W Element Protons Neutrons Electrons

Atomic Structure Protons Electrons Neutrons Youtube
Www Topperlearning Com Answer I Want The Symbol Number Of Protons Number Of Neutrons Number Of Electrons Distribution Of Electrons And Valency Of The First 30 Elements 3y90wojj

Slides Show
:max_bytes(150000):strip_icc()/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
Atom Diagrams Electron Configurations Of The Elements
Periodic Table Of Elements List With Protons Neutrons And Electrons

A Atomic Number
Cobalt60 is composed of 27 protons, 33 neutrons, and 27 electrons Cobalt60 (60Co or Co60) is a radioactive metal that is used in radiotherapy It produces two gamma rays with energies of 117 MeV and 133 MeV.

W element protons neutrons electrons. Number of Protons 53 Number of Neutrons 74 Number of Electrons 53 Melting Point 1135° C Boiling Point 1840° C Density 493 grams per cubic centimeter Normal Phase Solid Family Halogens Period 5 Cost $0 per 100 grams. So not all argon atoms have 22 neutrons, but most of them do in the natural world of 36 and 38 18 is Argon with symbol Ar Now we need to find the mass number of Argon Part I Protons, Electrons and Neutrons, continued Part !. Number of protons = 11 Number of electrons = 11 Number of neutrons (mass number atomic number) = 23 11 = 12.
Rare Earth Elements Basic Information Atomic Structure Isotopes Related Links Citing This Page Basic Information Name Radon Symbol Rn Atomic Number 86 Atomic Mass (22) amu Melting Point710 °C (215 K, 958 °F) Boiling Point618 °C ( K, 7924 °F) Number of Protons/Electrons 86 Number of Neutrons 136. *I recommend watching this in x125 15 speed This video goes over how to know how many Protons, Electrons, and Neutrons are in any given element whether o. What element has 27 protons and 33 neutrons Posted by February 18, 21.
How to find the Protons Neutrons and Electrons of an element on the Periodic Table Thursday, May 28, How many protons, neutrons, and electrons does Boron have?. On the periodic table, argon's atomic number is 18 Neutral Atom of Chlorine Atomic Number= 1. # of protons = 17 # of neutrons = 37 – 17 = # of electrons = 17 – 0 = 17 # of protons = 16 (the atomic number is not given, but can be found on the periodic table) # of neutrons = 32 – 16 = 16 # of electrons = 16 – (2) = 18 Additional Practice Try these on your own and check the answer below 78 Se 239 K ANSWERS 34 protons, 44.
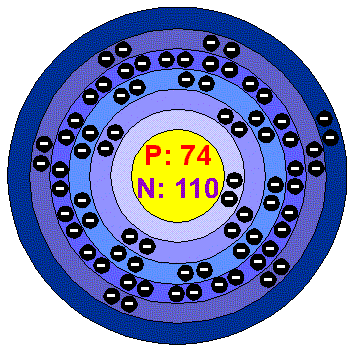
Electrons, protons and neutrons are the subatomic particles found inside every atom Protons are positively charged particles that are found in the nucleus of an atom. The addition or removal of (protons, neutrons, electrons) causes the element name to change 2 Atomic number is equal to the number of (protons, neutrons, electrons) 3 Atomic mass is equal to the number of (protons, neutrons, electrons) plus the number of (protons, neutrons, electrons) 4. So the nucleus contains 74 protons and 110 neutrons (the total = 184) A neutral atom contains the same number of electrons as it has protons, so a neutral tungsten atom has 74 electrons in the.
To know how to find protons, neutrons, and electrons in an isotope, let us consider the example of Chlorine isotopes, Chlorine – 35 ( 17 Cl 35) and Chlorine – 37 ( 17 Cl 37) Chlorine – 35 (17Cl35) Number of Electron = Number of Proton = Atomic Number = 17 Mass Number = 35. So the nucleus contains 74 protons and 110 neutrons (the total = 184) A neutral atom contains the same number of electrons as it has protons, so a neutral tungsten atom has 74 electrons in the. Protons,Electrons and Neutrons This page is an exercise in relating the number of protons, electrons and neutrons for an atom or monoatomic ion When you press "New Problem", an atomic symbol will appear in the first cell and several other cells will have values.
Tungsten hexacarbonyl W(CO) 6. Periodic Table Basics Learn how to use information from the periodic table to find the number of protons, neutrons, and electrons of an element The atomic n. The atomic number of an element is always the same as _____ the number of protons plus neutrons the number of neutrons the number of electrons the number of protons The atomic mass is the number of protons plus the number of neutrons.
For most of the remaining elements there are more neutrons than protons because extra neutrons are needed to keep the nucleus together by overcoming the mutual repulsion of the increasing numbers of protons concentrated in a very small space For example, silicon has 14 protons and 14 neutrons Its atomic number is 14 and its atomic mass is 28. The number of protons and electrons in an element are equal The number of protons and neutrons may be equal for some elements, but are not equal for all Figure 24 The Periodic Table of the Elements (credit RA Dragoset, A Musgrove, CW Clark, WC Martin). A particular atom will have the same number of protons and electrons and most atoms have at least as many neutrons as protons An element is a substance that is made entirely from one type of atom For example, the element hydrogen is made from atoms containing just one proton and one electron If you had very, very good eyes and could look at the atoms in a sample of hydrogen, you would notice that most of the atoms have no neutrons, some of them have one neutron and a few of them have two.
For example, a neutral chlorine atom has 17 protons and 17 electrons, whereas a Cl − anion has 17 protons and 18 electrons for a total charge of −1 All atoms of a given element are not necessarily identical, however The number of neutrons may vary to form different isotopes, and energy levels may differ, resulting in different nuclear. The periodic table provides a wealth of information for the elements However, if you can't read the periodic table it is not very helpful. For most of the remaining elements, there are more neutrons than protons, because extra.
Protons are the atomic number which is 13 14 (added onto the aluminum) = 27 therefore when you draw out the lewis dot it should have 3 on the outer shell 14 = 17 electrons if the protons and. All atoms contain protons, electrons, and neutrons (Figure 2) The only exception is hydrogen (H), which is made of one proton and one electron A proton is a positively charged particle that resides in the nucleus (the core of the atom) of an atom and has a mass of 1 and a charge of 1 Neutrons, like protons, reside in the nucleus of an atom They have a mass of 1 and no charge. Answers 1 Show answers Another question on Chemistry Chemistry, 2619 00 14 compare between the plasticity of edge and screw dislocations in bcc metals 15 why does a screw dislocation in a bcc metal needs high.
A 2page worksheet that gives students practice interpreting information on the periodic table Specifically, this worksheet challenges students to use an element's atomic number and mass number to calculate the number of protons, electrons and neutrons in 1 atom of that elementI use this workshee. Start studying Protons, Electrons, and Neutrons?, ROUNDING w/ SIGNIFICANT FIGURES, Converting with Metrics, Scientific notation and Standard Notation Learn vocabulary, terms, and more with flashcards, games, and other study tools. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number For hydrogen, the number of protons is 1 For zinc, the number of protons is 30 The element of an atom with 2 protons is always helium read more.
When you compare the masses of electrons, protons, and neutrons, what you find is that electrons have an extremely small mass, compared to either protons or neutrons On the other hand, the masses of protons and neutrons are fairly similar, although technically, the mass of a neutron is slightly larger than the mass of a proton. What element has 27 protons and 33 neutrons by Feb 18, 21 Uncategorized 0 comments Feb 18, 21 Uncategorized 0 comments. W 17 174 17 Ne 10 10 NOTE The number protons and electrons is equal to the atomic number To find neutrons, subtract the number of protons from the atomic mass To find the atomic mass, add the number of protons and neutrons O Oxygen ATOMIC NUMBER SYMBOL NAME ATOMIC MASS.
Atoms are made of protons, neutrons, and electrons Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral A neutral atom has the same number of protons and electrons (charges cancel each other out) An ion has an unequal number of protons and electrons If the charge is positive, there are more protons than electrons. For most of the remaining elements, there are more neutrons than protons, because extra. User Select all that applyAtoms are made up of _____ elements protons neutrons compounds electrons Weegy Sigmund Freud was an Austrian neurologist who became known as the founding father of psychoanalysis Score 6385samnPoints 4674.
Number of Protons and Neutrons An Atom determines an Element The purest type of atom is called an element Atoms are composed of three kinds of smaller particles, called protons, neutrons and electrons Protons and neutrons are made up of even smaller particles called quarks. Rare Earth Elements Basic Information Atomic Structure Isotopes Related Links Citing This Page Basic Information Name Tungsten Symbol W Atomic Number 74 Atomic Mass 184 amu Melting Point °C (3615 K, °F) Boiling Point °C ( K, 1020 °F) Number of Protons/Electrons 74 Number of Neutrons 110. When you compare the masses of electrons, protons, and neutrons, what you find is that.
Since the atomic number indicates both the number of protons and the number of electrons present in an atom, this isotope contains 19 protons and 19 electrons The number of neutrons in the isotope can be calculated from its mass number, which is written as a superscript in a nuclear symbol Mass Number = # of Protons # of Neutrons. 24 The Discovery of the Electron Atoms, the smallest particles of an element that exhibit the properties of that element, consist of negatively charged electrons around a central nucl 25 The Structure of The Atom An atom consists of a small, positively charged nucleus surrounded by electrons The nucleus contains protons and neutrons. Titanium Element Protons Neutrons And Electrons Diagram The wiring diagram on the opposite hand is particularly beneficial to an outside electrician Sometimes wiring diagram may also refer to the architectural wiring program The simplest approach to read a home wiring diagram is to begin at the source, or the major power supply.
Q A boron atom has an atomic number of 5 and a mass number of 11 How many neutrons does an atom of Boron have?. Element Symbol Atomic Number Mass Number Protons Neutrons Electrons Carbon 6 14 O 8 10 Potassium 19 19 41 Tin Sn 50 68 Zinc 64 30 66 30 68 30 Cobal t Co 27 32 Boron 5 6 10 5 26 28 Ion Symbol Atomic Number Mass Number Protons Neutrons Electrons. Best Answers if you look at the periodic table, phosphorus has 15 electrons and protons, and 16 neutrons read more Source socraticorg 0 0 Quick Answer Sulfur is an element with 16 neutrons Sulfur also has 16 protons and 16 electrons This results in an atomic number of 16 and an atomic mass of 32 read more.
An atom of a certain element has 28 protons, 28 electrons, and a mass number of 59 How many neutrons are in this atom?. What element has nine protons and 10 electrons 18 Feb 21 what element has nine protons and 10 electrons Posted in Uncategorized Leave a comment. Best Answers if you look at the periodic table, phosphorus has 15 electrons and protons, and 16 neutrons read more Source socraticorg 0 0 Quick Answer Sulfur is an element with 16 neutrons Sulfur also has 16 protons and 16 electrons This results in an atomic number of 16 and an atomic mass of 32 read more.
Ditungsten carbide (W 2 C) Tungsten fluoride (WF 5) 4;. *I recommend watching this in x125 15 speed This video goes over how to know how many Protons, Electrons, and Neutrons are in any given element whether o.

Lecture 4 Atoms And Atomic Structure Protons Neutrons And Electrons The Atom Is The Smallest Building Block Of An Element The Atom Is Composed Of A Core Called The Nucleus Where Most Of The Atoms Mass Is Concentrated The Nucleus Is Composed Of Two

Slides Show

Organization Of The Periodic Table Of The Elements Ppt Download
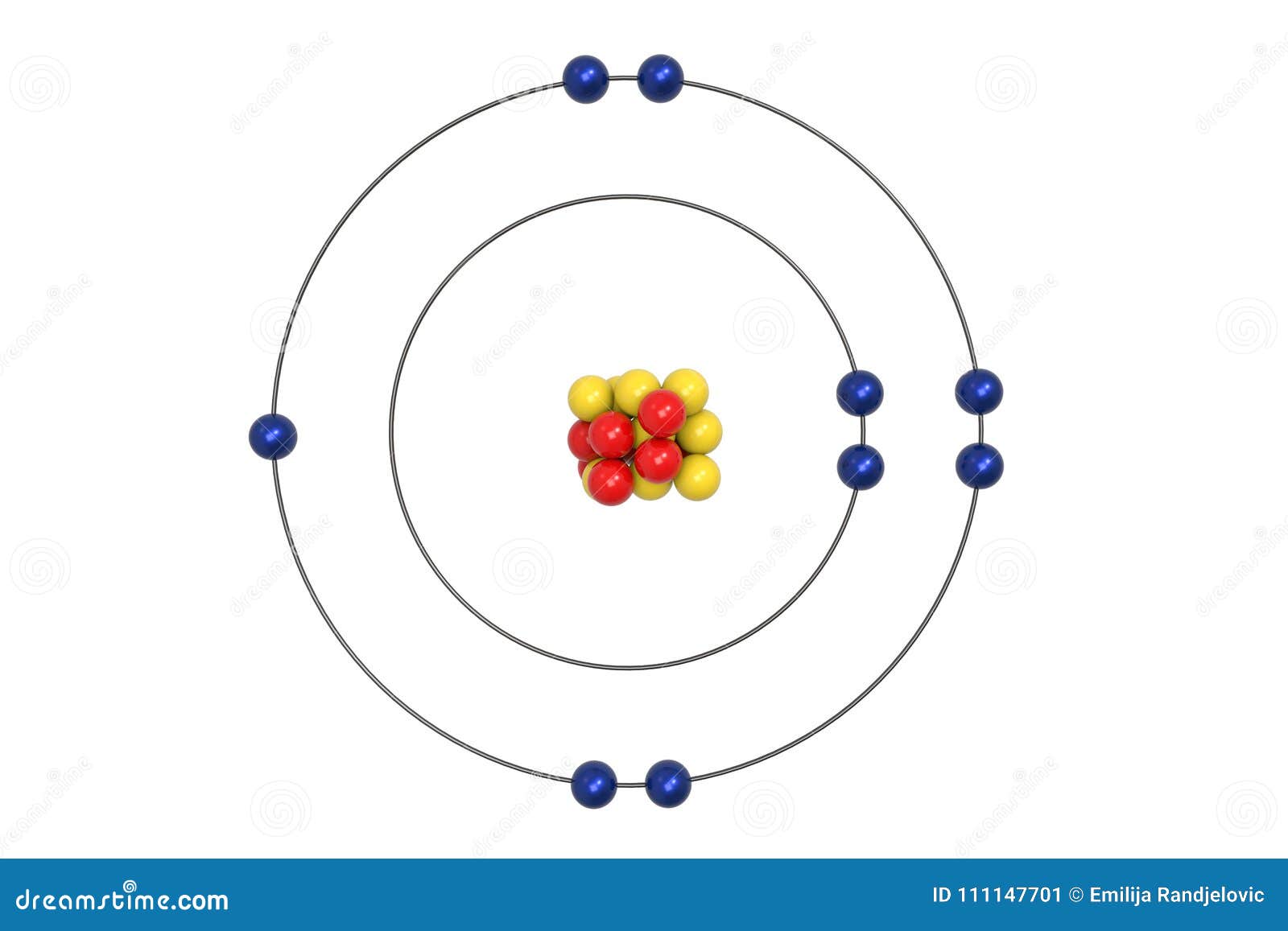
Fluorine Atom Bohr Model With Proton Neutron And Electron Stock Illustration Illustration Of Elements Core
How Many Protons Neutrons And Electrons Does Neon Have Quora

Protons Neutrons And Electrons Chapter 4 The Periodic Table Bonding Middle School Chemistry

What Is An Atom Live Science

Atomic Structure Atoms Molecules Quiz Quizizz
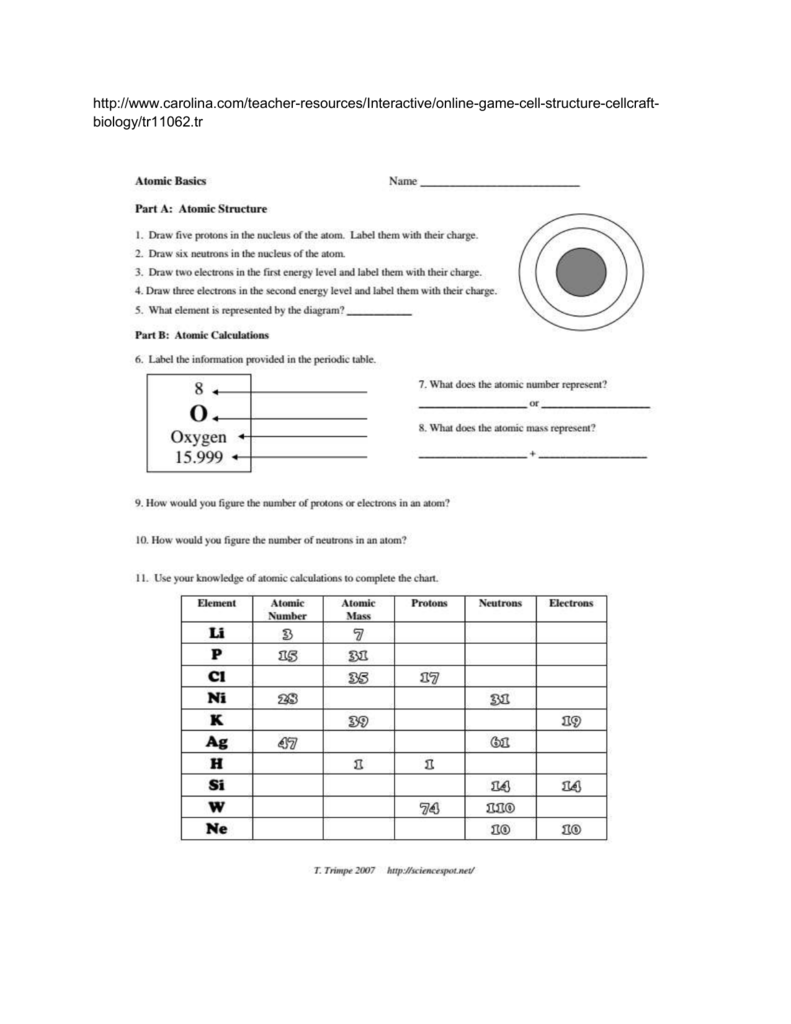
Atomic Basics Worksheet

Do Now Density Review Show Work 1 Find The Density Of An Object That Has A Mass Of 10g And A Volume Of 5 Cm Find The Density Of An Object

Electron Proton Twins Orderly Arranged In The Inside Of Bioatoms
Periodic Table Of Elements List With Protons Neutrons And Electrons
%252C_black_and_white.png?revision=2)
3 4 Atomic Mass And Atomic Number Chemistry Libretexts
Nearpod

Atomic Structure Ions And Bohr Diagrams

Atomic Structure
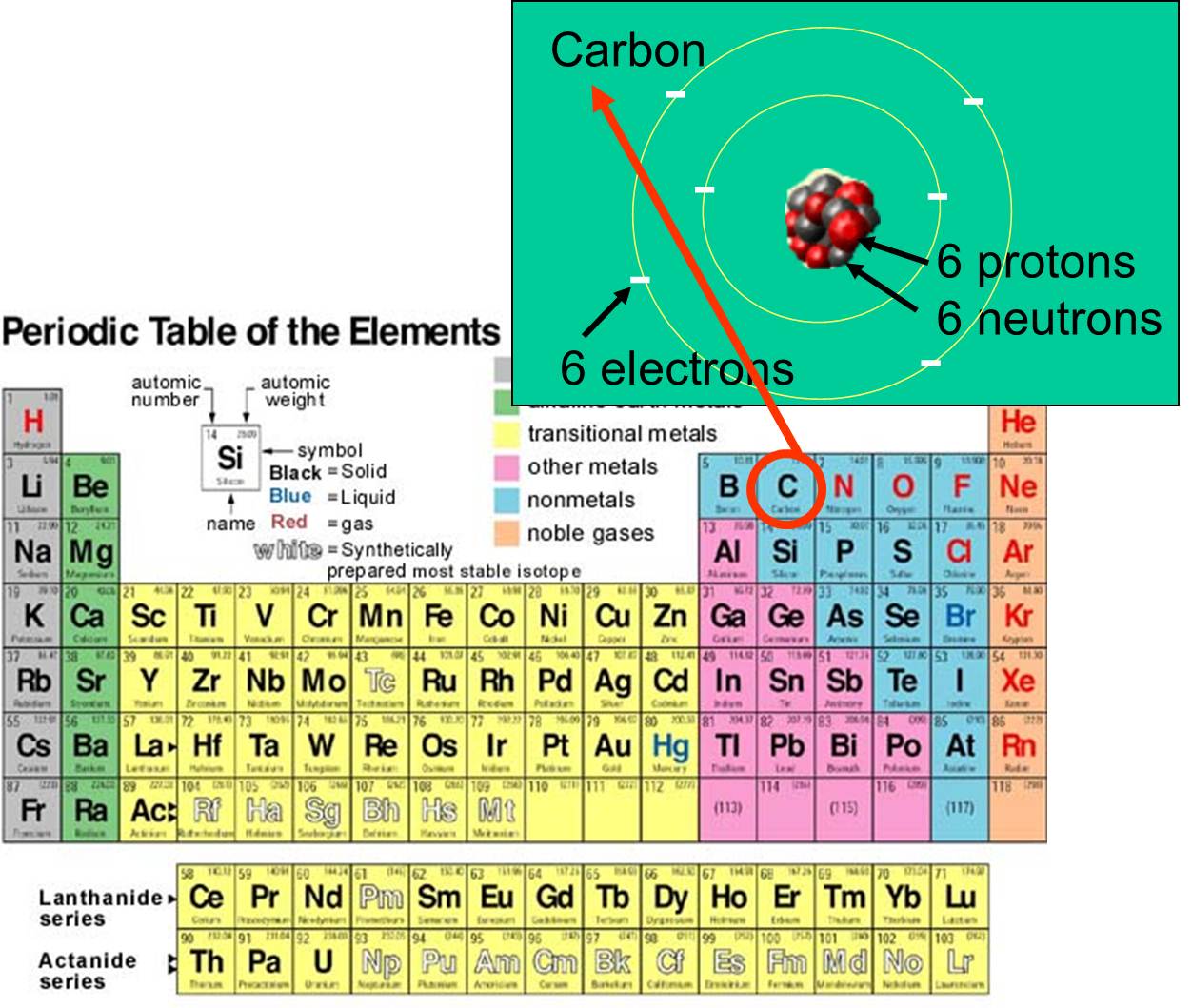
Chemical Elements Atoms

What Are The Parts Of An Atom

Atoms Isotopes Ions And Molecules Boundless Biology

Review With Answers
How Many Protons Neutrons And Electrons Are In Erbium Quora
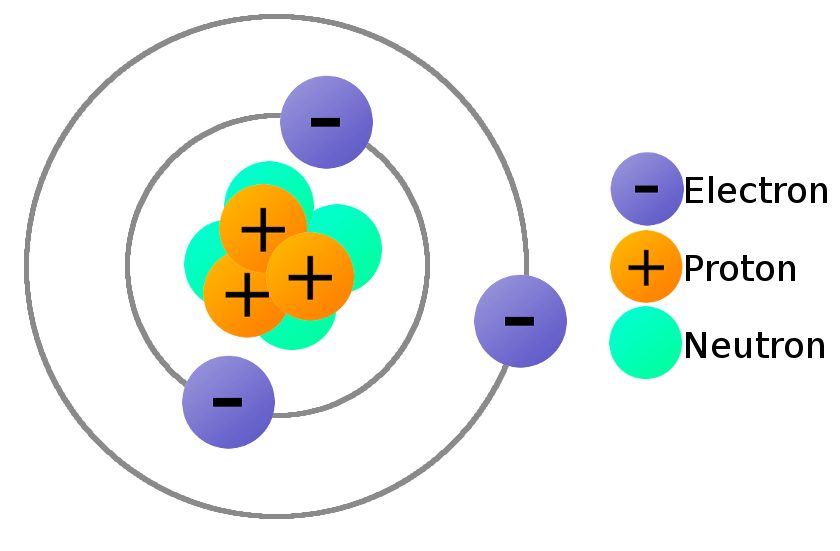
What Is Electricity Learn Sparkfun Com
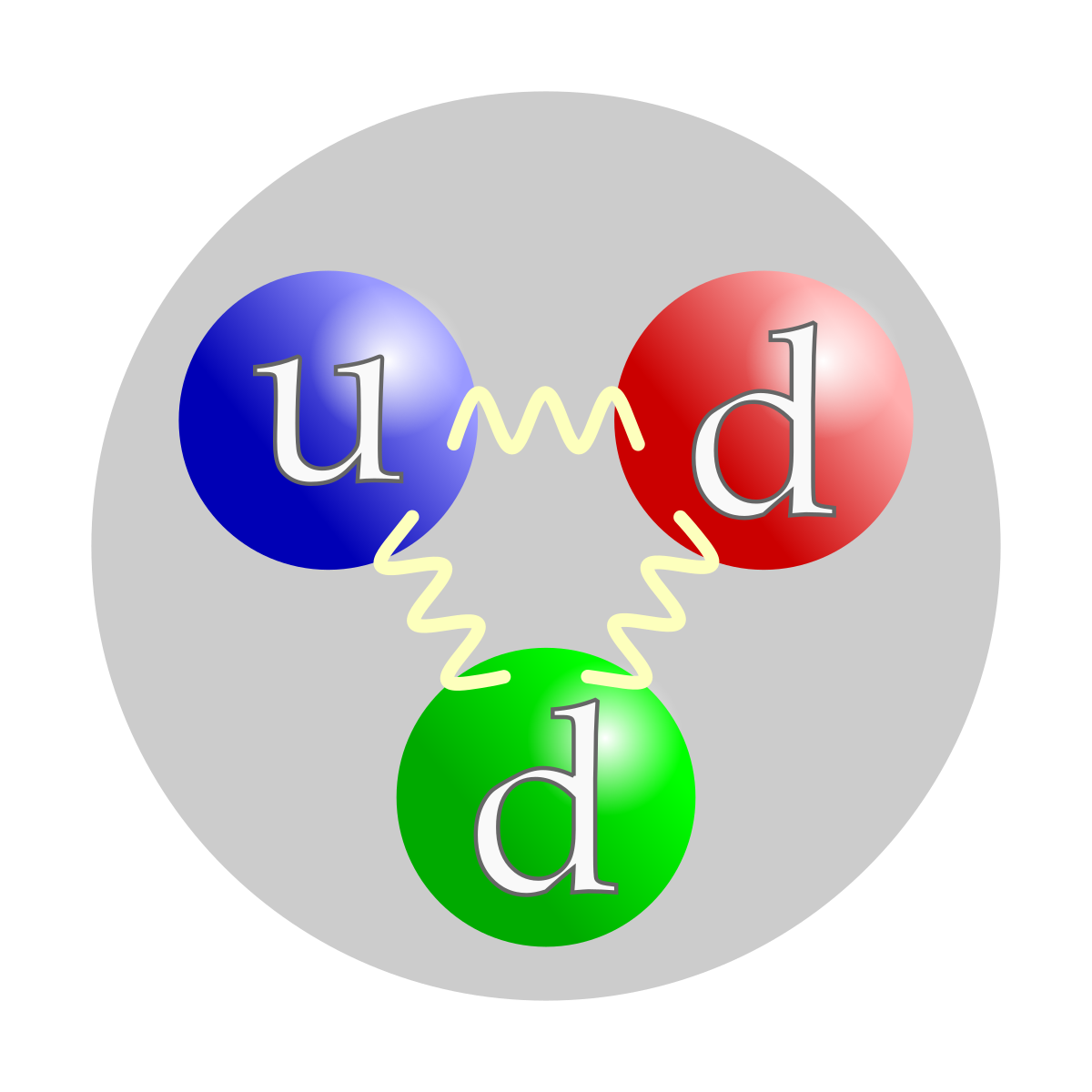
Neutron Wikipedia

Structure Of An Atom Learn Chemistry Class 9 Amrita Vidyalayam Elearning Network

Worked Example Identifying Isotopes And Ions Video Khan Academy
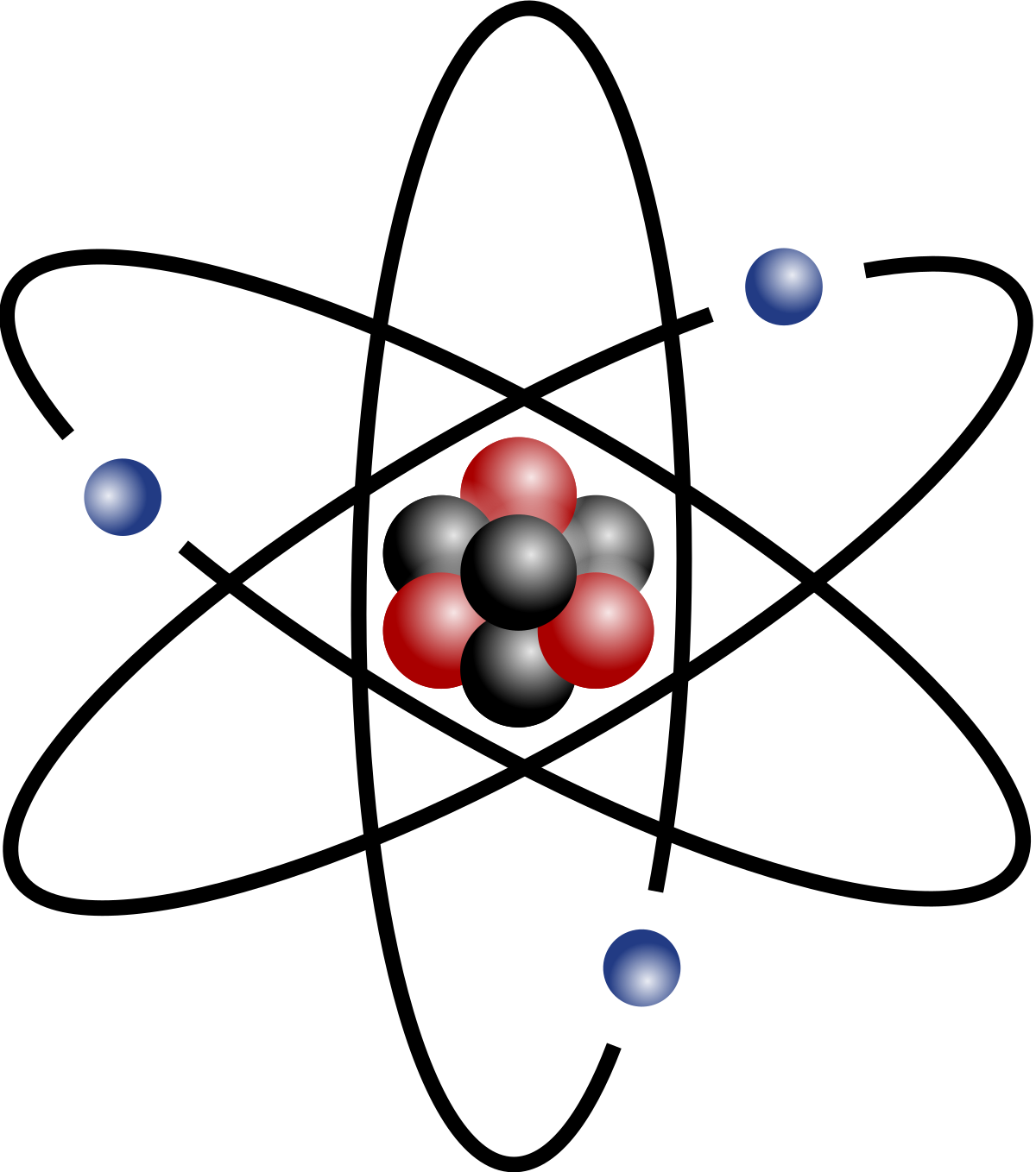
Atomic Mass Wikipedia
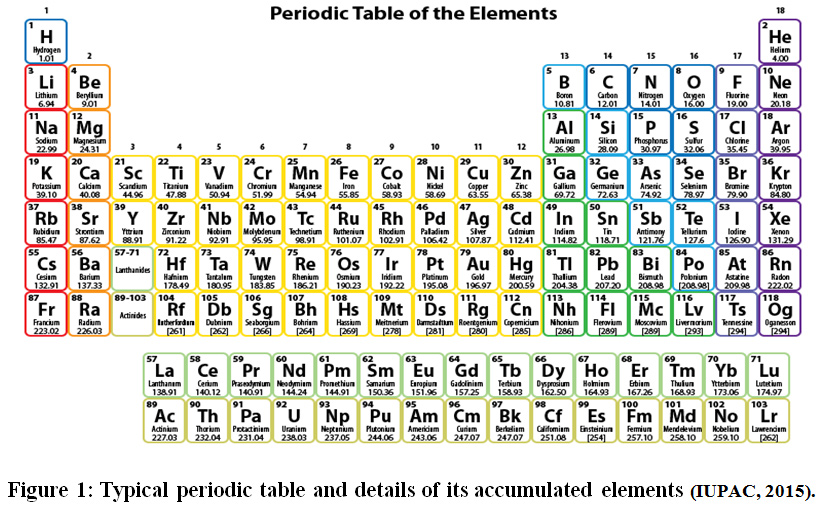
A Simplified Method For Estimating Atomic Number And Neutrons Numbers Of Elements Based On Period And Group Numbers In The Periodic Table Oriental Journal Of Chemistry

Mass Of Proton Definition Charge Discovery Properties With Videos

Average Atomic Mass Video Khan Academy
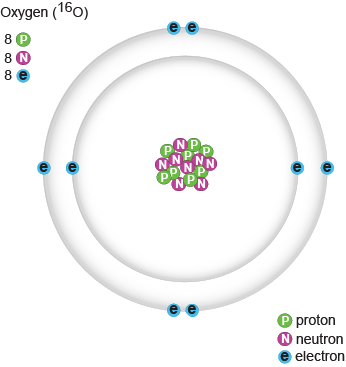
Biology Protons Neutrons And Electrons Shmoop Biology

Atomic Number Wikipedia
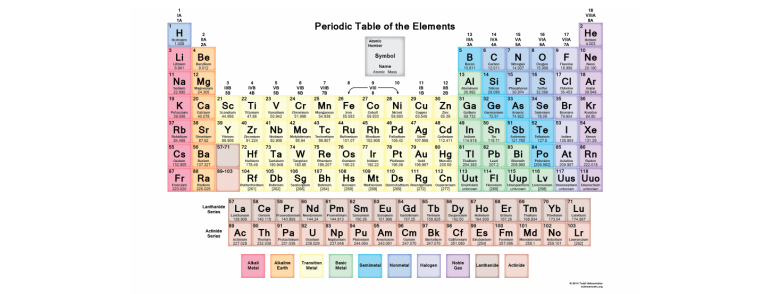
Periodic Table Year 9 Chemistry

Periodic Table Groups Periods Trends Patterns Comparison Properties Of Metals Non Metals Allotropes Summary Overview Gcse Chemistry Revison Notes Igcse O Level Ks4 Science

Isotopes And Atomic Mass Ck 12 Foundation

Atoms Protons Neutrons And Electrons A3 Anchor Poster Neutrons Protons Electrons
Ytterbium
Q Tbn And9gcstiuk49 Eq5yqqdh7btkte1ed Srd 9vl9zh06qg6c38kf1a1u Usqp Cau
The Science Of Electricity U S Energy Information Administration Eia

Basic Parts Of The Atom Protons Neutrons Electrons Nucleus Atom Atom Diagram Neutrons
Q Tbn And9gctrqlyzapewadl9ztnyvpkrz1qi4wxiagmjh1 Issxpidvd Svn Usqp Cau

Solved W S 40 Elements Name An Element Is A Pure Substa Chegg Com
Q Tbn And9gctyiilyu Wumkjajeu5opew2tpiss47pp9mklrjciu Usqp Cau

Ncert Exemplar Class 9 Science Solutions Chapter 4 Structure Of Atom Access Free Pdf

Atomicbasics

Atoms Isotopes Ions And Molecules Boundless Biology

Solved 1 The Atomie Number And Mass Number For An Atom W Chegg Com

General Organic And Biochemistry 8th Edition By Denniston Topping Woodrum And Caret Test Bank By Cndps8 Issuu
How To Find The Number Of Neutrons In An Isotope

Solved 1 The Atomic Number And Mass Number For An Atom W Chegg Com

Protons Neutrons And Electrons Practice Worksheet Youtube
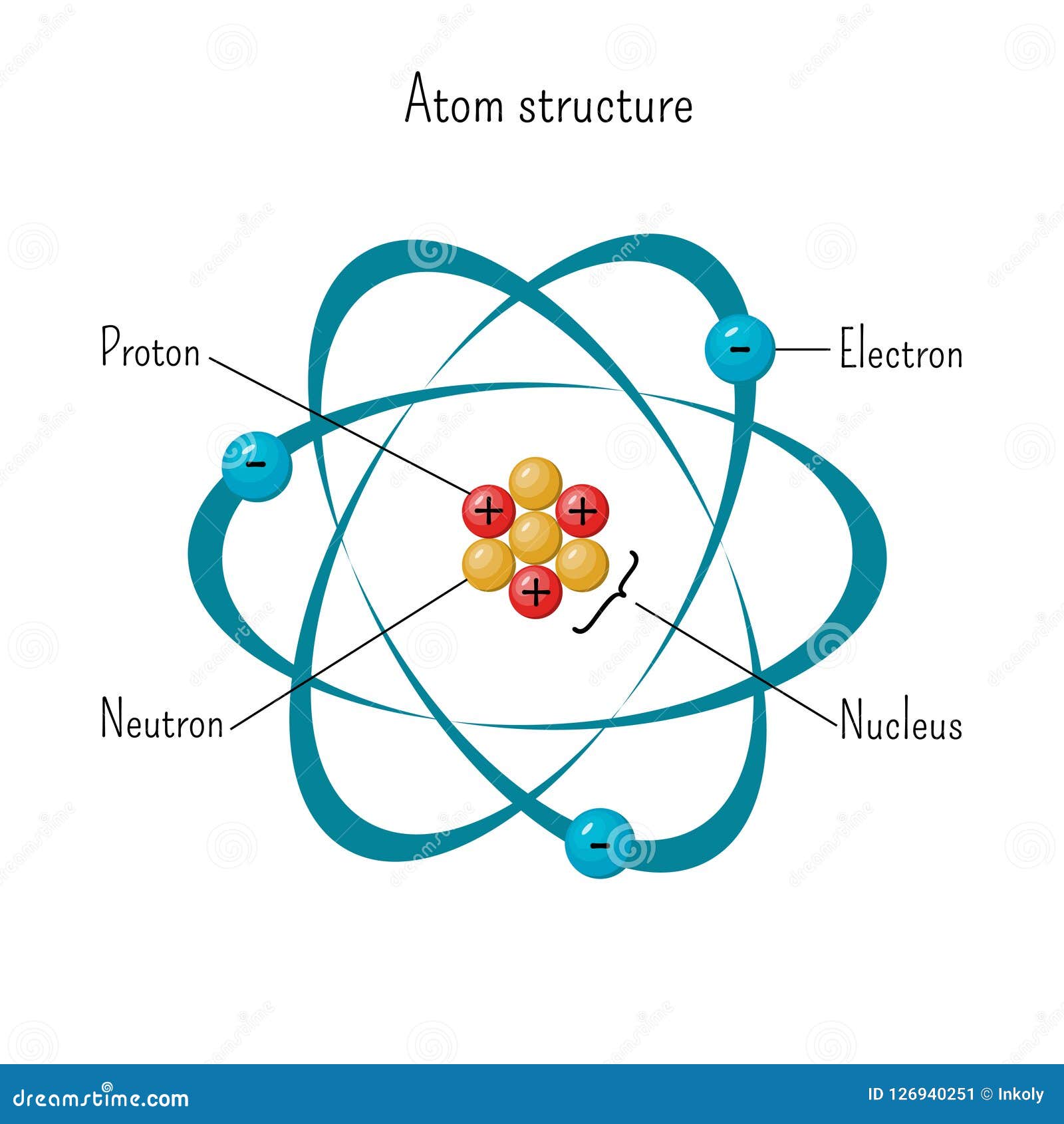
Simple Model Of Atom Structure With Electrons Orbiting Nucleus Of Three Protons And Neutrons Stock Vector Illustration Of Force Diagram
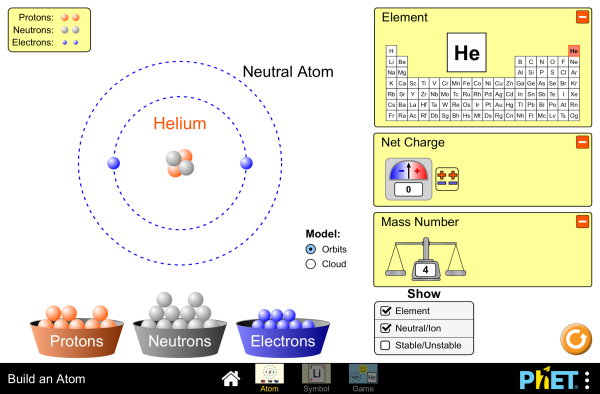
Build An Atom Atoms Atomic Structure Isotope Symbols Phet Interactive Simulations
What Isotope Has 18 Protons And 22 Neutrons Socratic
Q Tbn And9gcr2ajwuvf2szk7bcphrjvbjp 8ihvnnggsl5p7jd2dgrvy6ebec Usqp Cau

How To Find The Number Of Protons Electrons Neutrons For Nitrogen N Youtube
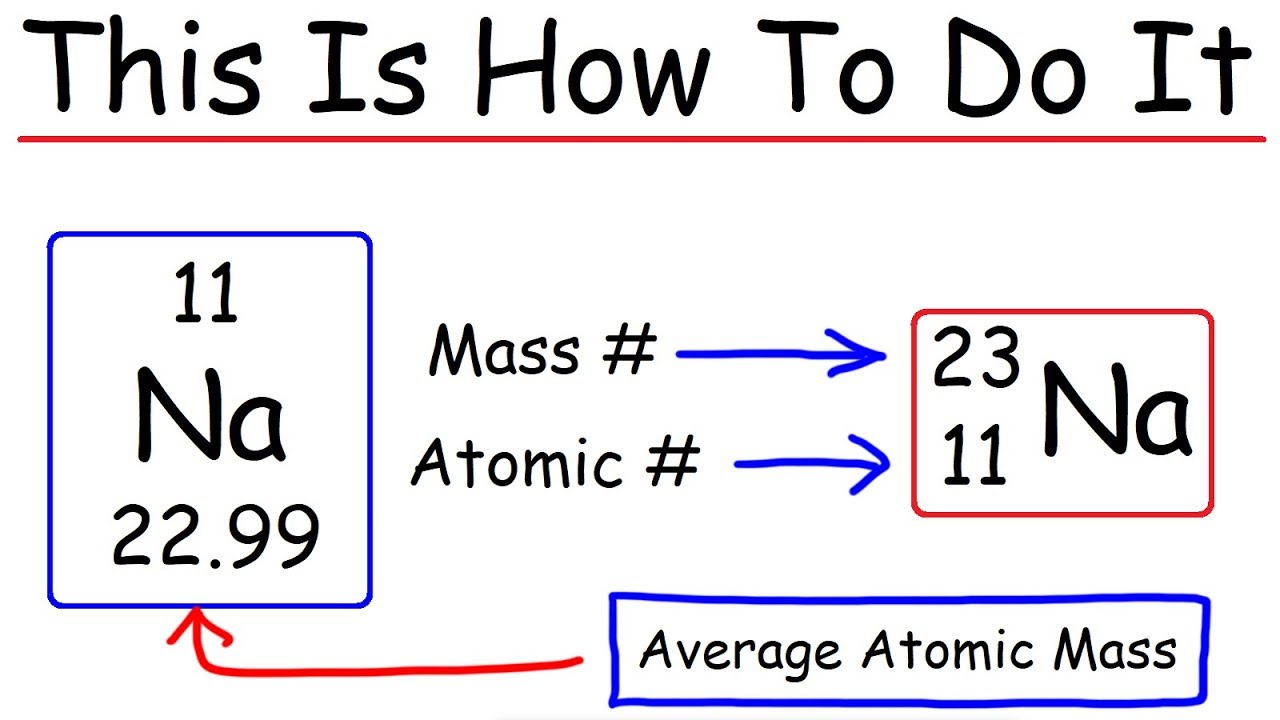
How To Calculate The Number Of Protons Neutrons And Electrons Chemistry Youtube
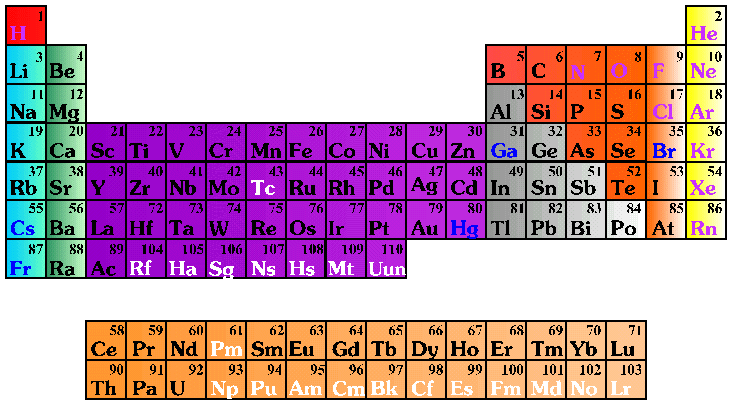
The Periodic Table By Energy Levels
Www Unf Edu Michael Lufaso Chem3610 Inorganic Chapter1 Pdf

Iron Has 26 Protons And Electrons And 30 Neutrons Atom Project Science Projects School Projects

Protons Neutrons And Electrons Chapter 4 The Periodic Table Bonding Middle School Chemistry
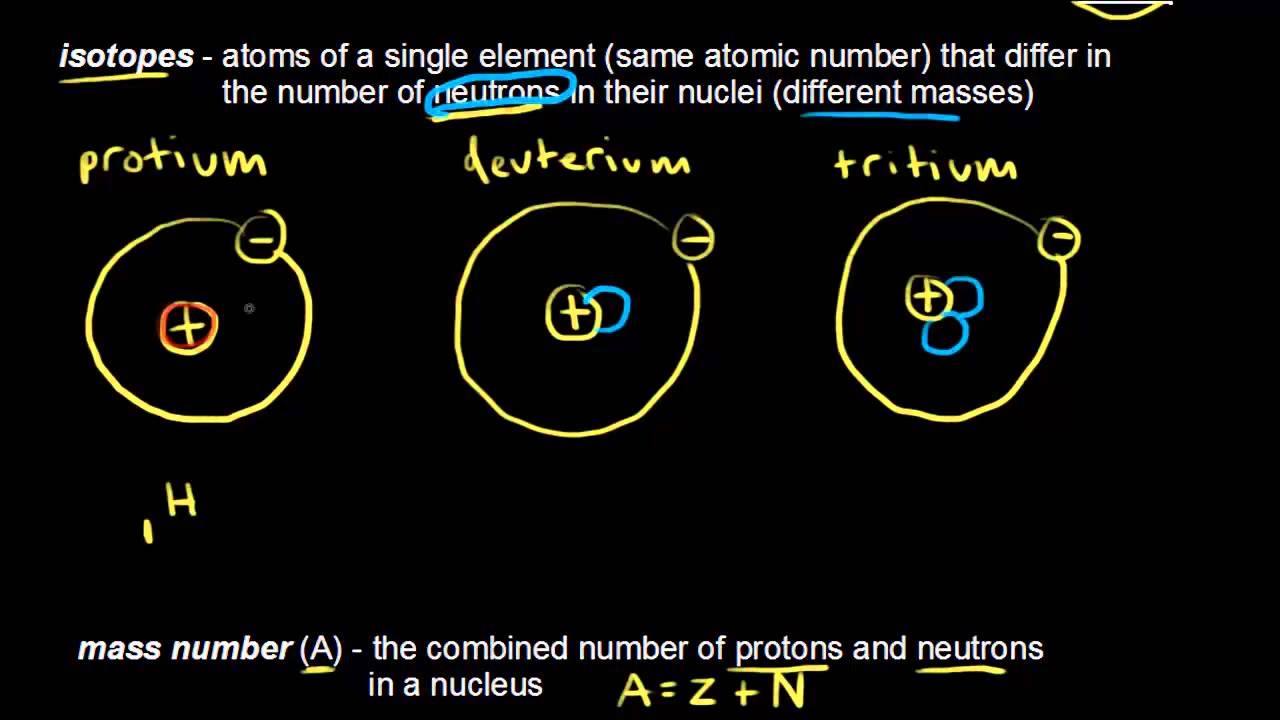
Atomic Number Mass Number And Isotopes Video Khan Academy
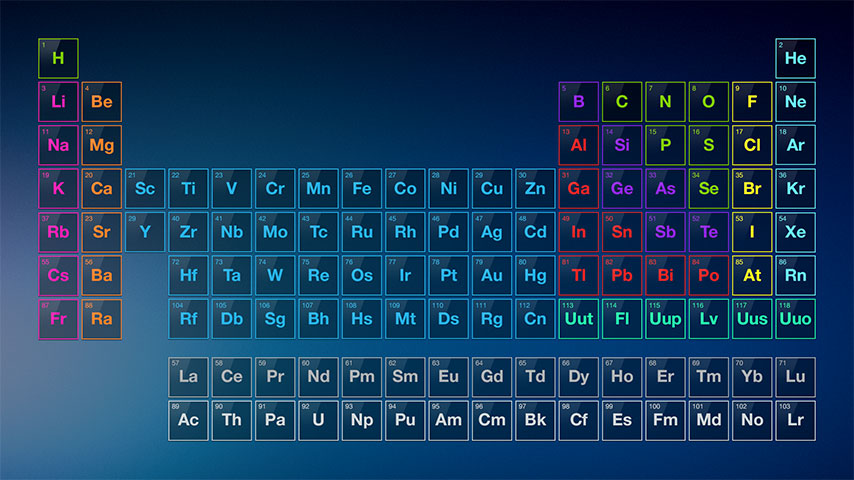
Goreact
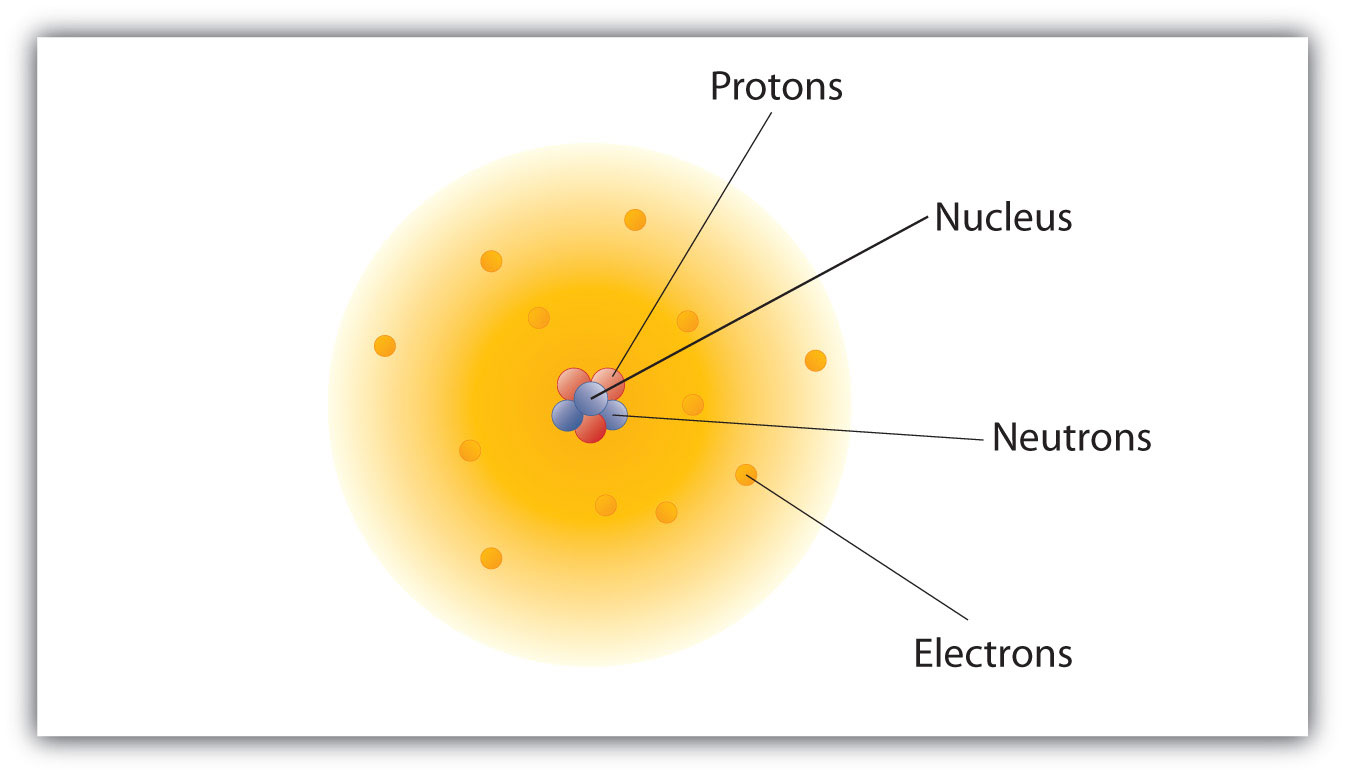
Atomic Theory
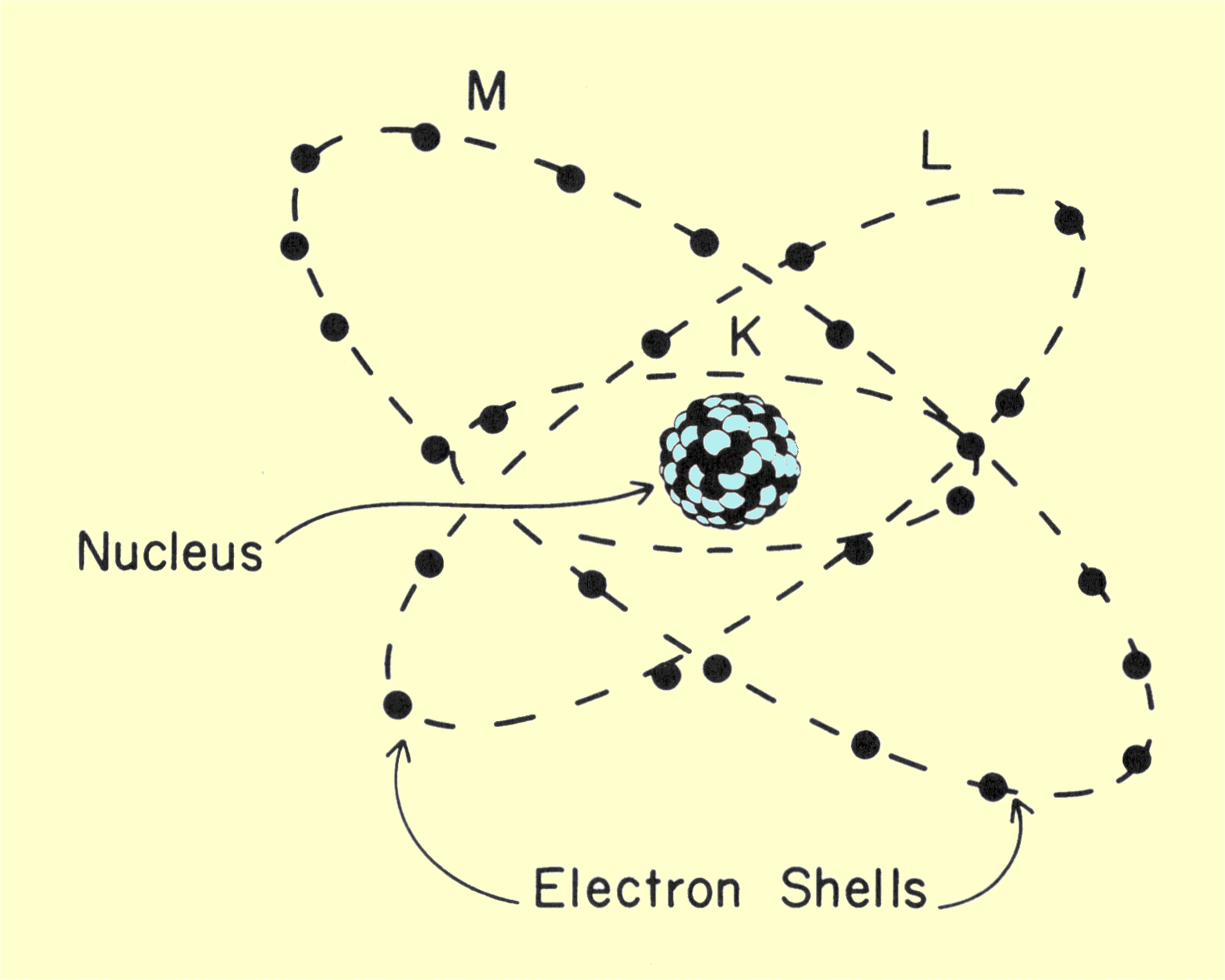
Characteristics And Structure Of Matter

Practice Determining The Number Of Protons Neutrons And Electrons With This Clever Chemistry Review Page Physical Science Teaching Chemistry Chemistry Review
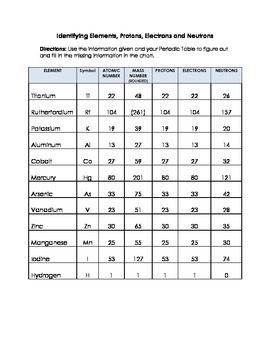
Periodic Table Of Elements List With Protons Neutrons And Electrons
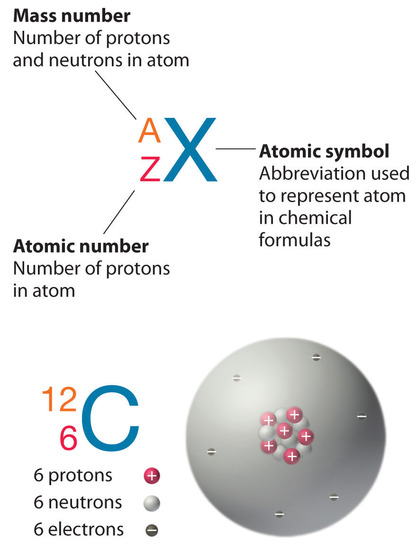
2 3 The Modern View Of Atomic Structure Chemistry Libretexts

10 Interesting Atom Facts

3e F Isotope Info Qs

Everything Worth Knowing About The Periodic Table Discover Magazine
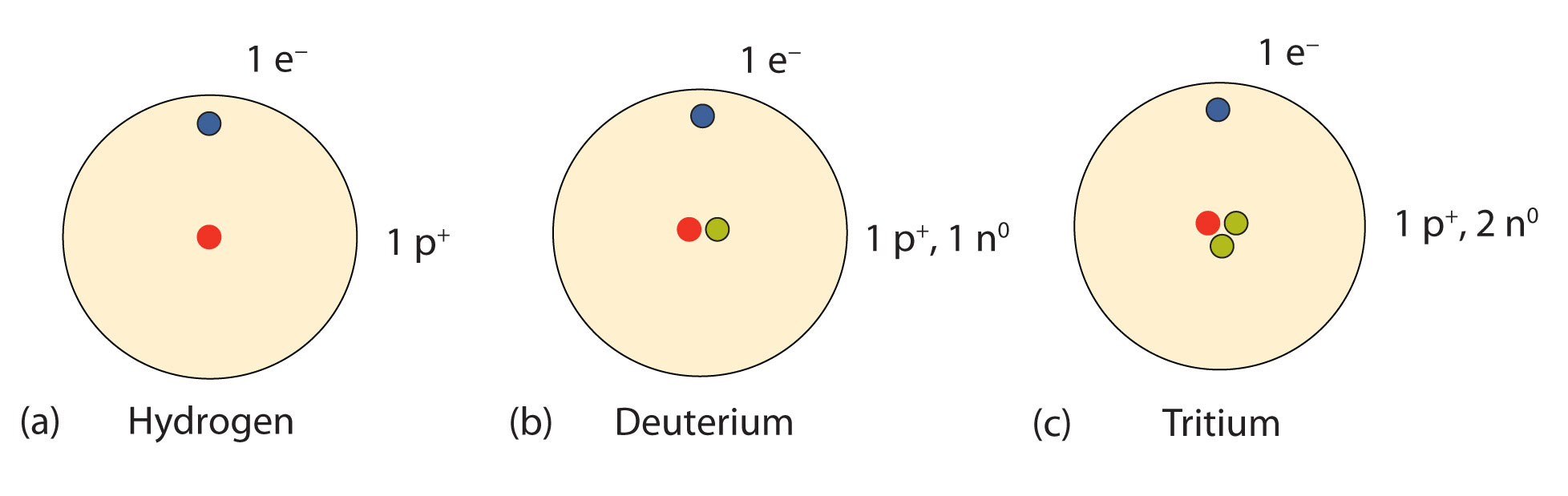
Nuclei Of Atoms

Determining Protons Neutrons And Electrons Of Atoms And Ions

Basic Chemistry Tutorial 2 Drawing Atoms Sciencemusicvideos

Solved Sample Problem 1 Write The Symbol Number Of Prot Chegg Com

Chapter 3 Elements And The Periodic Table Ppt Download
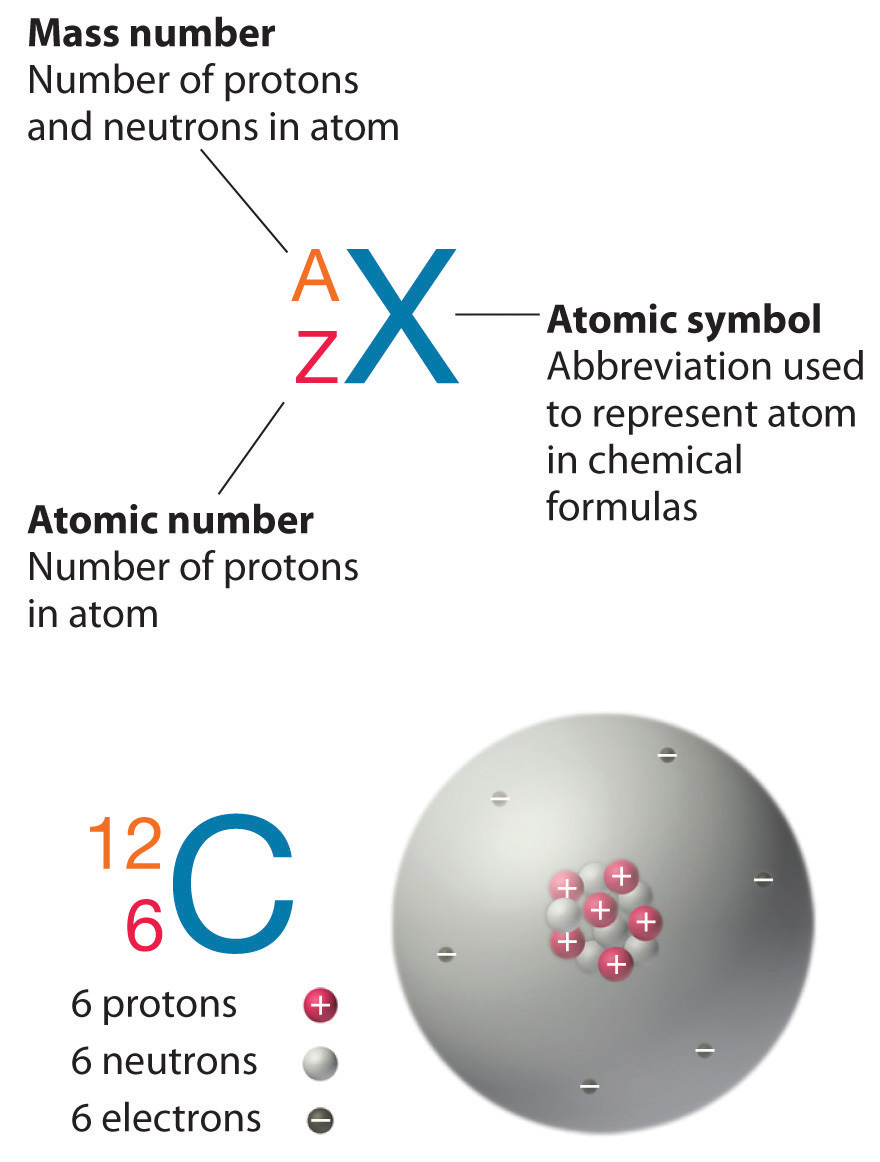
Chapter 1 6 Isotopes And Atomic Masses Chemistry Libretexts

Wait There Was An Exhibition Nik S Thoughts

3 Structures Of Atoms 3 1 1 4 Mcq Questions Igcse Chem Flashcards Quizlet
/GettyImages-523446050-5897be0a5f9b5874ee7c9fa6.jpg)
Number Of Protons Neutrons And Electrons In An Atom
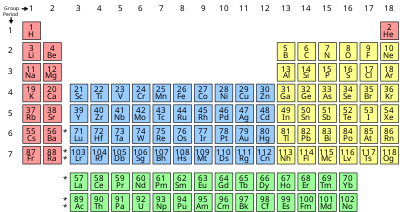
Chemical Element Wikipedia

Silver Sevens Hotel And Casino Las Silver Number Of Neutrons

How Many Protons And Neutrons Are In The Nucleus Of Isotope With Mass Of 68 926 Amu Lifeder English
What Is The Difference Between Protons Neutrons And Electrons Quora

Vector Tungsten Atom Stock Vector Royalty Free

Tungsten Atomic Structure Stock Image C018 3755 Science Photo Library
Www Topperlearning Com Answer I Want The Symbol Number Of Protons Number Of Neutrons Number Of Electrons Distribution Of Electrons And Valency Of The First 30 Elements 3y90wojj
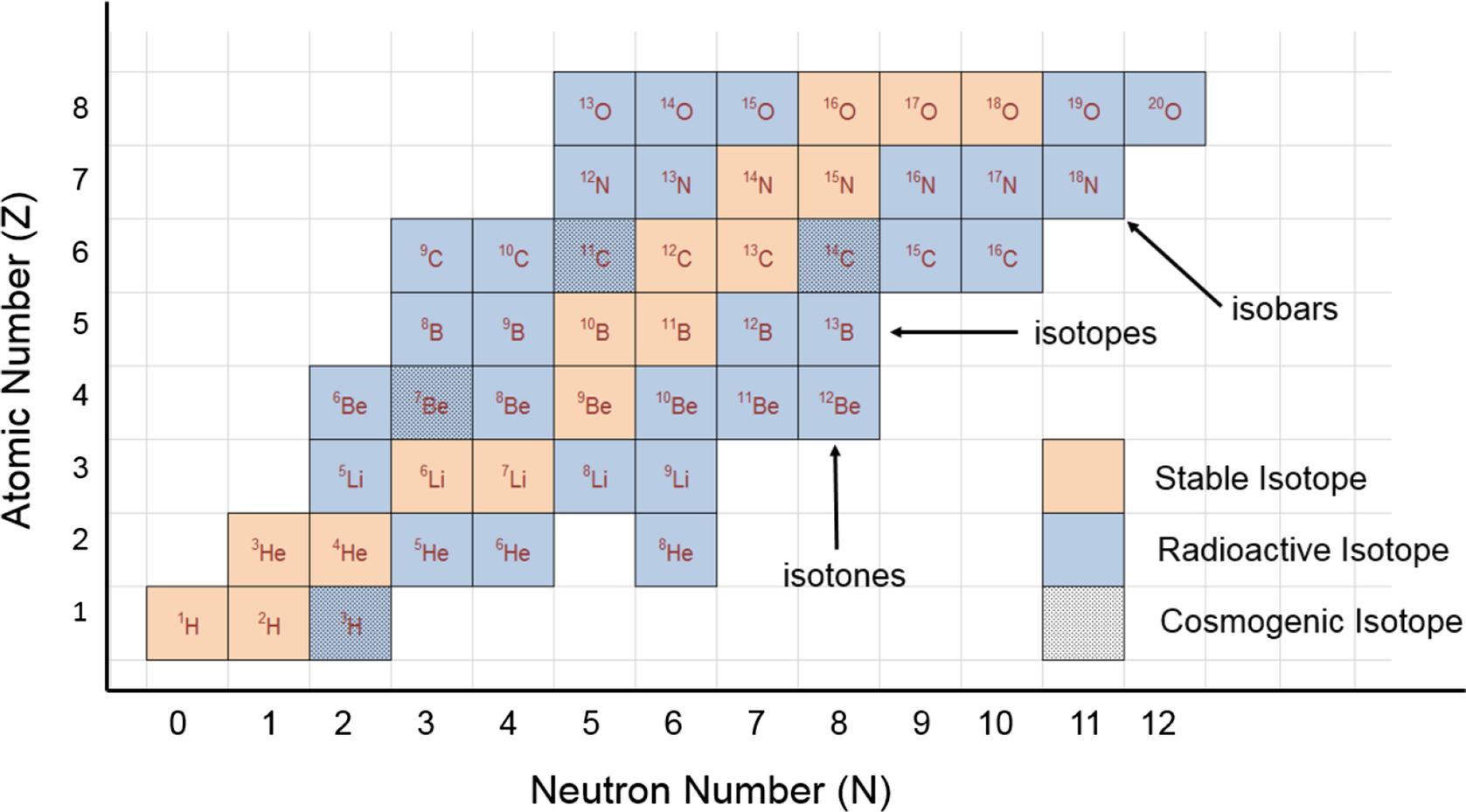
Atomic Number Mass Number And Isotopes Springerlink
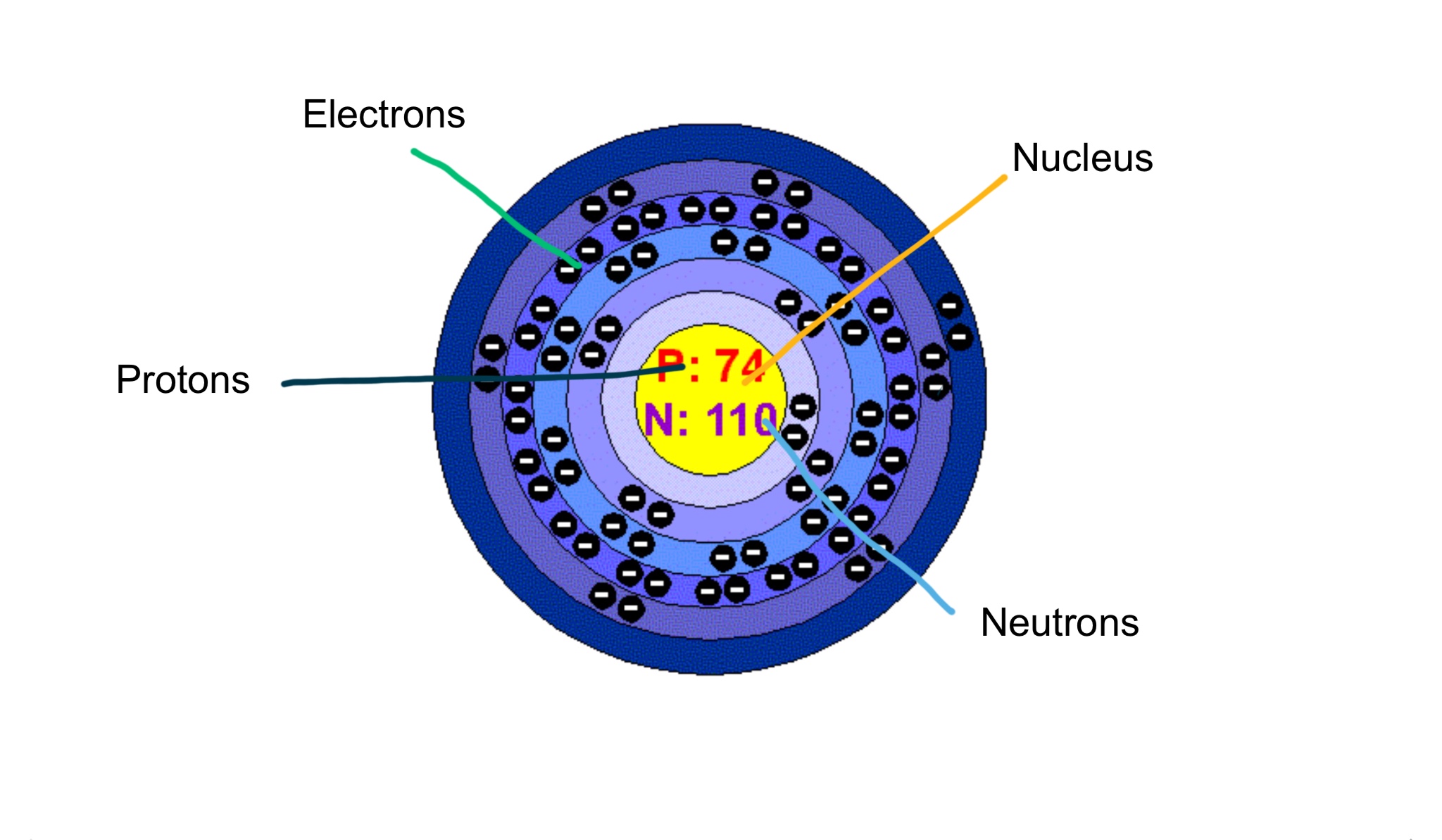
Tungsten Screen 6 On Flowvella Presentation Software For Mac Ipad And Iphone

Compare Nucleon Proton Neutron Number
Chemical Elements Com Tungsten W

Average Atomic Mass Video Khan Academy
Http Www Mvcsp Com Si Shared documents Che 2a Isotope packet Pdf

Basic Chemistry Tutorial 2 Drawing Atoms Sciencemusicvideos

Label Parts Of An Atom Learning In Hand With Tony Vincent Teaching Chemistry Chemistry Classroom Homeschool Science

Atomic Structure

An Atom Consists Of A Nucleus Nucleus Protons And Neutrons Electrons Electrons In Space About The Nucleus The Atom Nucleus Electron Cloud Ppt Download
Questions And Answers How Do I Find The Number Of Protons Electrons And Neutrons That Are In An Atom Of An Element

Test Bank Chapter 4 Middle School Chemistry



