N 15 Protons Neutrons Electrons
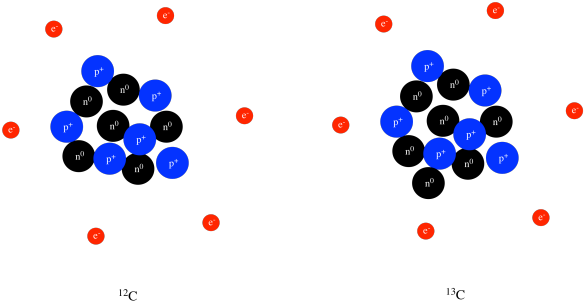
Structure Reactivity Atoms Protons Neutrons Electrons

Ppt 1 St Six Weeks Review Powerpoint Presentation Free Download Id

Worked Example Identifying Isotopes And Ions Video Khan Academy
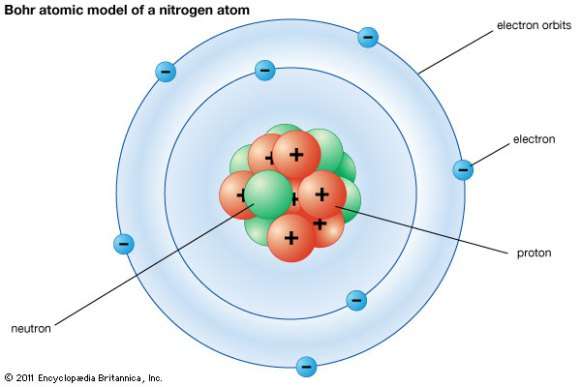
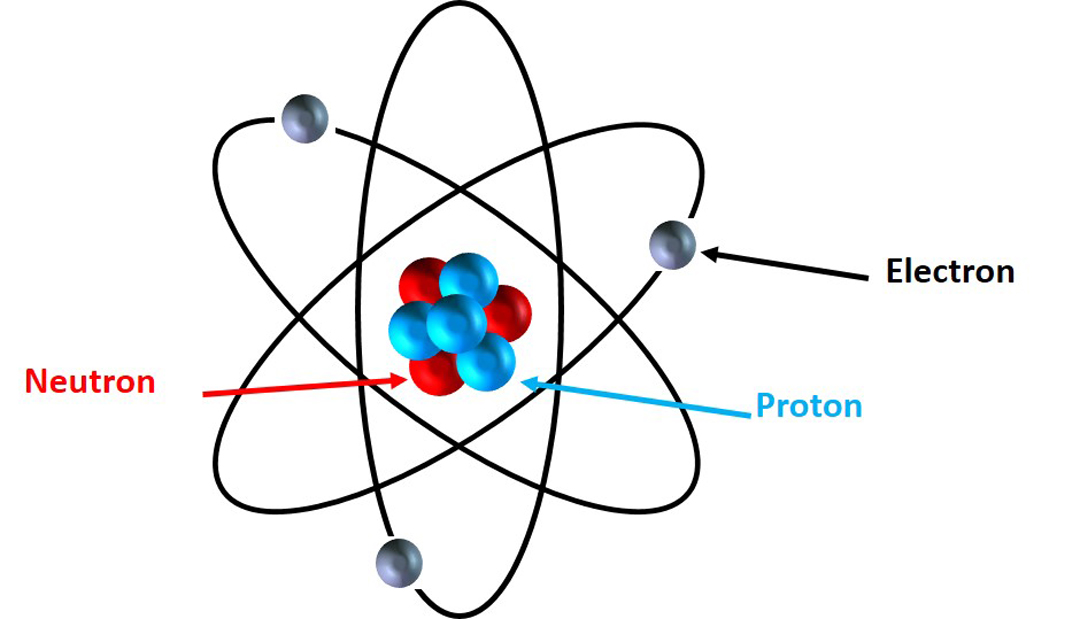
What Are The Parts Of An Atom
First Elements Storyboard By Oliversmith

Set 15 22 Chemistry Carlingford High School
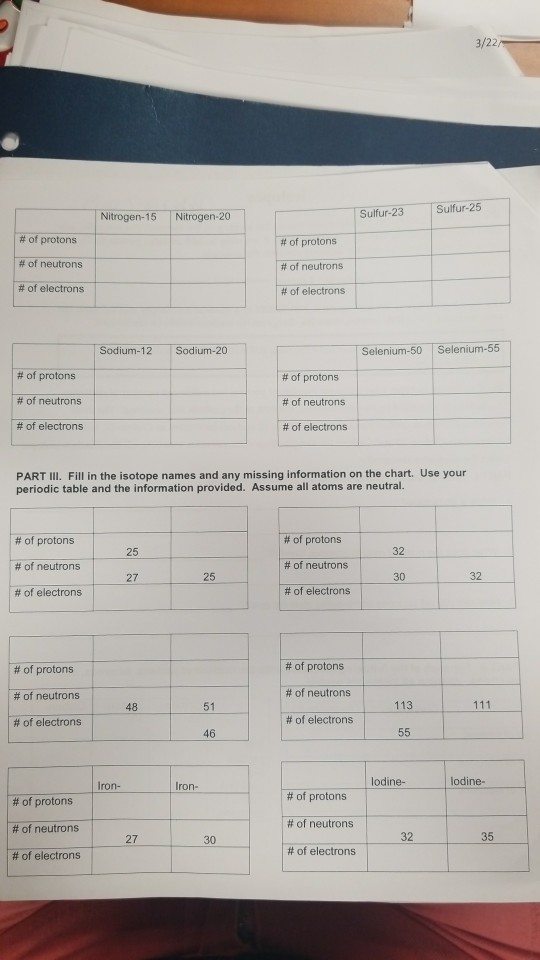
Determine the number of protons, neutrons and electrons in each and name them atomic number 26, mass number 58, charge of 2 atomic number 53, mass number 127, charge of 1− Answer a Iron, 26 protons, 24 electrons, and 32 neutrons Answer b Iodine, 53 protons, 54 electrons, and 74 neutrons.
N 15 protons neutrons electrons. Origin of Name From the Swedish words tung sten, meaning heavy stone The symbol comes from the German word wolfram Date and Place of Discovery Discovered in 1781;. Give the nuclear composition and isotopic notation for (a) an atom containing 27 protons, 32 neutrons, and 27 electrons (b) an atom containing 15 protons, 16 neutrons, and 15 electrons (c) an atom containing 110 neutrons, 74 electrons, and 74 protons. Oxygen17 is composed of 8 protons, 9 neutrons, and 8 electrons Both 17O and 18O are secondary isotopes, meaning that their nucleosynthesis requires seed nuclei Oxygen18 is composed of 8 protons, 10 neutrons, and 8 electrons Oxygen15, which is an unstable isotope of oxygen, is composed of 8 protons, 7 neutrons, and 8 electrons.
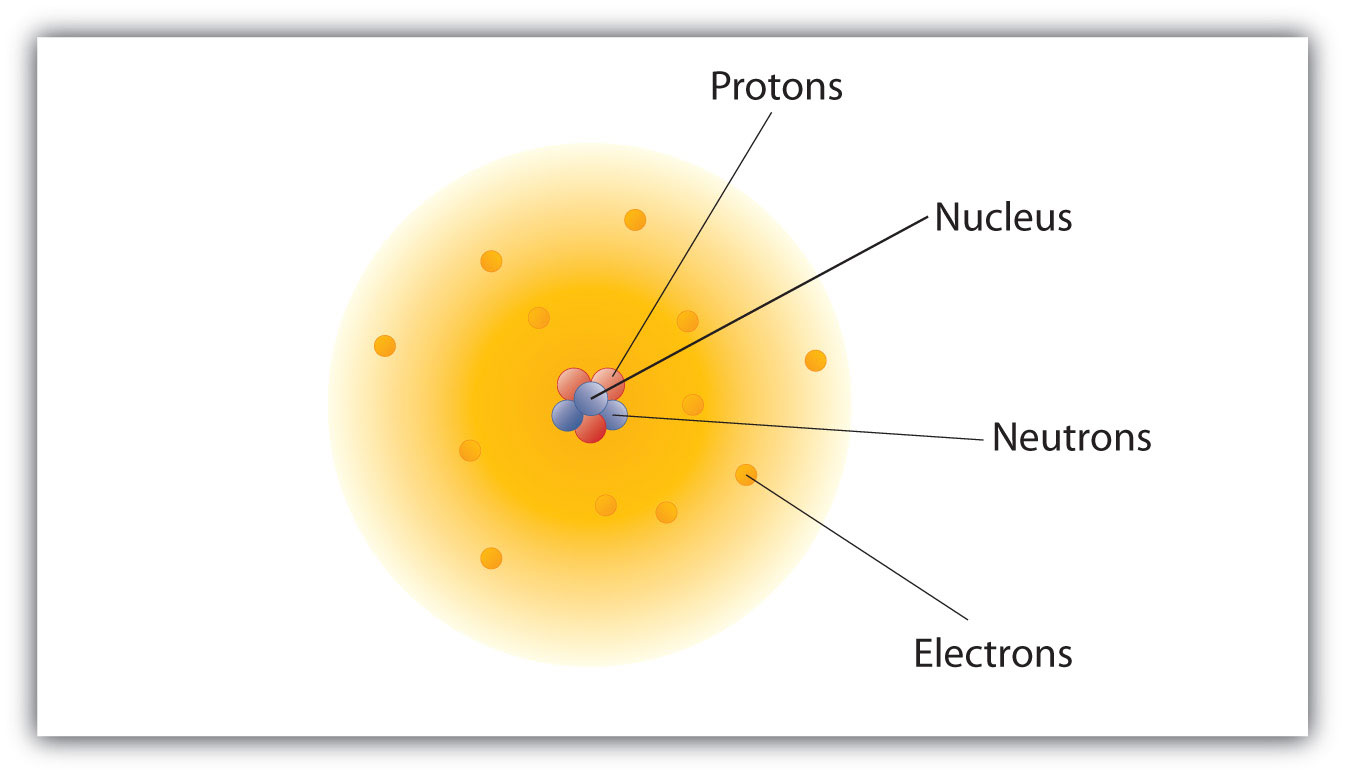
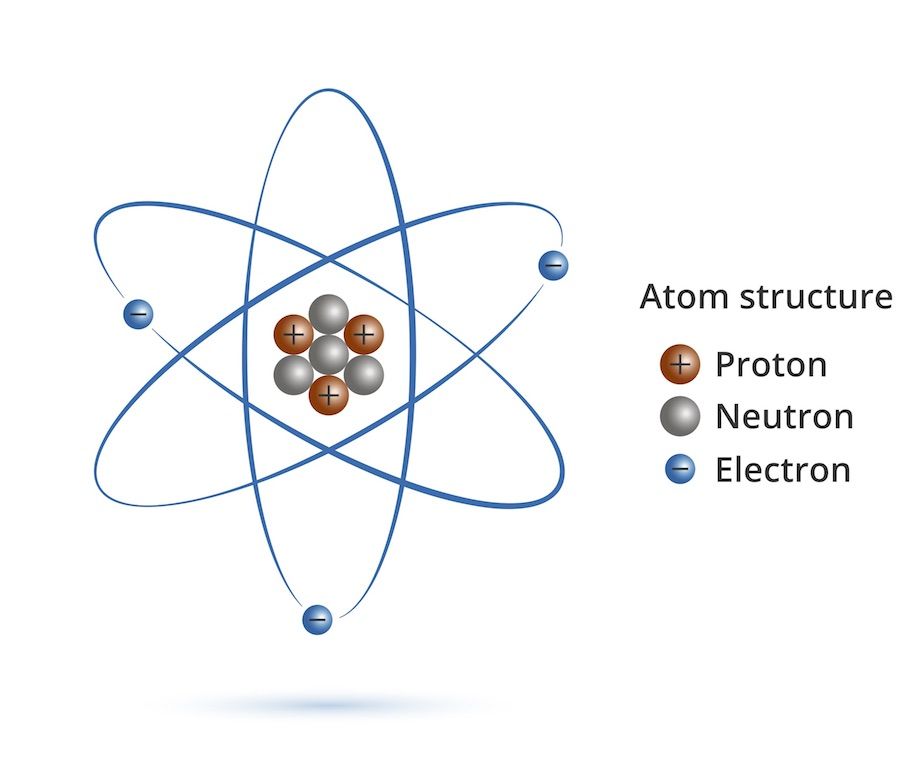
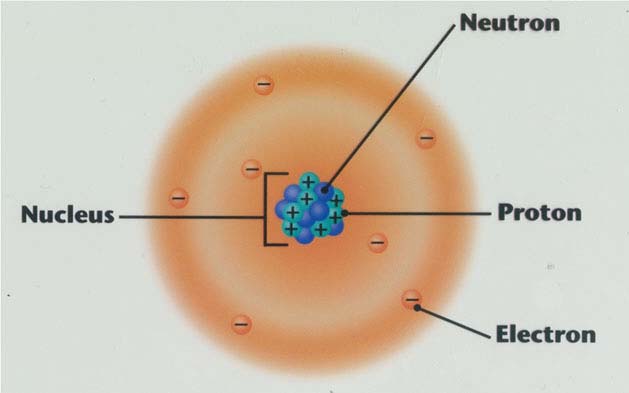
Protons, Electrons, and Neutrons Protons and neutrons are nuclear particles These particles reside inside the nucleus of an atom Electrons revolve around the nucleus of an atom. Chemistry General Chemistry Standalone book (MindTap Course List) Naturally occurring nitrogen is a mixture of 14 N and 15 N Give the number of protons, neutrons, and electrons in the neutral atom of each isotope. Determine the number of protons, neutrons and electrons in each and name them atomic number 26, mass number 58, charge of 2 atomic number 53, mass number 127, charge of 1− Answer a Iron, 26 protons, 24 electrons, and 32 neutrons Answer b Iodine, 53 protons, 54 electrons, and 74 neutrons.
Atoms having the same number of neutrons but different number of protons or mass number, eg 6 C 14, 8 O 16, 7 N 15 Isoelectronic Atoms, molecules or ions having same number of electrons eg N 2,CO, CN – Nuclear isomers atoms with the same atomic number and same mass number but with different radioactive properties Example of nuclear. APrerence of isotopes b Number of unpaired electrons c Spherical shape d Quantum number 9 Which of the following are not isotopes a 1 H 1 and 1 H 3 b 18 K 40 and Ca 40 c 6 C 14 and 7 N 14 d Both b and c 10 Charge present in the. An atom of the isotope 16 S31 consists of how many protons, neutrons, and electrons?.
Chemistry General Chemistry Standalone book (MindTap Course List) Naturally occurring nitrogen is a mixture of 14 N and 15 N Give the number of protons, neutrons, and electrons in the neutral atom of each isotope. The atomic nucleus is composed of protons and neutrons (Figure \(\PageIndex{1}\)) Protons and neutrons have approximately the same mass, but protons carry one unit of positive charge (e) and neutrons carry no charge These particles are packed together into an extremely small space at the center of an atom. This will give you the number of neutrons For example, carbon on periodic table is atomic number 6 That means 6 protons Above, it says Carbon12 12 is the mass number "Mass number" "Atomic number" = "neutrons" 126 = 6 neutrons Carbon 6 protons, 6 neutronsand so on One more Nitrogen Nitrogen is atomic number 7.
Give the nuclear composition and isotopic notation for (a) an atom containing 27 protons, 32 neutrons, and 27 electrons (b) an atom containing 15 protons, 16 neutrons, and 15 electrons (c) an atom containing 110 neutrons, 74 electrons, and 74 protons. Protons7 neutrons 15 electrons 7 Draw the complete symbol of an ion of Carbon14?. Boron is a chemical element with atomic number 5 which means there are 5 protons and 5 electrons in the atomic structureThe chemical symbol for Boron is B Significant concentrations of boron occur on the Earth in compounds known as the borate minerals There are over 100 different borate minerals, but the most common are borax, kernite, ulexite etc Natural boron consists primarily of two.
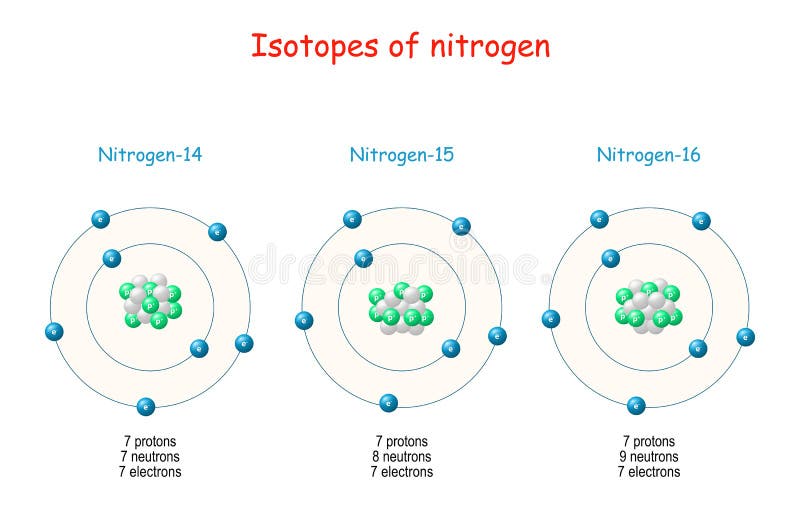
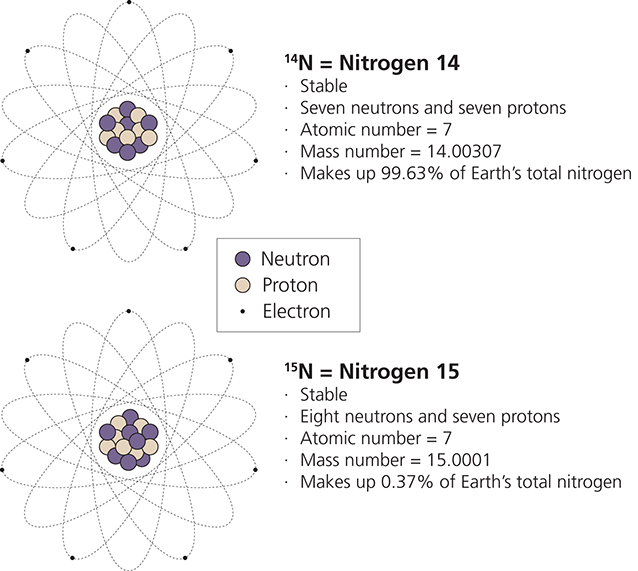
Since both N14 and N15 are neutral and have 7 protons, then there must also be 7 electrons in each of these isotopes It is in the number of neutrons that N14 and N15 differ The numbers in. List the number of protons, electrons, and neutrons for an atom of silver pro 47 nuet 61 elect 47 How many protons, electrons, and neutrons are in the isotope N15?. Since both N14 and N15 are neutral and have 7 protons, then there must also be 7 electrons in each of these isotopes It is in the number of neutrons that N14 and N15 differ.
Calculate the number of protons, neutrons, and electrons for an isotope of Nitrogen, N15 (Atomic mass of 15) 3 Complete the following table for neutral atoms and ions Symbol Atomic number Atomic mass Number of protons Number of neutrons Number of electrons Mg 2 S 2 26 norintion) for each of the following elements 5. N14 > 14 minus 7 = 7 and N15 > 15 7 = 8 Notice that 7 is the atomic number of nitrogen and I used it in both subtractions nmber of electrons you have to know the charge on the atom Suppose I have an atom of magnesium and it is neutral How many electrons?. How many neutrons in each?.
Get the detailed answer How many protons, neutrons, and electrons are in a neutral atom of sodium?. The atomic nucleus is composed of protons and neutrons (Figure \(\PageIndex{1}\)) Protons and neutrons have approximately the same mass, but protons carry one unit of positive charge (e) and neutrons carry no charge These particles are packed together into an extremely small space at the center of an atom. A Protons c Electrons b Neutrons d Nucleons 8 What makes the atomic mass fractional ?.
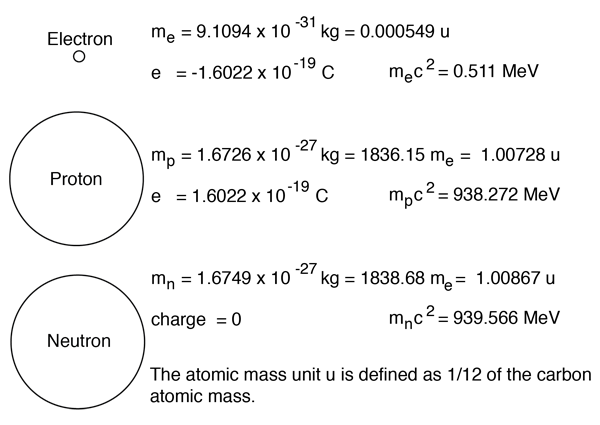
Atoms—and the protons, neutrons, and electrons that compose them—are extremely small For example, a carbon atom weighs less than 2 × 1023 g, and an electron has a charge of less than 2 × 1019 C (coulomb) When describing the properties of tiny objects such as atoms, we use appropriately small units of measure, such as the atomic mass unit (amu) and the fundamental unit of charge (e). The only naturally occurring isotopes of nitrogen are N14 and N15 1 State the number of protons in an atom of N15 1 2 State the number of electrons in each shell of a N14 atom in the ground state 1 3 Based on the atomic mass of the element nitrogen on the Periodic Table, compare the relative abundances of the naturally occurring isotopes of nitrogen. Oxygen17 is composed of 8 protons, 9 neutrons, and 8 electrons Both 17O and 18O are secondary isotopes, meaning that their nucleosynthesis requires seed nuclei Oxygen18 is composed of 8 protons, 10 neutrons, and 8 electrons Oxygen15, which is an unstable isotope of oxygen, is composed of 8 protons, 7 neutrons, and 8 electrons.
Subtracting the number of protons (7) from the element's mass number (14) makes it possible to determine the number of neutrons in the atom Many nitrogen atoms have seven neutrons (147 = 7) Some atoms of nitrogen, however, have eight electrons They are atoms of N15, an isotope of nitrogen. Name One day I will buy Also bought rings for research Atoms are made of protons, neutrons, and electrons a) 39 protons, 50 neutrons, 39 electrons b) 39 protons, neutrons, 39 electrons c) 39 protons, 50 neutrons, 50 electrons d) 50 protons, 39 neutrons, 50 electrons e) 39 protons, 11 neutrons, 39 electrons. N14 > 14 minus 7 = 7 and N15 > 15 7 = 8 Notice that 7 is the atomic number of nitrogen and I used it in both subtractions nmber of electrons you have to know the charge on the atom Suppose I have an atom of magnesium and it is neutral How many electrons?.
Switch to Home Your dashboard and recommendations Booster Classes Personalized courses, with or without credits Homework Help 37 million tough questions answered Study Guides Ace your next exam with ease. Identifying Protons, Neutrons And Electrons Of Atoms And Ions How To Discover Electrons A duration is all the aspects had in a straight row The atomic mass can be located below the icon for the elementMake sure that you round the atomic mass to the local whole number As an example, the atomic mass of boron is , however you can simply. A) 15 p, 16 n, 15 e b) 16 p, 15 n, 16 e c) 16 p, 31 n, 16 e d) 32 p, 31 n, 32 e A magnesium ion, Ca 2, has a) protons and 22 electrons b) protons and electrons c) protons and 18 electrons d) 22 protons and electrons 21.
Atomic Number – Protons, Electrons and Neutrons in Phosphorus Phosphorus is a chemical element with atomic number 15 which means there are 15 protons in its nucleusTotal number of protons in the nucleus is called the atomic number of the atom and is given the symbol ZThe total electrical charge of the nucleus is therefore Ze, where e (elementary charge) equals to 1,602 x 1019 coulombs. Get the detailed answer How many protons, neutrons, and electrons are in a neutral atom of sodium?. Nitrogen14 is composed of 7 protons, 7 neutrons, and 7 electrons 1)if a boron atom has 5 protons, 6 neutrons, and 5 electrons, it has a mass of a)11 b)10 c)6 2)the atomic number of an atom is the total number of in the nucleus.
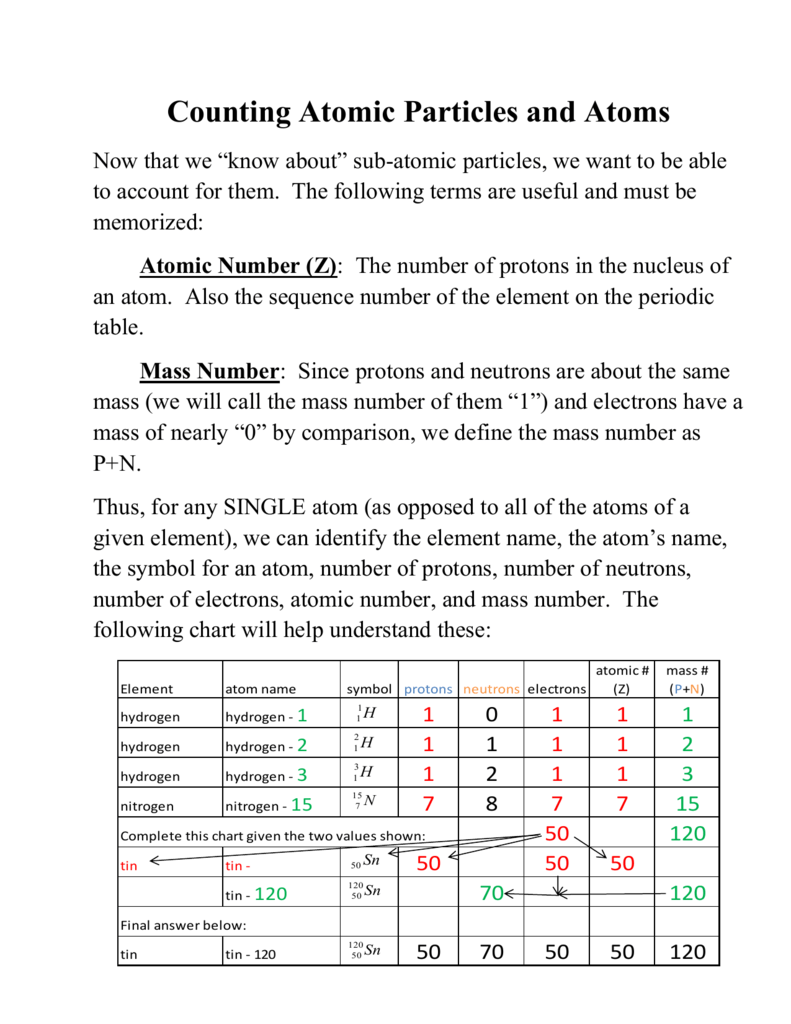
So the total number of neutrons and protons in the nucleus is 15 Find the number of neutrons N = 15 – 7 = 8 The 15N isotope has 8 neutrons in its nucleus Let’s calculate the number of neutrons for the 119Sn isotope Tin in the periodic system is numbered 50, which means that an atom contains 50 electrons and 50 protons Relative atomic. For any element, the number of protons and electrons is equal to the atomic number, so phosphorus has 15 Protons and electrons As for the neutrons, that differs depending on the isotope you are dealing with For phosphorus, the most common number of neutrons is 16. N14 > 14 minus 7 = 7 and N15 > 15 7 = 8.
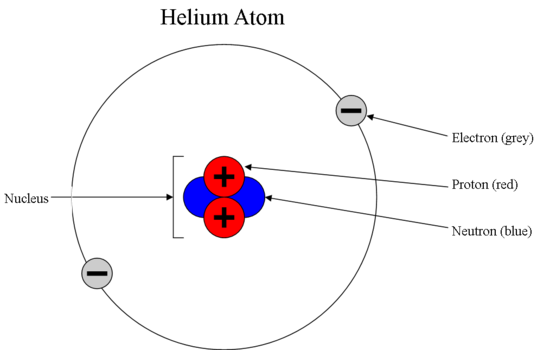
How many neutrons in each?. N15 protons 7, electrons 7, neutrons 8 What is the number protons, electrons, and neutrons for each of the following isotopes of Lithium (atomic number 3) Li6 Li7 Li8 Li6 protons 3, electrons 3, neutrons 3 Li7 protons 3, electrons 3, neutrons 4 Li8 protons 3, electrons 3, neutrons 5. The table below compares the properties of protons, neutrons, and electrons Protons and neutrons have about the same mass Electrons have a much smaller mass than protons or neutrons As a result, the nucleus contains most of an atom’s mass All atoms contain protons, neutrons, and electrons However, different types of atoms have different.
How many protons, neutrons and electrons are present in one atom of the following 1 H 2 H 3 H 13 N 14 N 15 N 79 Br 81 Br 1262 Bh What is the Atomic Weight of chlorine given that The exact mass of 35 Cl is 3496 and the relative abundance is 7553% The exact mass of 37 Cl is and the relative abundance is 2447%. Any of two or more species of atoms or nuclei that have the same number of neutrons c Any of two or more species of atoms or nuclei that have the same number of electrons 2 An atom of the isotope sulfur31 consists of how many protons, neutrons and electrons a 15 p, 16 n, 15 e b 16 p, 15 n, 16 e c 16 p, 31 n, 16 e d 32 p, 31 n, 32 e 3. Any of two or more species of atoms or nuclei that have the same number of neutrons c Any of two or more species of atoms or nuclei that have the same number of electrons 2 An atom of the isotope sulfur31 consists of how many protons, neutrons and electrons a 15 p, 16 n, 15 e b 16 p, 15 n, 16 e c 16 p, 31 n, 16 e d 32 p, 31 n, 32 e 3.
Determine the number of protons, neutrons and electrons in each and name them atomic number 26, mass number 58, charge of 2 atomic number 53, mass number 127, charge of 1− Answer a Iron, 26 protons, 24 electrons, and 32 neutrons Answer b Iodine, 53 protons, 54 electrons, and 74 neutrons. So the total number of neutrons and protons in the nucleus is 15 Find the number of neutrons N = 15 – 7 = 8 The 15N isotope has 8 neutrons in its nucleus Let’s calculate the number of neutrons for the 119Sn isotope Tin in the periodic system is numbered 50, which means that an atom contains 50 electrons and 50 protons Relative atomic. The atomic nucleus is composed of protons and neutrons (Figure \(\PageIndex{1}\)) Protons and neutrons have approximately the same mass, but protons carry one unit of positive charge (e) and neutrons carry no charge These particles are packed together into an extremely small space at the center of an atom.
The number after the "N", 15, is the mass number The mass number is the sum of the number of protons and the number of neutrons So, if you find a periodic table, you can find the atomic number. Switch to Home Your dashboard and recommendations Booster Classes Personalized courses, with or without credits Homework Help 37 million tough questions answered Study Guides Ace your next exam with ease. Atoms are made of protons, neutrons, and electrons Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral A neutral atom has the same number of protons and electrons (charges cancel each other out) An ion has an unequal number of protons and electrons.
So the total number of neutrons and protons in the nucleus is 15 Find the number of neutrons N = 15 – 7 = 8 The 15N isotope has 8 neutrons in its nucleus Let’s calculate the number of neutrons for the 119Sn isotope Tin in the periodic system is numbered 50, which means that an atom contains 50 electrons and 50 protons Relative atomic. The atomic mass refers to the number of protons and neutrons in an average isotope of the element Nitrogen on the Periodic Table has an Atomic Mass of The Nitrogen14 isotope makes up 996% of Nitrogen found in nature. Protons, Electrons, and Neutrons Protons and neutrons are nuclear particles These particles reside inside the nucleus of an atom Electrons revolve around the nucleus of an atom.
Atoms having the same number of neutrons but different number of protons or mass number, eg 6 C 14, 8 O 16, 7 N 15 Isoelectronic Atoms, molecules or ions having same number of electrons eg N 2,CO, CN – Nuclear isomers atoms with the same atomic number and same mass number but with different radioactive properties Example of nuclear. THIS SET IS OFTEN IN FOLDERS WITH elements quiz. Any of two or more species of atoms or nuclei that have the same number of neutrons c Any of two or more species of atoms or nuclei that have the same number of electrons 2 An atom of the isotope sulfur31 consists of how many protons, neutrons and electrons a 15 p, 16 n, 15 e b 16 p, 15 n, 16 e c 16 p, 31 n, 16 e d 32 p, 31 n, 32 e 3.

Chapter 5 P

Phosphorus Has The Atomic Number 15 And An Atomic Mass Of 31 Arnu How Many Protons Neutrons And Electrons Are In A Neutral Phosphorus Atom Homework Help And Answers Slader
Www Saultschools Org Cms Lib Mi Centricity Domain 222 Atoms family answers Pdf
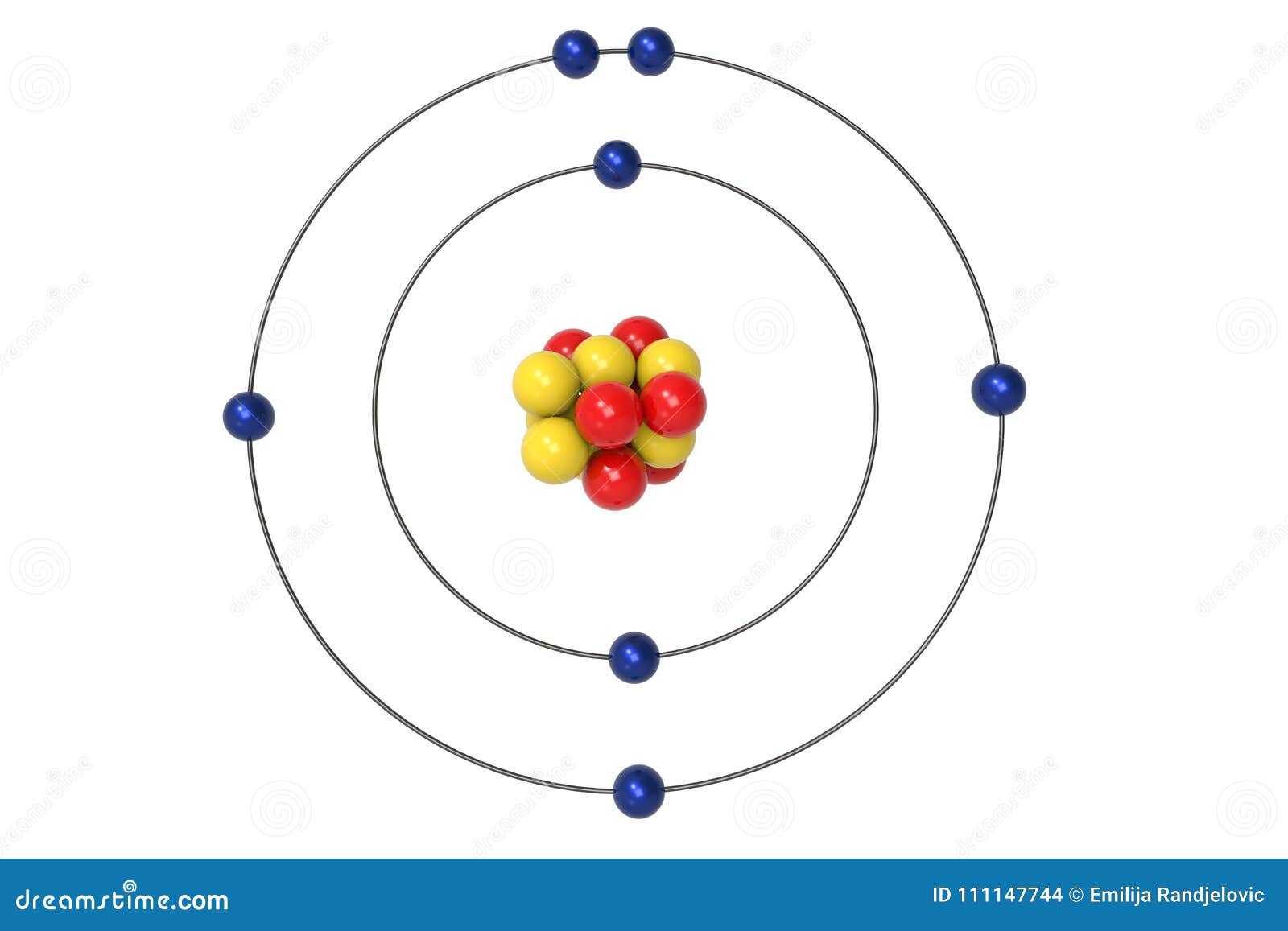
Nitrogen Atom Bohr Model With Proton Neutron And Electron Stock Illustration Illustration Of Background Biomolecule

2 Atoms Molecules And Ions Exercises Chemistry Libretexts
Atoms And Elements
Www Cpsk12 Org Cms Lib8 Mo Centricity Domain 3297 Atomic structure worksheet answers Pdf
Q Tbn And9gcreke2614cb4kchc9dtamvnuakfwio9pzeiq9acusxjrps9 Bh2 Usqp Cau

Physical Science The Structure Of Matter Ppt Video Online Download
Structure Reactivity Atoms Protons Neutrons Electrons

Solved 5 Practice A Show How Nitrogen 14 And Nitrogen Chegg Com

The Number Of Electrons Protons And Neutrons In A Species Are Equ
Ion Isotope Practice Key Youtube

Cbse Ncert Solution For Class 9 Chemistry Structure Of The Atom
Atomic Theory
What Are Isotopes Quora

Atomic Structure And Symbolism Chemistry I

8 5a Atomic Structure Flashcards Quizlet
Nitrogen Isotopes Stock Illustrations 8 Nitrogen Isotopes Stock Illustrations Vectors Clipart Dreamstime
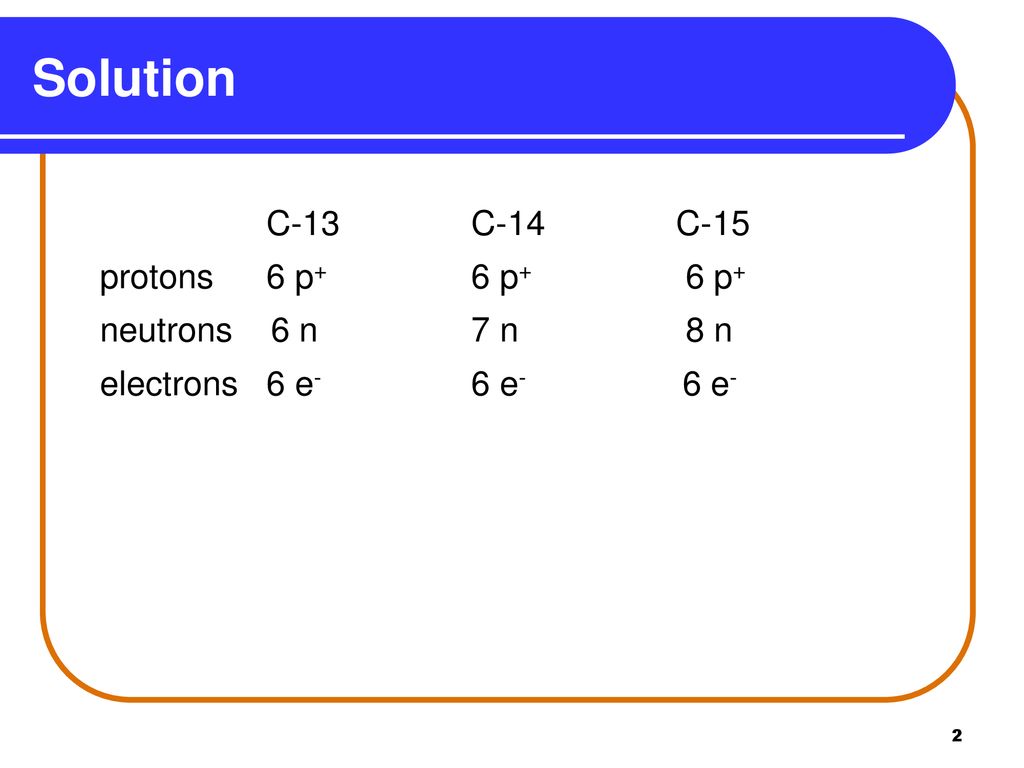
Learning Check Naturally Occurring Carbon Consists Of Three Isotopes Carbon 13 Carbon 14 And Carbon 15 State The Number Of Protons Neutrons And Ppt Download
Www Everettcc Edu Files Programs Academic Resources Transitional Studies Support Tutoring Center Chemistry W310 Protons Neutrons And Electrons Worksheet Pdf

2 Which Of The Following Describes The Difference Between Nitrogen 14 And Nitrogen 15 1 Point Brainly Com

How To Find The Number Of Protons Electrons Neutrons For Nitrogen N Youtube

1 01 Subatomic Particles Protons Neutrons Electrons And The Nucleus Week 1 Coursera

Dublin Schools Lesson Isotopes How Do The Number Of Subatomic Particles Differ For Atoms From The Same Element

How Is The N 15 Isotope Different From The N 14 Atom A N 15 Has One Less Proton Than N 14 B N 15 Has One Extra Neutron C N 15 Has One Extra Electron D N 15
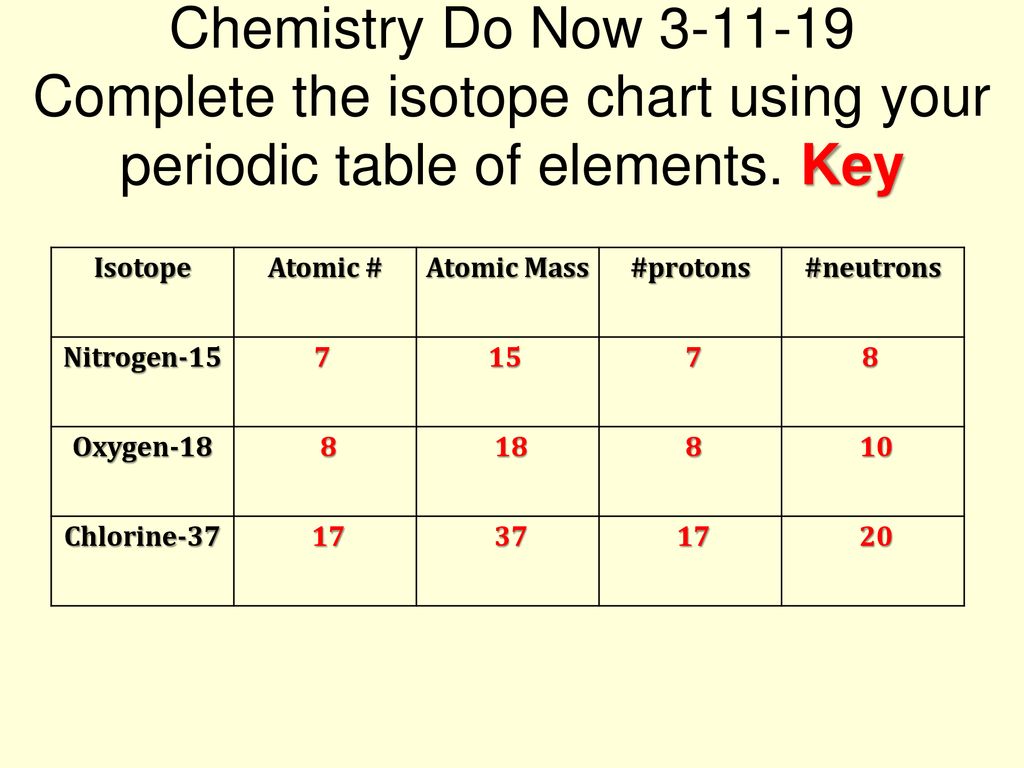
Atomic Atomic Mass Protons Neutrons Nitrogen 15 Oxygen Ppt Download

Lecture 4 Atoms And Atomic Structure Protons Neutrons And Electrons The Atom Is The Smallest Building Block Of An Element The Atom Is Composed Of A Core Called The Nucleus Where Most Of The Atoms Mass Is Concentrated The Nucleus Is Composed Of Two

Science Atomic Structure Flashcards Quizlet
What Is An Atom Live Science

Name Chemistry Teacher Period Mini

Solved Complete The Blanks Below For Each Element The Nu Chegg Com

What Are The First Elements Names And Symbols

The Ion N3 Has Protons And Clutch Prep

Atom Definition Structure History Examples Diagram Facts Britannica

Isotopes Of Nitrogen Stock Illustration

Solved Isotopes Atomic Number Mass Number And Number O Chegg Com

Atomic Structure Nucleons Atomic Number Ppt Download

Ppt Chapter 3 Atoms And Elements Powerpoint Presentation Free Download Id
Subatomic Particles Nemoquiz

Fall 11 Week 5 Atomic Structure Ppt Video Online Download

Nova Online Mystery Of The First Americans Close Encounters
Www Manhassetschools Org Cms Lib Ny Centricity Domain 796 Atoms practice qs answer key Pdf

How To Find The Number Of Protons Electrons Neutrons For Magnesium Mg Youtube

Atomic Structure Electrons Protons Neutrons And Atomic Models
1 Carbon 12 Atom Has A Mass Of Exactly 12 Amu

Radioactivity And Radiolabeling The Bumbling Biochemist

Spsphysicalscience Licensed For Non Commercial Use Only Ii C 2 Elements And Compounds Assessment
2 3 Atomic Structure And Symbolism General Chemistry 1 2
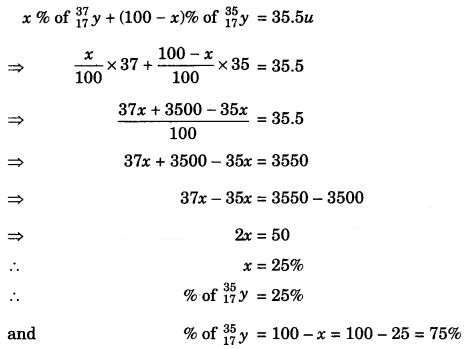
Structure Of The Atom Class 9 Extra Questions And Answers Science Chapter 4

How Many Neutrons And Protons Are There In The Following Nuclei 6 13c 8 16o 12 24mg 26 56fe 38 sr
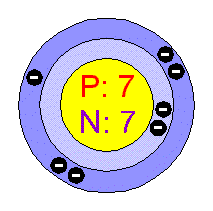
Chemical Elements Com Nitrogen N

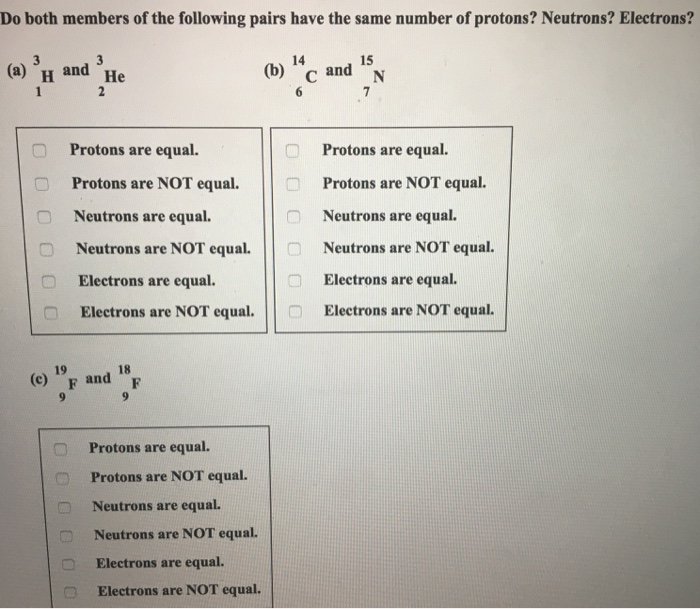
Solved Do Both Members Of The Following Pairs Have The Sa Chegg Com
How Solar Cells Work

10 15 Review Atomic Structure

Q11 Draw The Atomic Diagrams Of The Following Elements Showi Lido
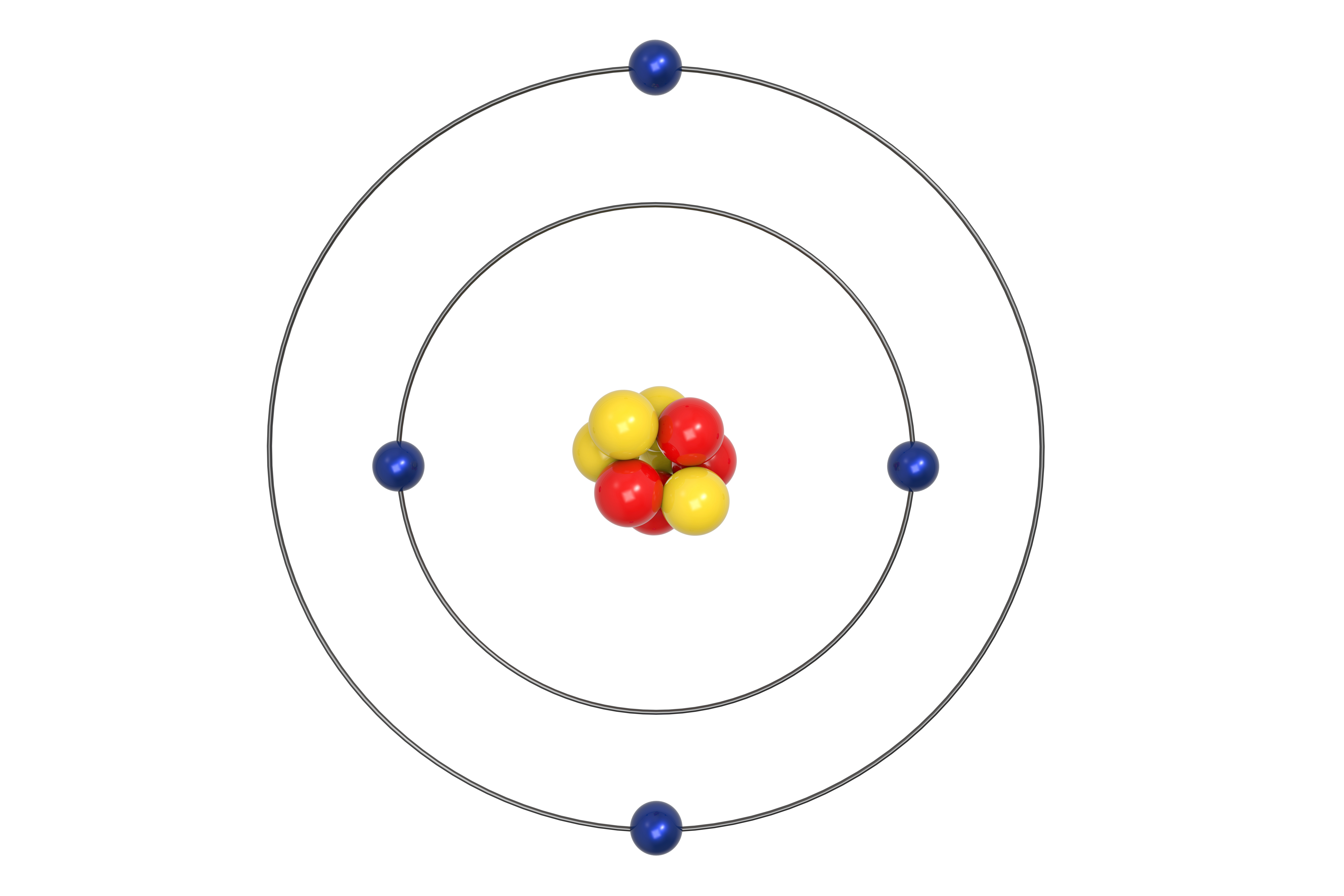
How To Find How Many Protons Neutrons Electrons Are In Isotopes

Atoms Ppt Download

B What Are The Stable Isotopes Of Nitrogen N 14 N 15 C List Two Radioactive Course Hero
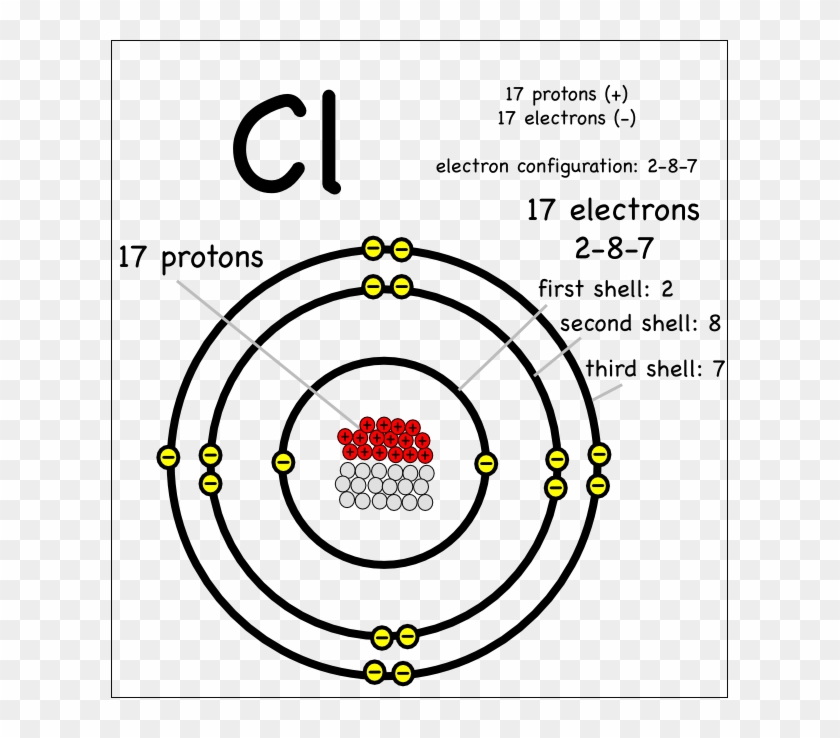
An Introduction To Ionic Bonding Montessori Muddle Mg Element Protons Neutrons Electrons Free Transparent Png Clipart Images Download

Isotopes Nitrogen Diagram Structure Nitrogen Atoms Stock Vector Royalty Free
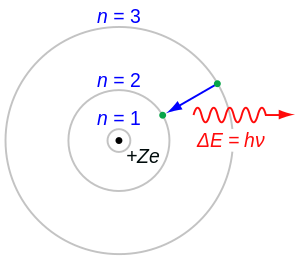
Atomic Number Wikipedia

Radioactivity And Radiolabeling The Bumbling Biochemist

Isotopes Chemistry Socratic

Luj Zu 5 Calculate The Number Of Electrons Protons And Neutrons In The Following Molecules Ions
How Many Protons Neutrons And Electrons Are In Silicon Quora

Atoms And Elements The Nature Of Matter Matter Refers To Anything That Takes Up Space And Has Mass Matter Refers To Anything That Takes Up Space And Ppt Download

Lecture 4 Atoms And Atomic Structure Protons Neutrons And Electrons The Atom Is The Smallest Building Block Of An Element The Atom Is Composed Of A Core Called The Nucleus Where Most Of The Atoms Mass Is Concentrated The Nucleus Is Composed Of Two
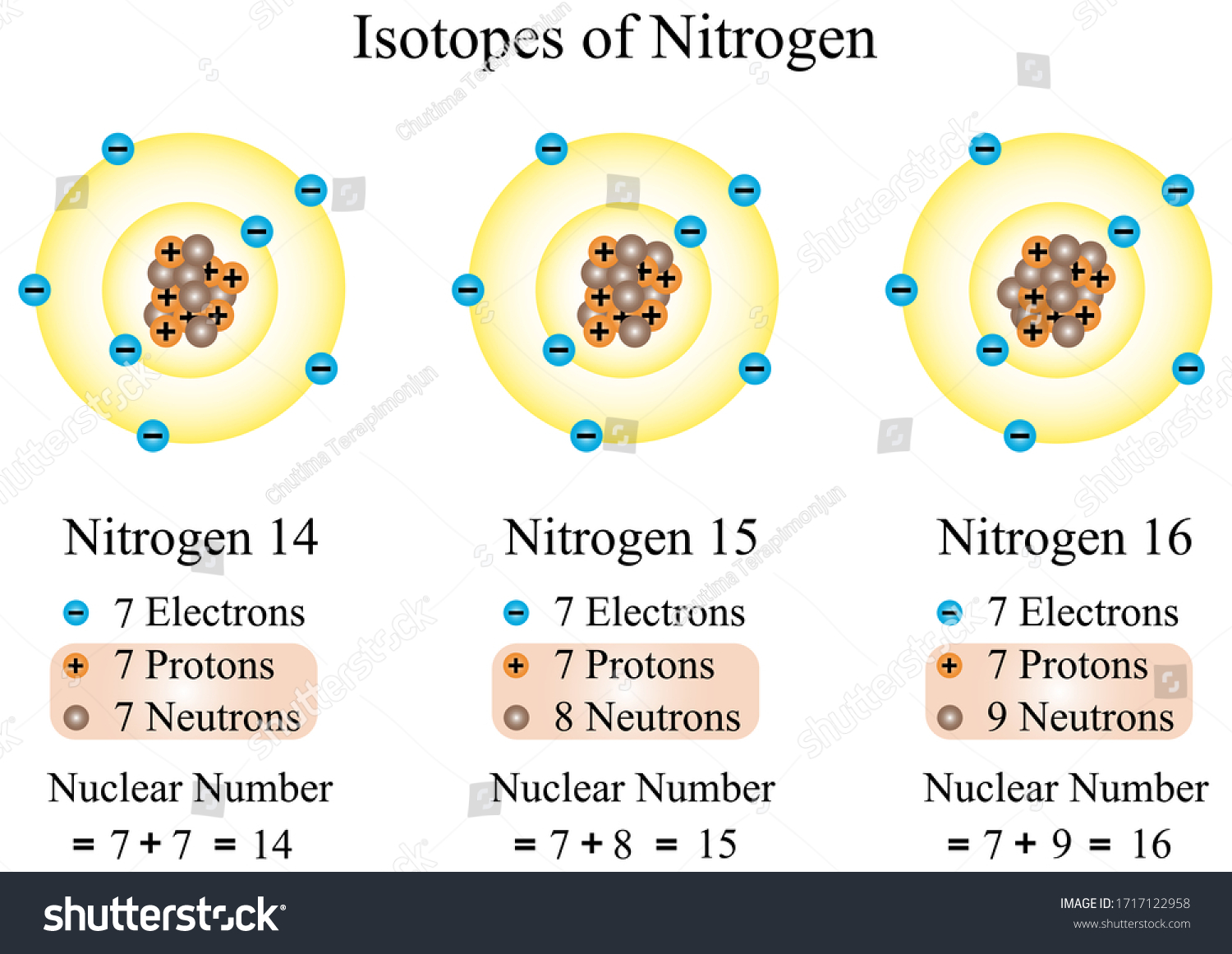
Illustration Chemical Isotopes Nitrogen All Atoms Stock Vector Royalty Free
Isotopes In Environmental Science U S National Park Service

The Atom And The Periodic Table Ppt Video Online Download

The Number Of Electrons Protons And Neutrons In A Species Are Equ

Atomic Number Mass Number And Isotopes Video Khan Academy

Isotopes Answers Doc Isotope Proton

Introduction To The Atom Protons Neutrons Electron Nucleus Atomi
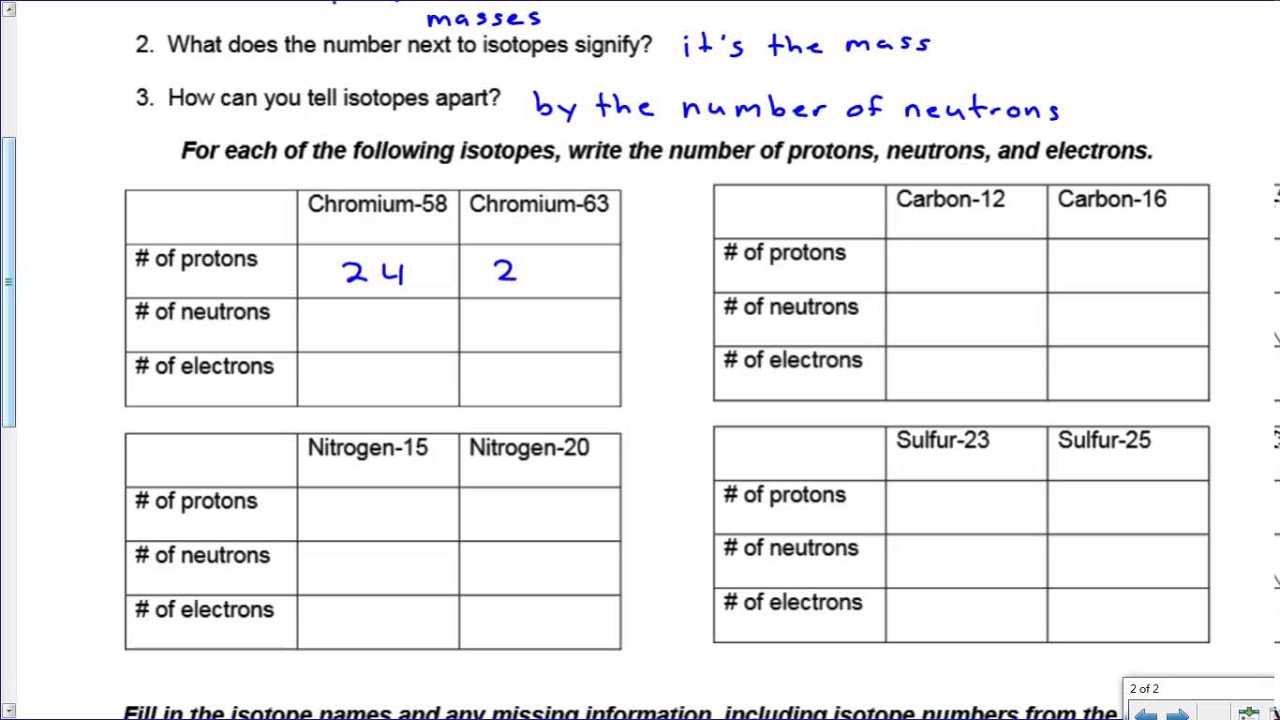
Solved 3 22 Nitrogen 15 Nitrogen Sulfur 23 Sulfur 25 Chegg Com
Sub Atomic Particles Chemistry Libretexts

Atom Wikipedia

File Electron Shell 007 Nitrogen Svg Wikimedia Commons Electron Configuration Atom Diagram Electrons

Atoms And The Periodic Table By The End Of This Unit You Should Be Able To Explain Ppt Download

10 15 Review Atomic Structure
Q Tbn And9gcrqsfkbpshao7szli0adyzwe2lbthton Gifrjf24hcv0eko3zq Usqp Cau

Chm 45 Week 1 Practices F Studocu

Cbse Ncert Solution For Class 9 Chemistry Structure Of The Atom
Q Tbn And9gcrqsfkbpshao7szli0adyzwe2lbthton Gifrjf24hcv0eko3zq Usqp Cau
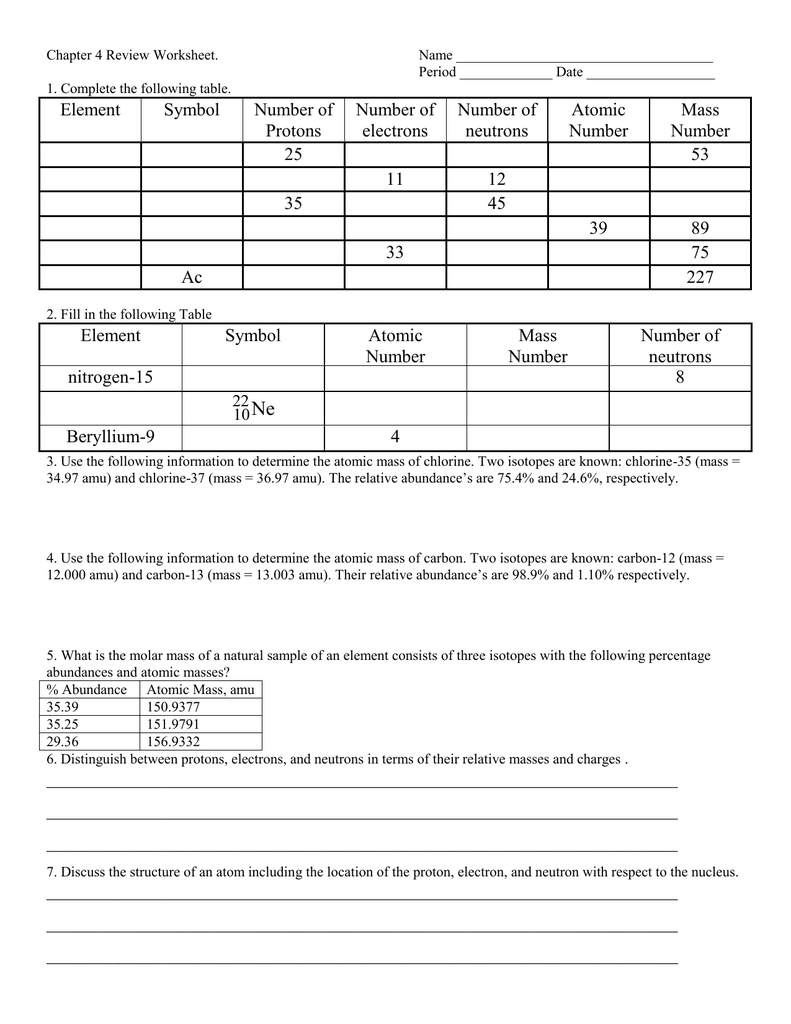
Chapter 4 Review Worksheet Name

How To Find The Number Of Protons Electrons Neutrons For Nitrogen N Youtube
Www Gateschili Org Site Handlers Filedownload Ashx Moduleinstanceid 2113 Dataid 9934 Filename Unit 3 quiz 1 practice quiz key Pdf
Http Centritto Weebly Com Uploads 4 2 7 6 Topic 2 12 Mc Practice Pdf
Just What Is An Isotope And How Can We Use It To Unlock The Secrets Of Deep Water Sharks Save Our Seas Foundation
Www Sd308 Org Cms Lib Il Centricity Domain 2521 Beanium lab scanned Pdf

Slides Show

Ncert Exemplar Class 9 Science Solutions Chapter 4 Structure Of Atom Access Free Pdf

Chapter 2 Atoms Ions And The Periodic Table Pdf Free Download

Solution How Many Protons Neutrons And E Clutch Prep
Solved Do Both Members Of The Following Pairs Have The Sa Chegg Com

Q11 Draw The Atomic Diagrams Of The Following Elements Showi Lido



