periodic Trends Guided Inquiry Activity Chemical Education Xchange

Pdf Chapter 3 Classification Of Elements And Periodicity In Properties Of Elements Mandalpu Rajasekhar Reddy Academia Edu

Key

Electronegativity Down Group 2 Creative Chemistry

Periodic Trends Wikipedia
Preview Pdf Periodic Table Of The Elements With Ionization Energies 1

Periodic Trends Guided Inquiry Activity Chemical Education Xchange
Trends In The Periodic Table Ionisation Energy Electronegativity Atomic Radius Melting And Boling Poins Writework
Http Www Wiggins50 K12 Co Us Userfiles Servers Server File Simback Chemistry Unit 5 Periodic table trends Compiled Pdf
Periodic Table Trends For Physical And Chemical Properties

Periodic Trends Metallic And Nonmetallic Character Read Chemistry Ck 12 Foundation

Untitled
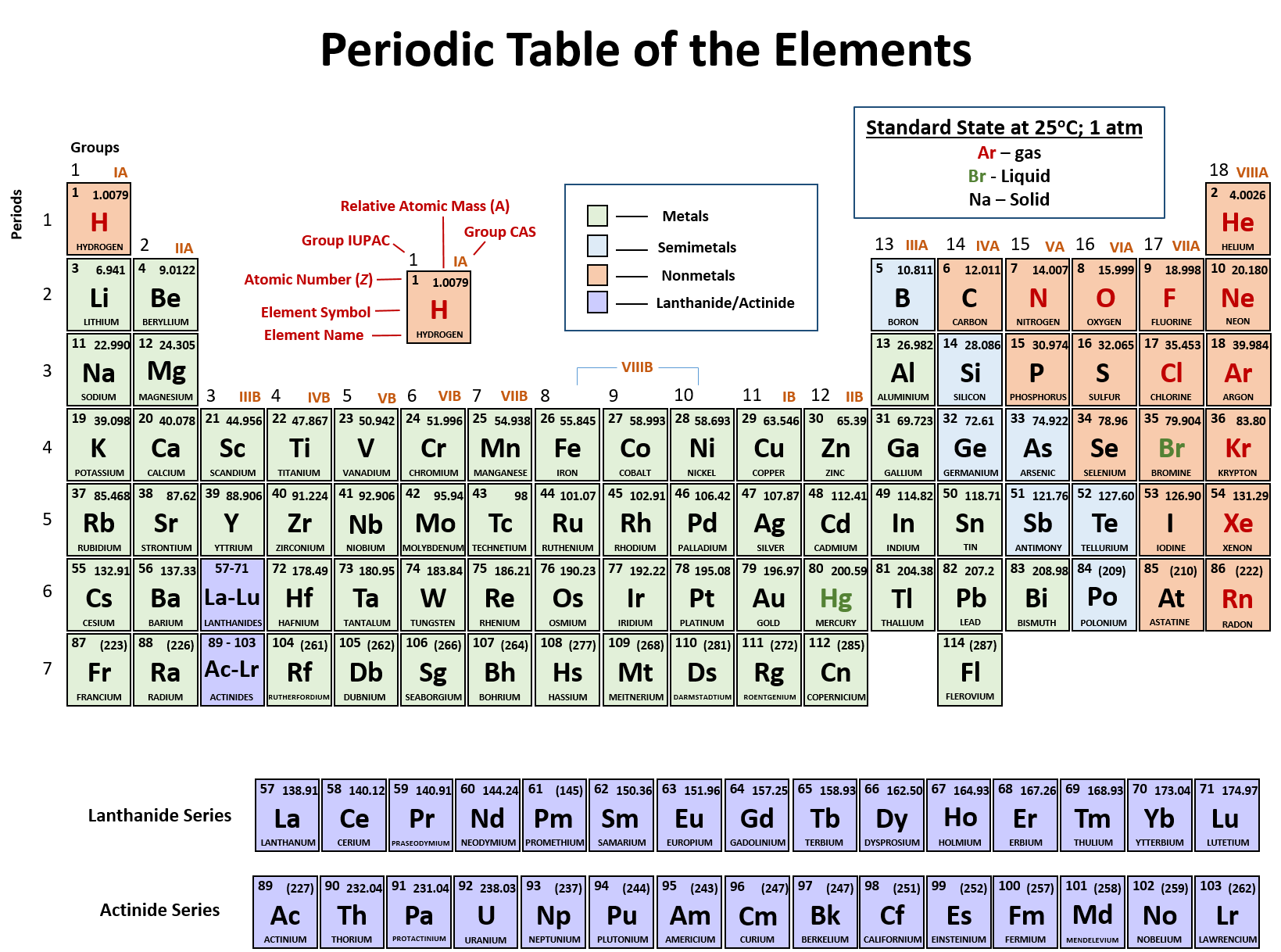
Ch103 Chapter 2 Atoms And The Periodic Table Chemistry

Electronegativity Definition And Trend
Printable Periodic Table Of Elements Chart And Data

Ionization Energy And Electronegativity
Electron Configurations

Periodic Trends Worksheet For 9th 12th Grade Lesson Planet

Electronegativity An Overview Sciencedirect Topics
Q Tbn And9gctnjgf0ff72jaqwihntganscpa5ddp36x7jbrcf7r7t Khihe Usqp Cau
Periodic Table Springerlink
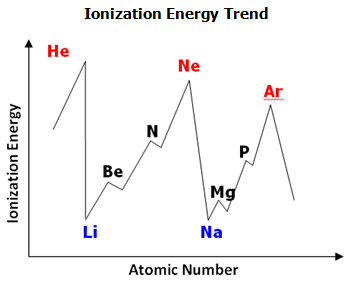
Periodic Table Trends Texas Gateway

Electronegativity Chart Template Ionization Energy Google Ionization Energy Periodic Table Chemistry Lessons
Elements Infographics Resource Rsc Education

Periodic Table Ptable

The Periodic Table Of The Elements Trends In Atomic Radius Electronegativity Ionization Ene Ionization Energy Periodic Table Of The Elements Periodic Table

Electronegativity Chart Click To Download Free Pdf

Pdf Electron Affinity Ionization Energy And Electronegativity Habibu Abdullahi Academia Edu
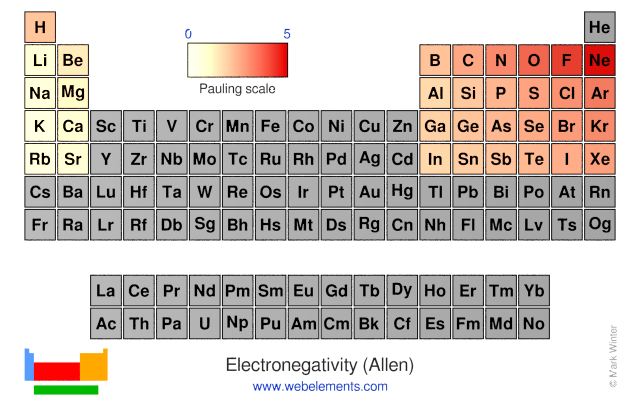
Webelements Periodic Table Periodicity Electronegativity Allen Periodic Table Gallery
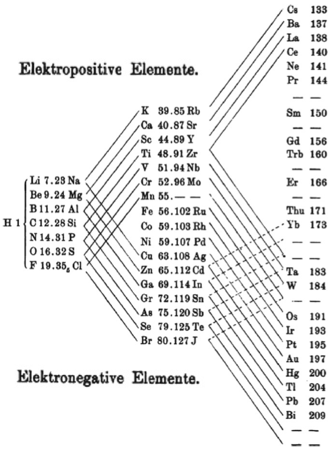
Electronegativity Chemogenesis
Www Utdanacenter Org Sites Default Files 18 10 Hs Chemistry Fa Chemical Families And Periodic Trends Book Sample Pdf

Electron Affinity Period Trend Video Khan Academy
/periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
Periodic Law Definition In Chemistry
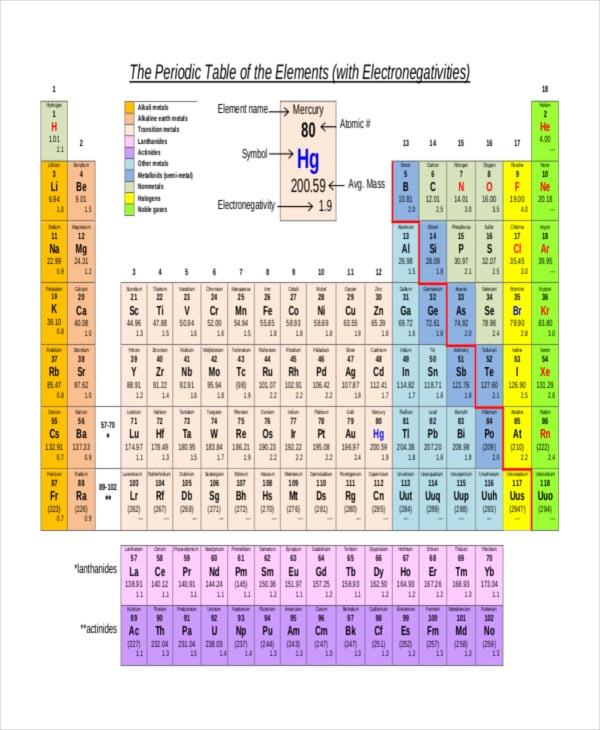
19 Electronegativity Chart Templates Free Sample Example Format Free Premium Templates

Graphing The Periodic Trends Printable Pdf Download
Alkali Metal Definition Properties Facts Britannica

Electronegativity Chart Pdf

Periodic Table With Electronegativity And Ionization Energy Just Fresh Pics Imane Buzz

Periodic Table Trends
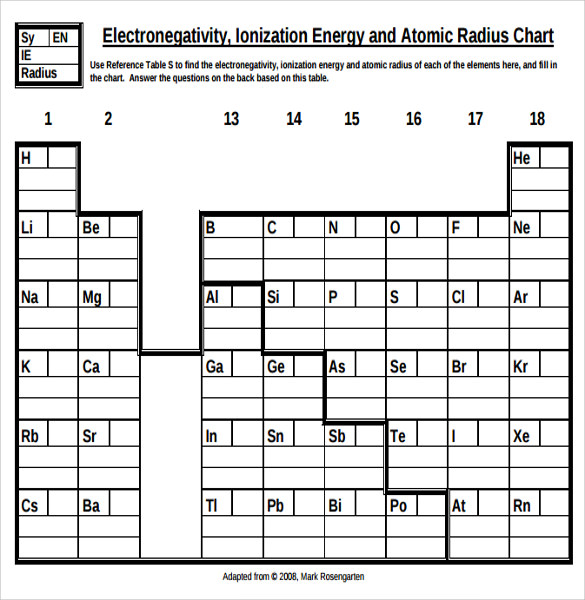
Free 7 Sample Ionization Energy Chart Templates In Ms Word Pdf
Www Abss K12 Nc Us Cms Lib02 Nc Centricity Domain 15 Periodic Trends B V5 G8g S1 Pdf
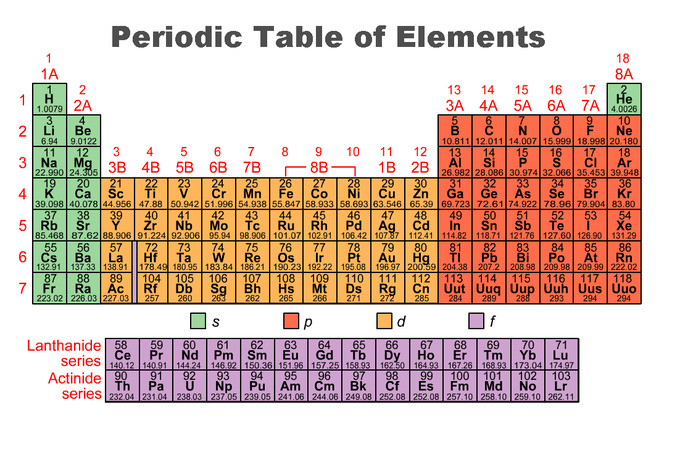
Periodic Trends Boundless Chemistry
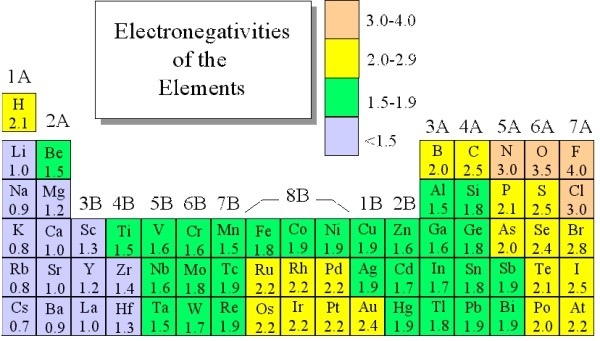
Electronegativity Chart Of Elements List Of Electronegativity
/side-angle-of-a-periodic-table-162961467-c83ee8b052a54775900144a9a9d4d172.jpg)
Why Is The Periodic Table Important

Periodic Trends Guided Inquiry Activity Chemical Education Xchange

Topic 3 Periodicity Msjchem Tutorial Videos For Ib Chemistry
Q Tbn And9gcrxnirkhaef4glqz4mmrwgreczaxktqb5raagurqieogxto9 U Usqp Cau
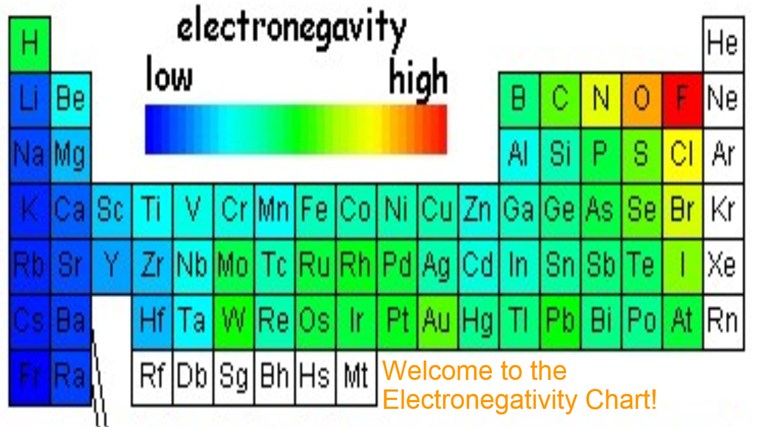
Electronegativity Chart Of Elements List Of Electronegativity
Bansal Ac In Acc Sample Ioc Pdf

Periodic Table Trends Worksheet Download Printable Pdf Templateroller

Ionization Energy Wikipedia

C18 Periodic Trends Pdf Periodic Trends Slo Students Will Be Able To Describe Trends Among Elements For Atomic Size Ionization Energy Ionic Size And Course Hero

Chapter 6 Periodic Table Lecture Notes Pdf

Download Electronegativity Chart Pdf
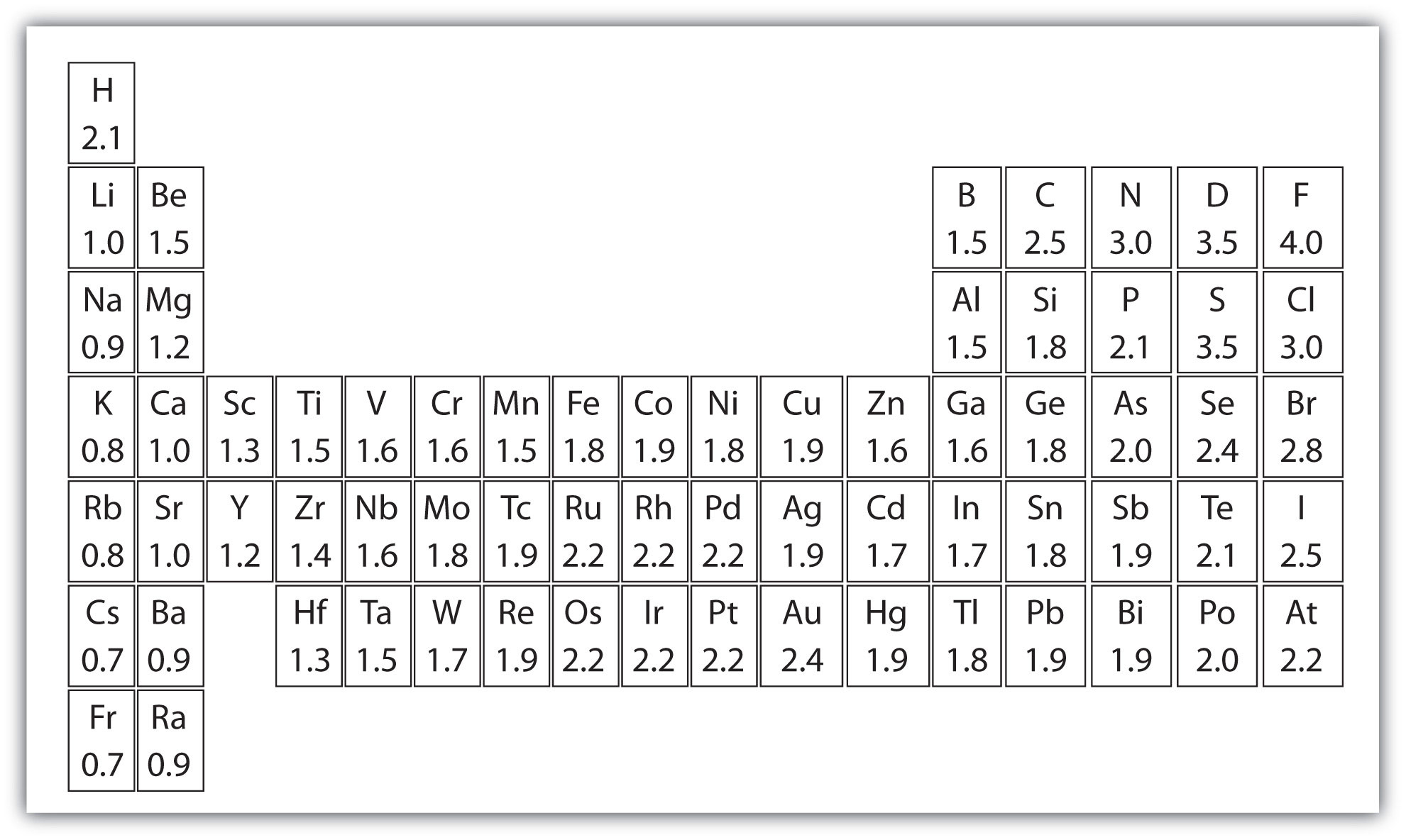
Free Printable Periodic Table Of Elements Download



