15 7 N Protons Neutrons Electrons

Solved Part I Multiple Choice 2 Pts Each Toi Ly One A Chegg Com

Basic Atomic Structure Ppt Download
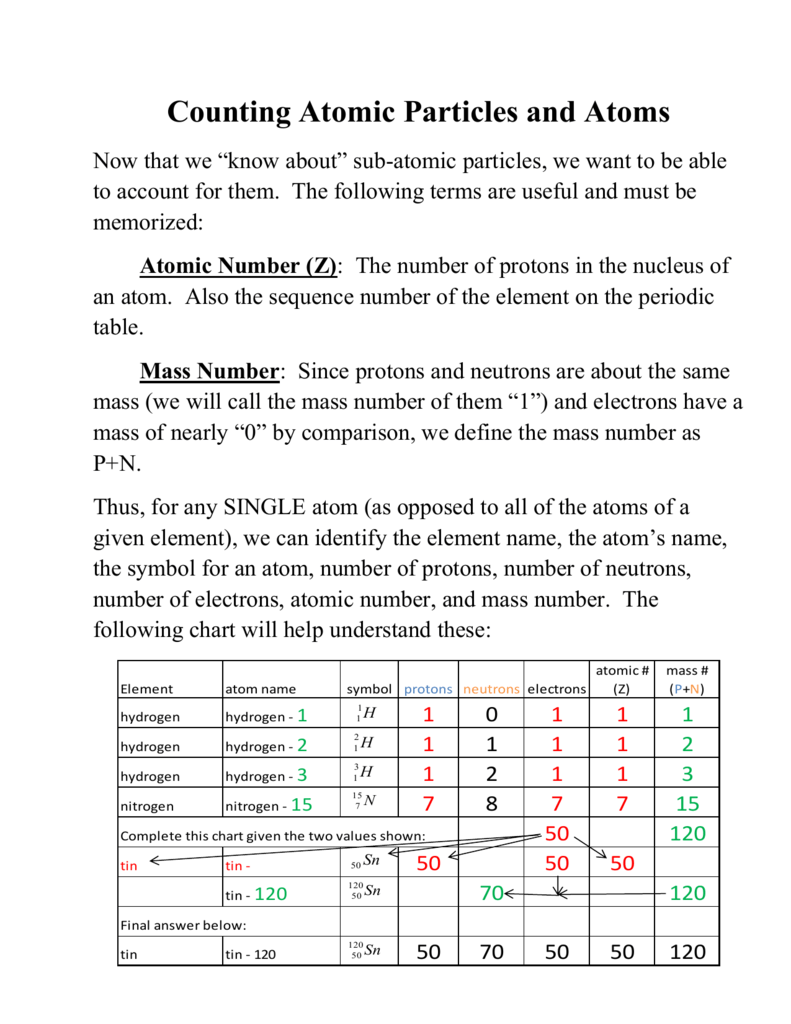
1 Carbon 12 Atom Has A Mass Of Exactly 12 Amu

Radioactivity And Radiolabeling The Bumbling Biochemist
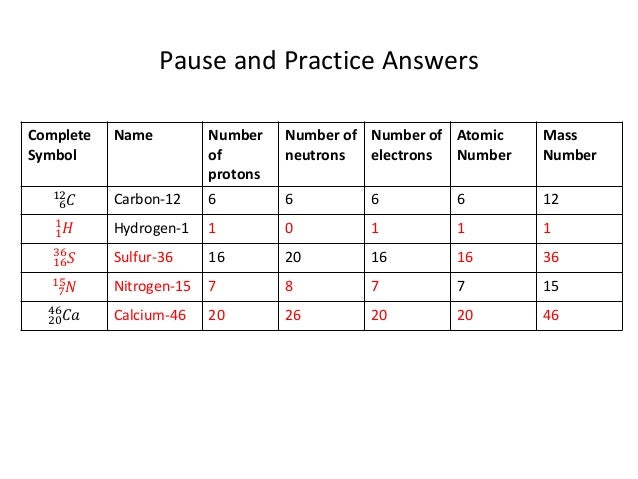
Unit 3 2 Structure Of The Atom
Www Saultschools Org Cms Lib Mi Centricity Domain 222 Atoms family answers Pdf
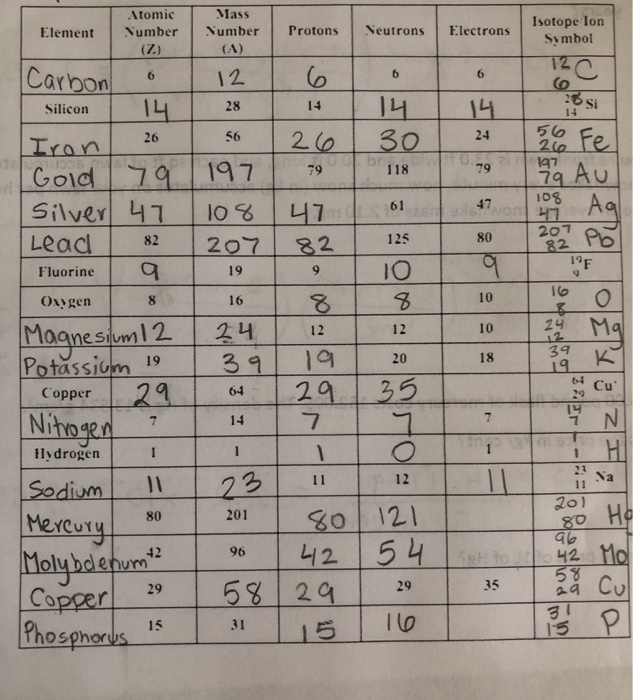
View Copy_of_Protons_Neutrons_Electrons_Practice from CHE 123 at Armijo High Name_Justin Wu_Date_Period_ Please read note on page 2!.

15 7 n protons neutrons electrons. Subsequently, one may also ask, how many protons neutrons and electrons does rubidium 87 contain?. Elements 1, with Protons, Electrons, Neutrons, and Atomic Mass 21 terms Isotopes. Because the atomic number of nitrogen is 7, it has 7 protons Then we take the mass number (protons neutrons) and subtract the number of protons to find the number of neutrons 15 7 = 8.
So, because this atom has 7 protons, its atomic number is 7 which is Nitrogen (N) The mention of names of specific companies or products does not imply any intention to infringe their proprietary rights It explains how we use cookies (and other locally stored data technologies), how thirdparty cookies are used on our Website, and how you can manage your cookie options A actual An atom of. 7 electrons, 7 protons, and 7 neutrons Nitrogen14 is actually an isotope of nitrogen, so right from the start, you can say that it is a neutral atom Isotopes are atoms that have the same number of protons but different numbers of neutrons in the nucleus You know that nitrogen14 has 7 protons in the nucleus because it is an isotope of nitrogen, which has an atomic number equal to 7. Name Nitrogen Symbol N Atomic Number 7 Atomic Mass atomic mass units Number of.
How to solve Give the number of protons, electrons, and neutrons in neutral atoms of the following isotope _{15} ^{31} \rm{P} By signing up,. A neutron also has about the same diameter as a proton, or \(17 \times 10^{15}\) meters As you might have already guessed from its name, the neutron is neutral In other words, it has no charge whatsoever and is therefore neither attracted to nor repelled from other objects Even though electrons, protons, and neutrons are all types of. For example, I tell you I have two isotopes of nitrogen, mass numbers of 14 and 15 How many neutrons in each?.
Question 15 SURVEY 30 seconds Report an issue Q The sum of protons and neutrons around the nucleus answer choices atomic number An oxygen atom has 8 protons, 8 neutrons, and 8 electrons What is the mass number?. Elements 1, with Protons, Electrons, Neutrons, and Atomic Mass 21 terms Isotopes. Protons, Neutrons, and Electrons Practice 1 How many n's are.
Nitrogen has seven protons Anything with seven protons within its nucleus (and only seven) is an atom of nitrogen The mass number (15) is the sum of the protons and neutrons 15 7 = 8 neutrons. Protons, Neutrons, and Electrons Practice 1 How many n's are. Protons Neutrons & Electrons of All Elements (List Images) February 26, 21 February 21, 21 by Admin Protons, neutrons and electrons of all elements are mentioned in the table below.
Because matter is electrically neutral, and positive and negative particles must be equal Electrons have negligible mass;. Give the number of protons, electrons, and neutrons in neutral atoms of each of the following isotopes a) 3 7Li b) 52 125Te c) 47 109Ag d) 7 15N. (d) 15,7 N 7 protons, 7 electrons, 8 neutrons (e) 31,15 P 15 protons, 15 electrons, 16 neutrons Explanation In the case of the isotopes in neutral atoms, the number of electrons and protons is equal to the atomic number, the number of neutrons is equal to the number of the isotope minus the atomic number (a) 7,3 Li The number of protons 3.
Which atom would have an atomic mass of 30?. A neutron also has about the same diameter as a proton, or \(17 \times 10^{15}\) meters As you might have already guessed from its name, the neutron is neutral In other words, it has no charge whatsoever and is therefore neither attracted to nor repelled from other objects Even though electrons, protons, and neutrons are all types of. For example, I tell you I have two isotopes of nitrogen, mass numbers of 14 and 15 How many neutrons in each?.
Elements 1, with Protons, Electrons, Neutrons, and Atomic Mass 21 terms Isotopes. (d) 15,7 N 7 protons, 7 electrons, 8 neutrons (e) 31,15 P 15 protons, 15 electrons, 16 neutrons Explanation In the case of the isotopes in neutral atoms, the number of electrons and protons is equal to the atomic number, the number of neutrons is equal to the number of the isotope minus the atomic number (a) 7,3 Li The number of protons 3. So we have seven protons and electrons and then 15 minus seven or eight neutrons, and we just do that with all of them So for 16 has 16 protons and electrons and 33 minus 16 or 17 neutrons.
Nickel60 is composed of 28 protons, 32 neutrons, and 28 electrons Nickel61 is composed of 28 protons, 33 neutrons, and 28 electrons Nickel61 is the only stable isotope of nickel with a nuclear spin (I = 3/2), which makes it useful for studies by EPR spectroscopy Nickel62 is composed of 28 protons, 34 neutrons, and 28 electrons. Protons Neutrons Electrons \(^{15}_{7}N\) 3\(^{42}_{}Ca\) 2 \(^{118}_{50}Sn\) 0. An atom is made up of protons, neutrons, and electrons The nucleus contains protons and neutrons and determines the atomic mass of an element On the other hand, the number of protons determines.
Worksheet Protons, Neutrons, and Electrons Complete the following chart Proton Neutron Electron Relative Charge Positive Neutral Negative Relative Mass 1 1 Location Nucleus Nucleus Electron charge Significance Identity, charge, mass Mass Charge Formula # p = mass # n # n = mass – # p # e = # p Fill in the following chart for the. The positively charged protons tend to repel each other, and the neutrons help to hold the nucleus together The number of protons is the atomic number, and the number of protons plus neutrons is the atomic mass For hydrogen, the atomic number is 1 because there is one proton and no neutrons For helium, it is 4 two protons and two neutrons. Answer choices 6 8 10 16 s Question 21 SURVEY 30 seconds Report an issue.
A neutron also has about the same diameter as a proton, or \(17 \times 10^{15}\) meters As you might have already guessed from its name, the neutron is neutral In other words, it has no charge whatsoever and is therefore neither attracted to nor repelled from other objects Even though electrons, protons, and neutrons are all types of. But yeah blame the The universe is made of protons neutrons electrons and morons vintage shirt Additionally,I will love this party not in charge the sad part is that you actually believe this which clearly explains all the Republican governors cancelling the reopening what a joke of a comment. An electron has very little mass by comparison, protons have a positive charge, electrons have a negative charge, and neutronsnone The nucleus of 87 Rb contains 37 protons(p) and 50 neutrons(n) What is rubidium found in?.
D) the same number of electrons e) the same number of shells 11) Chemists often represent the structure of atoms using p, n, and e to indicate the numbers of protons, neutrons, and electrons;. Give the number of protons, electrons, and neutrons in neutral atoms of each of the following isotopes a) 3 7Li b) 52 125Te c) 47 109Ag d) 7 15N. Nitrogen15 is composed of 7 protons, 8 neutrons, and 7 electrons Two sources of nitrogen15 are the positron emission of oxygen15 and the beta decay of carbon15 Nitrogen15 presents one of the lowest thermal neutron capture cross sections of all isotopes Nitrogen16 is composed of 7 protons, 9 neutrons, and 7 electrons.
15 7 N Atomic number is Z =7 Number of protons np =Z = 7 Number of electrons ne = np = 7 7 15 N Atomic number is Z = 7 Number of protons n p = Z = 7 Number of electrons n e = n p = 7 Mass. Anything with seven protons within its nucleus (and only seven) is an atom of nitrogen The mass number (15) is the sum of the protons and neutrons 15 7 = 8 neutrons The number of electrons. View Copy_of_Protons_Neutrons_Electrons_Practice from CHE 123 at Armijo High Name_Justin Wu_Date_Period_ Please read note on page 2!.
A) 10p, 10n, 10e C 15p, On, 15e b) 15p, 15n, 15e d Op, 15n, 15e 12) The number of electrons surrounding an. Nitrogen has 7 electrons when it is neutral It will need three more electrons to have a 3 charge 73 = 10 Element Protons Neutrons Electrons Charge Ar 15 Mo As 11 0 O 2 Write the symbol When you write the atomic symbol you do the following Title Atom Author. Subtracting the number of protons (7) from the element's mass number (14) makes it possible to determine the number of neutrons in the atom Many nitrogen atoms have seven neutrons (147 = 7) Some atoms of nitrogen, however, have eight electrons They are atoms of N15, an isotope of nitrogen.
Atoms are made of protons, neutrons, and electrons Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral A neutral atom has the same number of protons and electrons (charges cancel each other out) An ion has an unequal number of protons and electrons. N14 > 14 minus 7 = 7 and N15 > 15 7 = 8 Notice that 7 is the atomic number of nitrogen and I used it in both subtractions nmber of electrons you have to know the charge on the atom. Calculate numbers of protons, neutrons, and electrons by using mathematical expressions (13) p = 11 n = 23 11 = 12 e = 11 0 = 11 4 Alternatively, you can also calculate the atomic number, atomic mass, and charge 5 Choose your element Let’s assume that it is the sulfide anion 6 Find the numbers of protons, neutrons, and electrons.
Protons Neutrons & Electrons of All Elements (List Images) February 26, 21 February 21, 21 by Admin Protons, neutrons and electrons of all elements are mentioned in the table below. This website uses cookies to improve your experience while you navigate through the website It doesnt matter how many elecrons or neutrons it has For example, removing an electron from an atom of krypton forms a krypton ion, which is usually written as Kr This means that the unit as a whole is electrically neutral its positive and negative vibes cancel out Try these on your own and check. 7 electrons, 7 protons, and 7 neutrons Nitrogen14 is actually an isotope of nitrogen, so right from the start, you can say that it is a neutral atom Isotopes are atoms that have the same number of protons but different numbers of neutrons in the nucleus You know that nitrogen14 has 7 protons in the nucleus because it is an isotope of nitrogen, which has an atomic number equal to 7.
Number of protons equals the number of neutrons ratio of the number of neutrons to the number of electrons is 11 ratio of the number of neutrons to the number of protons is 21. The mass number depends on the number of protons contained within the nucleus, PLUS the number of neutrons Given that we have 15N, there must be 8 neutrons contained within the nucleus Capisce?. In the periodic table, the elements are listed in order of increasing atomic number Z The protons exist in the nuclei of typical atoms, along with their neutral counterparts, the neutrons Therefore, the number of electrons in neutral atom of Nitrogen is 7 Main purpose of this project is to help the public to learn some interesting and important information about chemical elements and many.
N14 > 14 minus 7 = 7 and N15 > 15 7 = 8 Notice that 7 is the atomic number of nitrogen and I used it in both subtractions nmber of electrons you have to know the charge on the atom. 7 protons 7 neutrons 10 electrons element February 19, 21 by Comments are off. The number of protons is unique for each element, so Nitrogen will always have 7 protons There is usually the same number of electrons as there are protons, so that would be 7 too (unless there is.
Neutral Nitrogen has an atomic number of 7 (from periodic table) thus it has 7 protons and 7 electrons (equal p and e) Since it is an ion with charge 3, it means that it gained 3 electrons therefore, N^3 has 7 protons and 10 electrons. Protons Neutrons & Electrons of All Elements (List Images) February 26, 21 February 21, 21 by Admin Protons, neutrons and electrons of all elements are mentioned in the table below.

Solved Do Both Members Of The Following Pairs Have The Sa Chegg Com
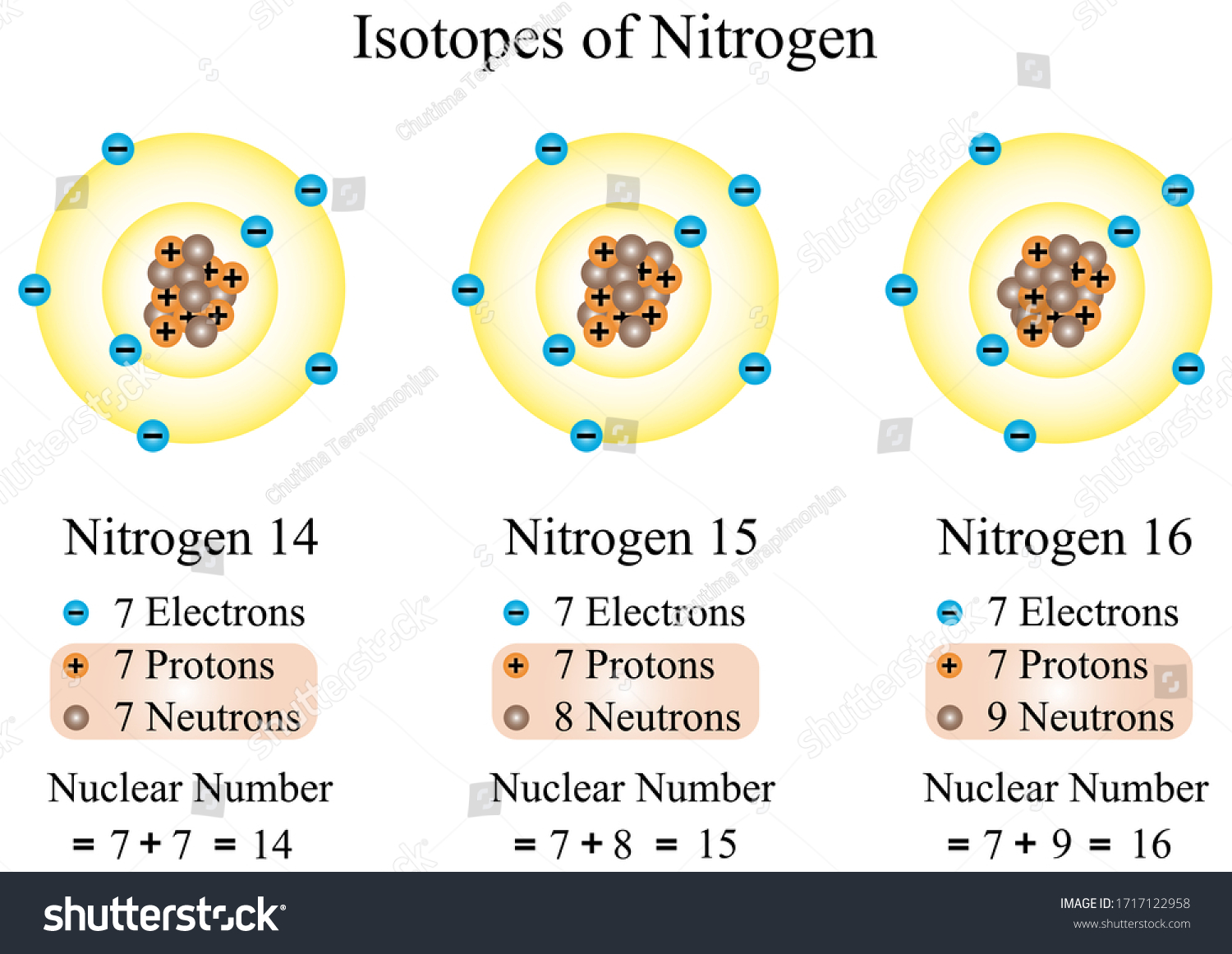
Illustration Chemical Isotopes Nitrogen All Atoms Stock Vector Royalty Free

Isotopic Notation Chemistry 11 Definition Of An Isotope Isotopes Are Atoms With The Same Number Of Protons But Different Number Of Neutrons Isotopes Ppt Download

The Ion N3 Has Protons And Clutch Prep

O Level Chemistry 05 15 13

Atom Wikipedia

Solved I Choose The Correct Answer Showing Your Detaile Chegg Com

Atomic Number Number Of Protons Always An Integer Ppt Download

How To Find The Number Of Protons Electrons Neutrons For Nitrogen N Youtube
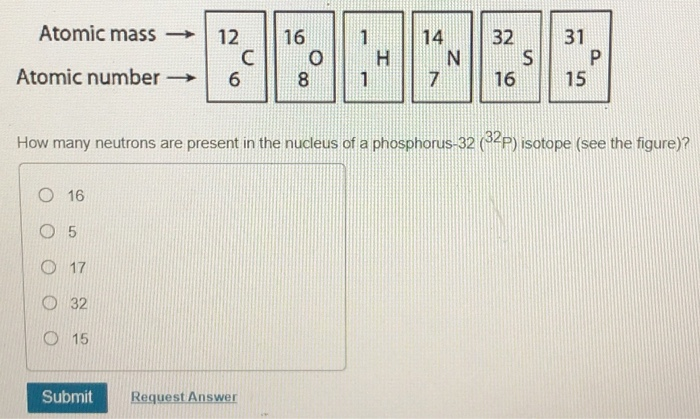
Solved A B D E Which Drawing In The Figure Depicts Th Chegg Com

The Number Of Protons Neutrons And Electrons In Particles A To E

Solved Complete The Blanks Below For Each Element The Nu Chegg Com

Solved 130 15 The Atomic Number Of An Atom Of 130xe Is Chegg Com

Solved How Many Protons Neutrons And Electrons Are In A Chegg Com

How Many Neutrons And Protons Are There In The Following Nuclei 6 13c 8 16o 12 24mg 26 56fe 38 sr

Radioactivity And Radiolabeling The Bumbling Biochemist
Www Scasd Org Cms Lib5 Pa Centricity Domain 1441 Chm1r 4 Pdf
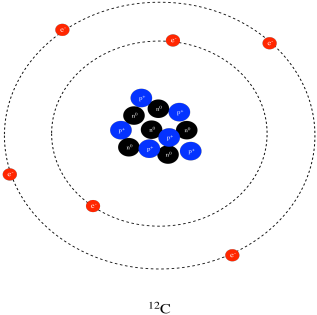
Structure Reactivity Atoms Protons Neutrons Electrons

An Atom Has Electronic Configuration 2 8 7 A What Is The Atomic Number Of This Element B Youtube
Q Tbn And9gcqtxt5uvohwgqexjto0f3um5 Wubockh3z7osukeywcfc4ry8 Usqp Cau

Atomic Structure And Symbolism Chemistry For Majors

Notes 4 3 Atomic Number Mass Number Isotopes And Atomic Mass Ppt Download

Solution How Many Protons Neutrons And E Clutch Prep
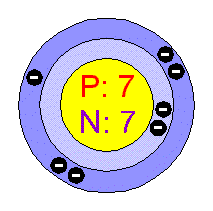
Chemical Elements Com Nitrogen N

Write The Complete Symbol For The Atom With The Given Atomic Number Z And Atomic Mass A I Z 17 A 35 Ii Z 92 A 233 Iii Z 4 A 9
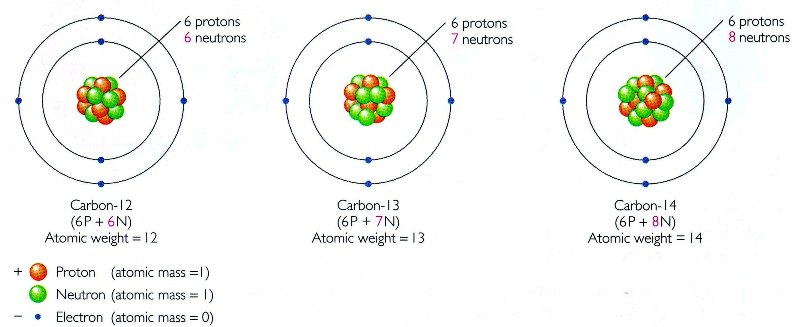
O Level Chemistry 05 15 13
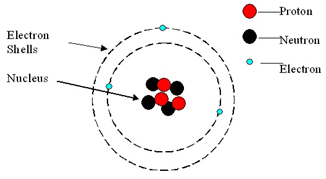
1 15 Know The Structure Of An Atom In Terms Of The Positions Relative Masses And Relative Charges Of Sub Atomic Particles Tutormyself Chemistry
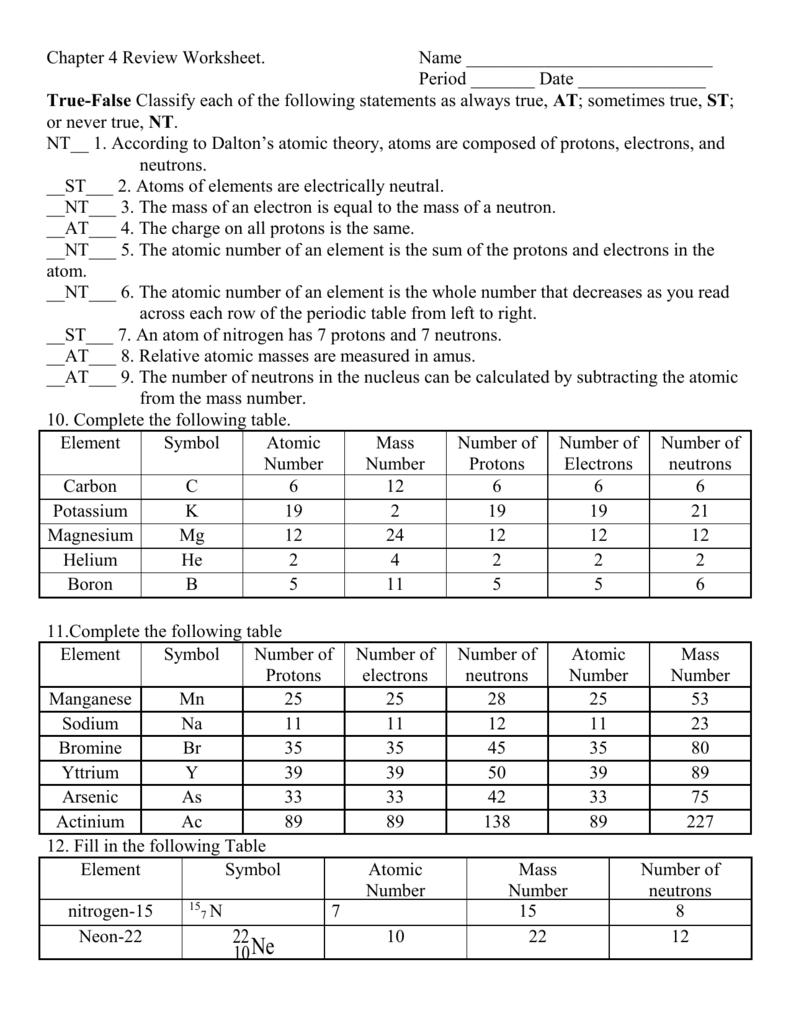
Chapter 4 Review Worksheet Name
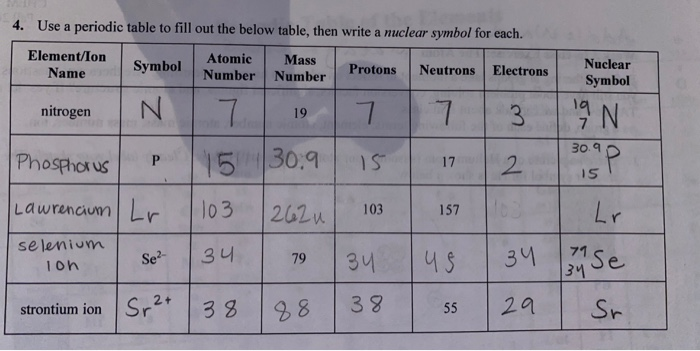
Solved 4 Use A Periodic Table To Fill Out The Below Tabl Chegg Com

File Electron Shell 007 Nitrogen Svg Wikimedia Commons Electron Configuration Atom Diagram Electrons

How Are Electrons Distributed In Different Orbits Electronic Configuration
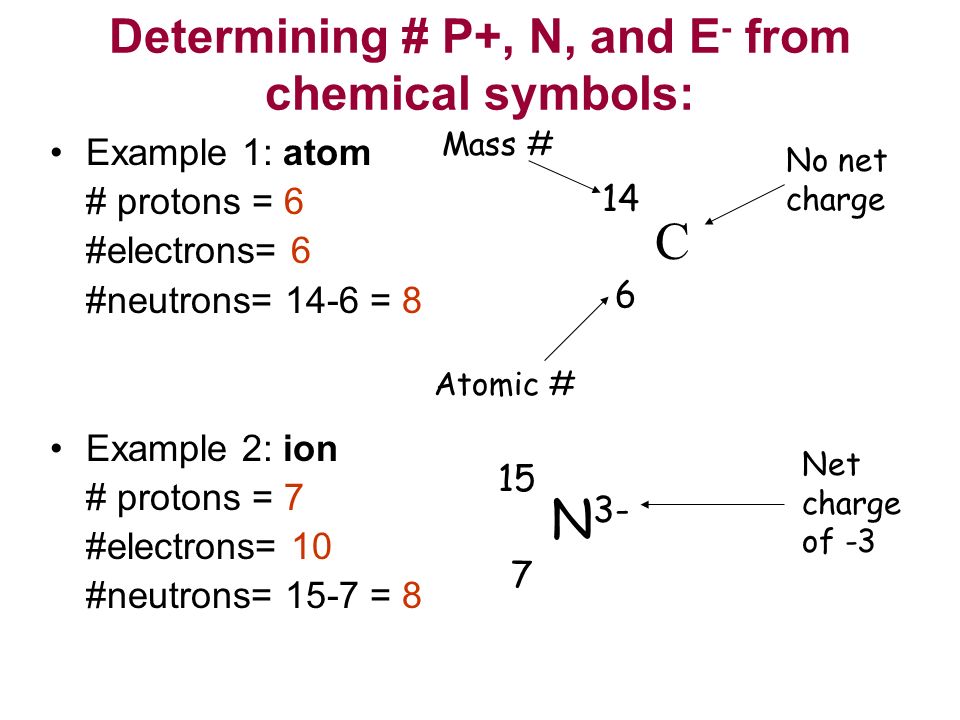
Warm Up 2 10 What Do You Know About Atoms And The Periodic Table What Do You Think An Atom Looks Like Ppt Download
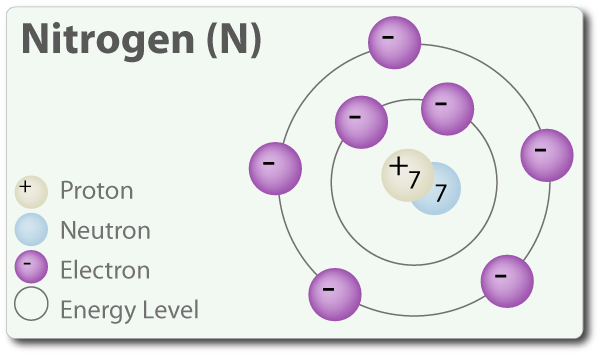
Question 4b2b1 Socratic
Q Tbn And9gcrynkusoqel S461uigmf0todl81sknigwjhtrhhhmo2pdvorri Usqp Cau

Ncert Exemplar Class 9 Science Solutions Chapter 4 Structure Of Atom Access Free Pdf

Ncert Exemplar Class 9 Science Solutions Chapter 4 Structure Of Atom Access Free Pdf

Hydrogen Elements Number Of Protons 1 Neutrons 0 Electrons 1 Ppt Video Online Download

Ncert Exemplar Class 9 Science Solutions Chapter 4 Structure Of Atom Access Free Pdf

10 15 Review Atomic Structure
Www Manhassetschools Org Cms Lib Ny Centricity Domain 796 Atoms practice qs answer key Pdf
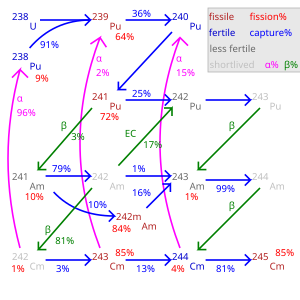
Neutron Wikipedia

Isotopes And Atomic Mass Ck 12 Foundation
Atomic Number Mass Number And Isotopes Video Khan Academy

Lecture 4 Atoms And Atomic Structure Protons Neutrons And Electrons The Atom Is The Smallest Building Block Of An Element The Atom Is Composed Of A Core Called The Nucleus Where Most Of The Atoms Mass Is Concentrated The Nucleus Is Composed Of Two

Protons Neutrons Electrons Isotopes Average Mass Number Atomic Structure Atoms Vs Ions Youtube
The Periodic Table By Energy Levels
What Is The Isotope Of 15 Protons And 15 Electrons And 17 Electrons Quora

Fall 11 Week 5 Atomic Structure Ppt Video Online Download
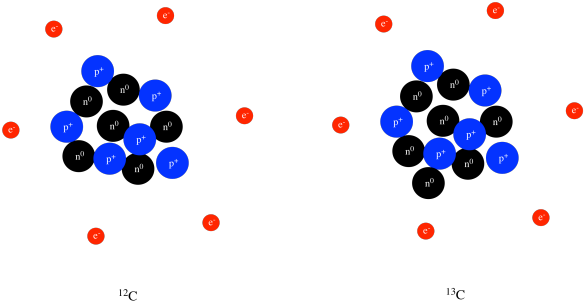
Structure Reactivity Atoms Protons Neutrons Electrons
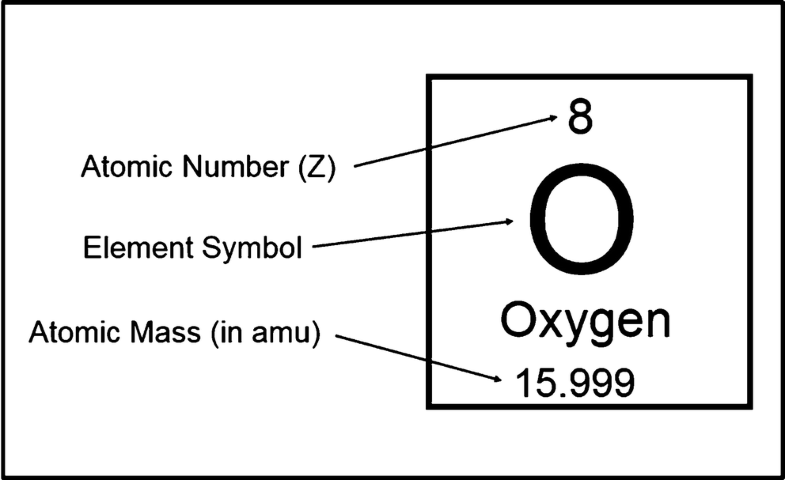
Atomic Number Mass Number And Isotopes Springerlink
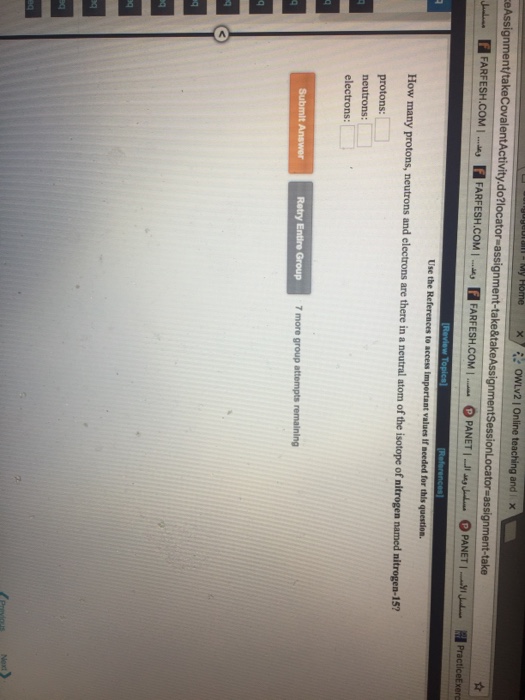
Solved How Many Protons Neutrons And Electrons Are There Chegg Com

Isotopes And Ions Isotopes Atoms Of The Same Element That Have Different Masses Have The Same Number Of Protons And Electrons But Different Numbers Of Ppt Download
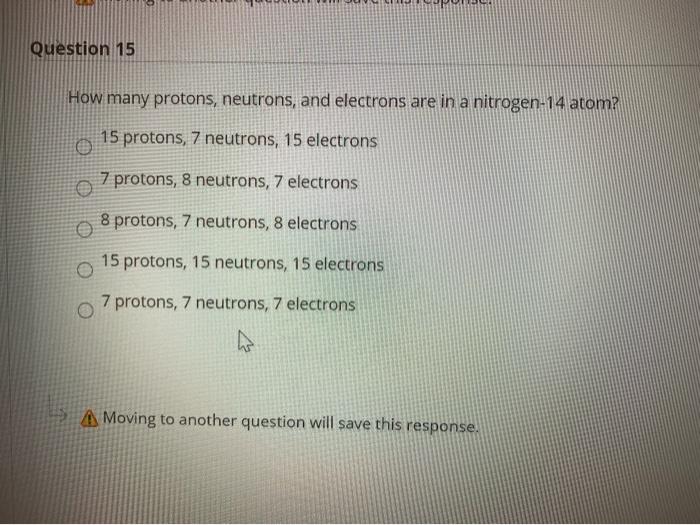
Solved Question 15 How Many Protons Neutrons And Electr Chegg Com

Atomic Structure Protons Electrons Neutrons Youtube
Q Tbn And9gcrqsfkbpshao7szli0adyzwe2lbthton Gifrjf24hcv0eko3zq Usqp Cau

The Numbers Of Electrons Protons And Neutrons In A Species Are Equal To 18 16 And 16 Youtube

Lakhmir Singh Chemistry Class 9 Solutions For Chapter 4 Structure Of Atom Free Pdf

How To Find The Number Of Protons Neutrons And Electrons

Solved 1 A Give The Number Of Protons Neutrons And El Chegg Com
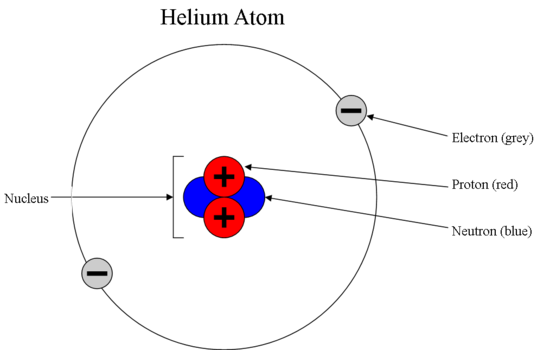
Sub Atomic Particles Chemistry Libretexts

Atomic Structure Nucleons Atomic Number Ppt Download
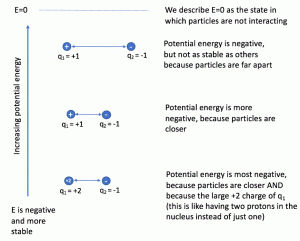
2 3 Atomic Structure And Symbolism General Chemistry 1 2

How To Find The Number Of Protons Electrons Neutrons For Nitrogen N Youtube
Introduction To The Atom Video Khan Academy

Review With Answers

Atoms Of Different Elements Having The Same Mass Number Are Known As

B What Are The Stable Isotopes Of Nitrogen N 14 N 15 C List Two Radioactive Course Hero
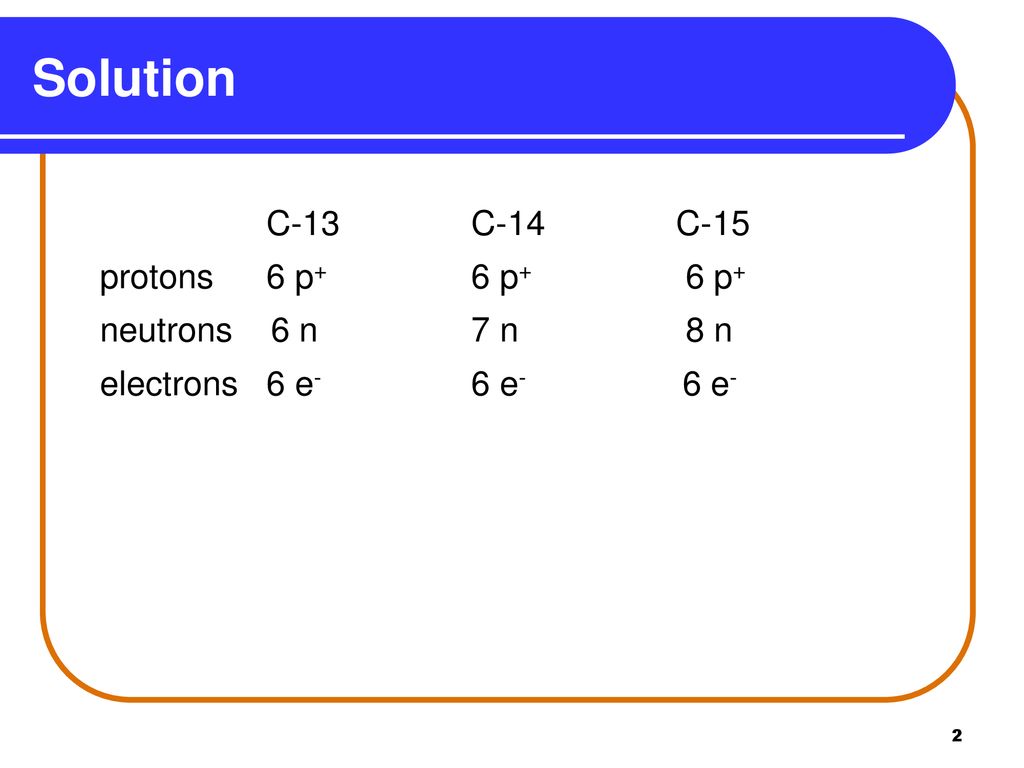
Learning Check Naturally Occurring Carbon Consists Of Three Isotopes Carbon 13 Carbon 14 And Carbon 15 State The Number Of Protons Neutrons And Ppt Download

Atoms And Elements The Nature Of Matter Matter Refers To Anything That Takes Up Space And Has Mass Matter Refers To Anything That Takes Up Space And Ppt Download
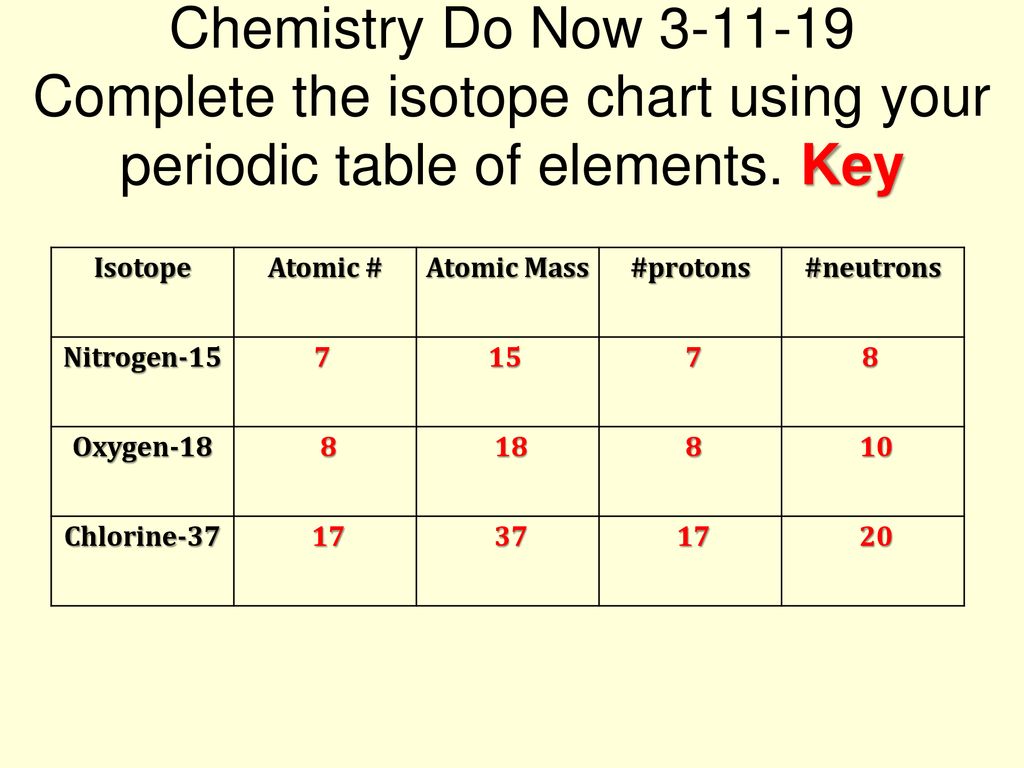
Atomic Atomic Mass Protons Neutrons Nitrogen 15 Oxygen Ppt Download

Solved 1 An Atom Of Nitrogen With A Mass Number Of 15 An Chegg Com
Http Www Lfatq Org Uk Wp Content Uploads Sites 14 01 Lfatq Intervention Resources Chemistry Topic 1 Pdf
Neutron Wikipedia

How To Find The Number Of Protons Electrons Neutrons For Potassium K Youtube

Difference In 17cl 35 And 17cl 37 Is Of
Http Katrinaschneider Weebly Com Uploads 2 1 5 8 Physci2 2 Pdf
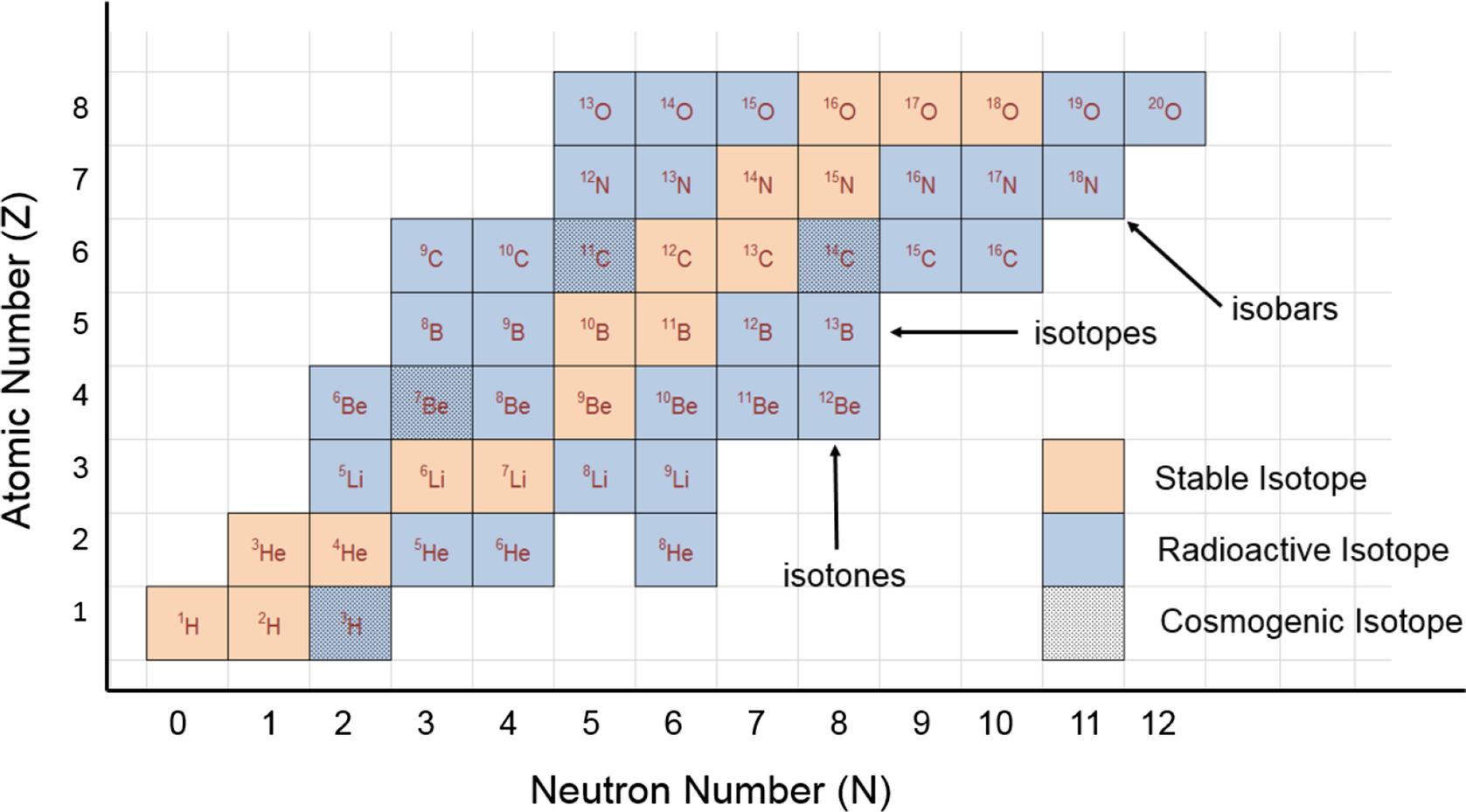
Atomic Number Mass Number And Isotopes Springerlink

Ncert Exemplar Class 9 Science Solutions Chapter 4 Structure Of Atom Access Free Pdf

Dublin Schools Lesson Isotopes How Do The Number Of Subatomic Particles Differ For Atoms From The Same Element

Isotopes Of Beryllium Wikipedia

First Elements In The Periodic Table Ppt Video Online Download

The Number Of Protons Neutrons And Electrons In Particles A To E

Calculate The Number Of Electrons Protons And Neutrons In The Fol

Protons Neutrons And Electrons Flashcards Quizlet

Cbse Ncert Solution For Class 9 Chemistry Structure Of The Atom
Q Tbn And9gcrqsfkbpshao7szli0adyzwe2lbthton Gifrjf24hcv0eko3zq Usqp Cau

Worked Example Identifying Isotopes And Ions Video Khan Academy
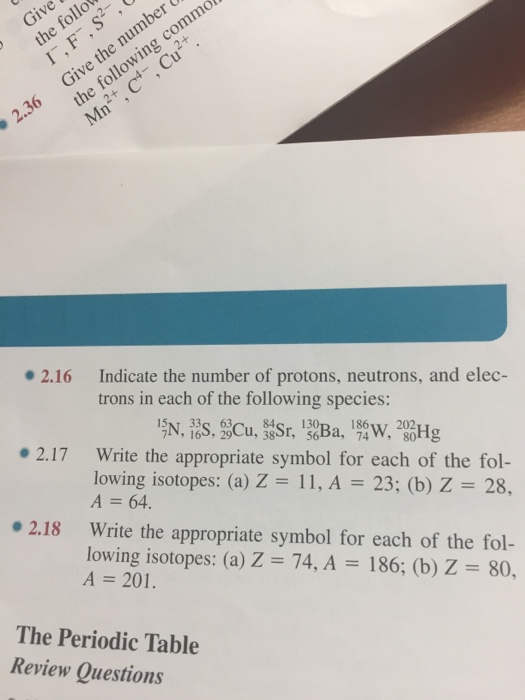
Solved Indicate The Number Of Protons Neutrons And Electr Chegg Com

Protons

Luj Zu 5 Calculate The Number Of Electrons Protons And Neutrons In The Following Molecules Ions

Average Atomic Mass Video Khan Academy
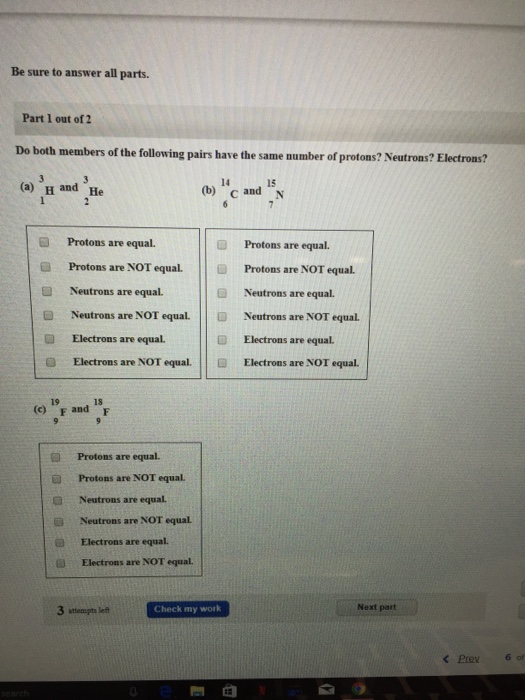
Solved Do Both Members Of The Following Pairs Have The Sa Chegg Com

Atoms Ppt Download
Solved Atomic Number Element Mass Sumber A Protons Neut Chegg Com
Www Cpsk12 Org Cms Lib8 Mo Centricity Domain 3297 Atomic structure worksheet answers Pdf

Atomic Structure And Symbolism Chemistry For Majors
Www Manhassetschools Org Cms Lib Ny Centricity Domain 796 Atoms practice qs answer key Pdf

How To Find The Number Of Protons Electrons Neutrons For Nitrogen N Youtube



