A Jug Of Water Will Usually Be Cooler Than The Surrounding Air This Is Because

How To Calculate The Time Needed To Cool Food Down Food Crumbles
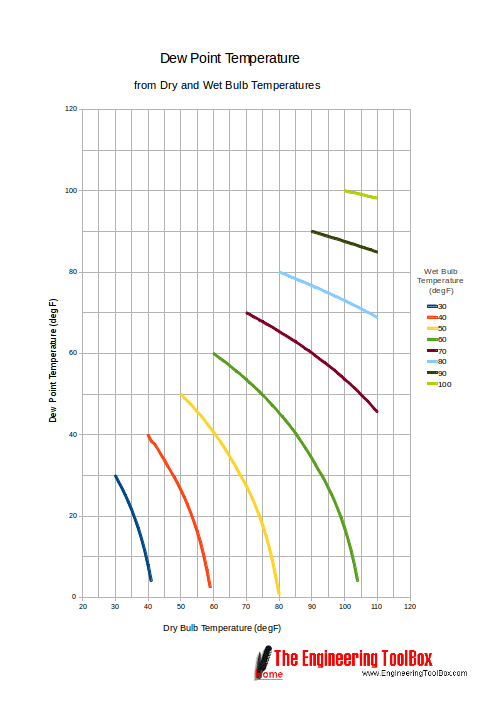
Dry Bulb Wet Bulb And Dew Point Temperatures
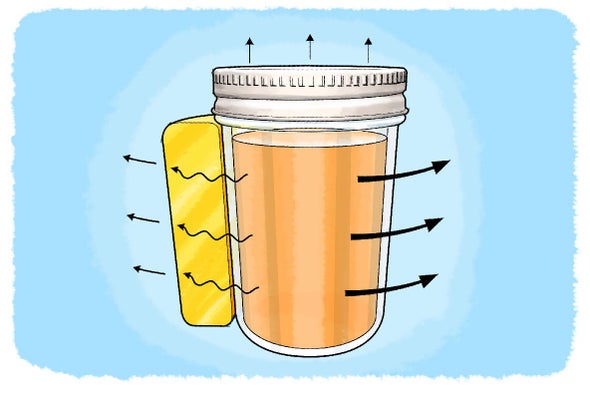
Stay Warm With Thermal Insulation Scientific American

Stop Heat From Escaping Testing Insulation Materials Activity Teachengineering

Phase Change And Latent Heat Physics

Why Do Some Drinks Sweat Wonderopolis
The heat sink has to be cooler than the object that needs cooling or it will not do its job For example, a bucket of ice will cool a bottle of wine because it becomes a sink for heat in the liquid.
/GettyImages-604349952-5aa6e0eea18d9e0038915154.jpg)
A jug of water will usually be cooler than the surrounding air this is because. Water vapor, water vapour or aqueous vapor is the gaseous phase of waterIt is one state of water within the hydrosphereWater vapor can be produced from the evaporation or boiling of liquid water or from the sublimation of iceWater vapor is transparent, like most constituents of the atmosphere Under typical atmospheric conditions, water vapor is continuously generated by evaporation and. The bottle will transfer its heat to the surrounding cold water, and that water will only become increasingly warm due to the bucket’s insulation In this scenario, the bottle will transfer its heat until it essentially becomes the same temperature as the water, and then it cannot become any cooler. Hotter, less dense air rises (just like the hot red water) while colder, more dense air sinks (like the cold blue water) The warmer air is less dense, Donaher explains, because its molecules are.
The evaporation from the wet muslin is reduced when air contains more water vapor The Wet Bulb temperature is always between the Dry Bulb temperature and the Dew Point For the wet bulb, there is a dynamic equilibrium between heat gained because the wet bulb is cooler than the surrounding air and heat lost because of evaporation. A condition in which water droplets in air do not freeze even when the air cools below the freezing point Dew Moisture that is condensed onto objects from the atmosphere, usually during the night, when the ground and leaf surfaces become cooler than the surrounding air. Stable air has an ELR less than the MAR—06°C/100m On a graph, an ELR to the right of the MAR is stable Unstable air has an ELR greater than the DAR—1°C/100m On a graph, an ELR to the left of the DAR is unstable Conditionally unstable air has an ELR between the DAR and the MAR –means that the air is unstable if saturated, stable if dry So, dry air is more stable than moist air and.
The presence of the ice cubes causes the plastic sheet to become cooler than the surrounding air Since heat travels from a hotter to a cooler region, we would expect the warmer water vapour from the air in the bottle to lose heat to the cooler plastic sheet and condense to form water droplets Thus, the answer for Part (a) would be. Hence, warm air rises spontaneously—if that air is warmer than air immediately around it at the same altitude, on small scales Stability and Instability One way that air (on small scales) can become warmer than immediately surrounding air and rise spontaneously occurs when air is in contact with, say, sunheated ground or an ocean surface. Stable air has an ELR less than the MAR—06°C/100m On a graph, an ELR to the right of the MAR is stable Unstable air has an ELR greater than the DAR—1°C/100m On a graph, an ELR to the left of the DAR is unstable Conditionally unstable air has an ELR between the DAR and the MAR –means that the air is unstable if saturated, stable if dry So, dry air is more stable than moist air and.
The evaporation from the wet muslin is reduced when air contains more water vapor The Wet Bulb temperature is always between the Dry Bulb temperature and the Dew Point For the wet bulb, there is a dynamic equilibrium between heat gained because the wet bulb is cooler than the surrounding air and heat lost because of evaporation. However, water heats up much more slowly than land and so the air above the land will be warmer compared to the air over the ocean The warm air over the land will rise throughout the day, causing low pressure at the surface Over the water, high surface pressure will form because of the colder air To compensate, the air will sink over the ocean. Water cools slower than air so it will increase the rate of condensation A metal cone could also be added to the smaller bottle to speed condensation My idea was to have it put in a car and have the a/c run over the external of the reservoir to cool the water and then have it stored in the car for drinking water.
For flowing water to freeze, the surrounding air has to be colder than 32°F, because the flowing water mixes with itself So, the colder water on the surface mixes with the warmer water from the bottom, and the average temperature is somewhere between the two. The presence of the ice cubes causes the plastic sheet to become cooler than the surrounding air Since heat travels from a hotter to a cooler region, we would expect the warmer water vapour from the air in the bottle to lose heat to the cooler plastic sheet and condense to form water droplets Thus, the answer for Part (a) would be. However, water heats up much more slowly than land and so the air above the land will be warmer compared to the air over the ocean The warm air over the land will rise throughout the day, causing low pressure at the surface Over the water, high surface pressure will form because of the colder air To compensate, the air will sink over the ocean.
Hot air with a low relative humidity may actually contain more water vapor than cool air with a high relative humidity True If very cold air is brought indoors and warmed with no change in its moisture content, the saturation vapor pressure of this air will ____ and the relative humidity of this air will ____. Cooler Air over the Ocean In the morning, the land has been radiating heat into space all night and, because it takes less energy loss to decrease the temperature of the land than it does to decrease the temperature of the ocean, the land is cooler than the ocean As a result, the air above the land is cooler than is the air above the ocean. Your right hand should feel cooler—this is because the alcohol boils at a lower temperature (181°F) than the water (212°F) This means that the alcohol will evaporate from your skin faster, carrying heat energy with it as it evaporates, making your right hand feel cooler than your left hand (for more information about thermal energy, see.
Water is a great conductor of changes in temperature I am sure if you had a log that was 978 degrees F, it would not feel as hot as the water And a sheet of copper at 978 degrees F might feel hotter than the water It is also true that the normal temperature of the surface of your hand is cooler than your core temperature (986 F). Usually, air at a beach is a few degrees cooler than the air just a few miles inland during the day Likewise, during the night, the air at the coast may not cool down quite as much as at inland locations I think that the temperature of the ocean water affects the surrounding air because of the constant breezes that blow over it I'm not. Convection The transfer of energy by movements of masses in a liquid or a mass In meteorology the energy transfer is vertical Convection in the atmosphere If the ground is hot, energy is transferred to the air molecules by conduction Heated air parcel will rise and cooler air sinks to replace the rising hot air.
The bottle creates a pool of cold air, and because the density of colder air is higher than that of warmer air, the cooler air stays inside the wine cooler The air in the insulating walls is slightly warmer than the air surrounding the bottle, but cooler than the air surrounding the cooler. Answer choices because they are large bodies of salt water that easily evaporate salt into the air because they are large bodies of salt water that fresh water can be evaporated from because the salt water condenses easily to become a gas in the air because the salt water runs off onto land to fill lakes and streams. This heated air expands to become less dense than the cooled air above Variations in surface heating and other factors result in surrounding cooler, denser air pushing in and forcing the lighter air up In this way the warm air is carried upward This motion of the air, resulting in the transport and mixing of its properties, is called.
Water can condense on hot parts only if those parts are cooler than the dew point of the air inside the engine If an engine is not allowed to come to full operating temperature, the water that has condensed on the walls of the block or on other cooler parts will not be evaporated, nor will the water vapor inside the engine be driven off and. When plants evaporate water into the surrounding air, they modify the properties of the air (usually assumed to be saturated or 100% Rh) and the humidity of the On warm days, forests tend to be cooler than deserts because the trees are actively cooling their leaves and the surrounding air through transpiration. Why Does Preventing Condensation on a Bottle Keep it Cool?.
I keep him in a thick n sturdy 3 gallon Italian glass refillable water bottle It is quite heavy and he seems to be enjoying his new home My question is this Since I have not heated the water in any way and do not have a heater installed in the 3 gallon jug,shouldn't the 3 gallon jug of water that he swims in be the same temp as the surrounding air or slightly cooler or warmer or ?. The thin, warm, moist layer of air over the pond then mixes with the cooler air from the land As it cools, condensation occurs and a fog forms It looks like steam rising off the water, hence the. If the mirror is much cooler than the water and if there is a significant amount of vapor in the air, you may actually see streams of condensed water running down the mirror Light traveling through the film of water bends, creating a clouded appearance on the mirror.
2 The Salt Water Bottle An old cowboy trick is to fill milk jugs, or other sealed plastic containers, with salt water and place a few in the stock tank Saltwater has a lower freezing point than freshwater and tends to stay liquid even in the coldest weather In fact a water to salt mixture of about 31 won’t freeze until around 5 Fahrenheit. That’s why running water is usually cooler than standing water The movement of the water exposes more of it to the air, increasing the amount of evaporation In cases of whitewater, where the water is being thrown into the air by rocks and other obstacles, evaporation increases even more, making the water even cooler. The wind speed tends to decrease after sunset because at night the surface of the Earth cools much more rapidly than does the air above the surface During the day it is very easy for the air to mix and cause surface gusts If there is a low pressure area or storm in the region the winds will blow day or night.
Look for water around and under rocks Rocks slow evaporation, so dew or rainwater may linger around them a little longer Turn over halfburied stones in the desert just before dawn and dew may form on their surface (This works because the base of the stone is cooler than the surrounding air) 9. Cooler Air over the Ocean In the morning, the land has been radiating heat into space all night and, because it takes less energy loss to decrease the temperature of the land than it does to decrease the temperature of the ocean, the land is cooler than the ocean As a result, the air above the land is cooler than is the air above the ocean. Lifting or lowering a layer of air changes causes the air to stretch out or compact as the air density aloft increases or decreases When the air stretches out, it cools more quickly The air at the top of the layer will be cooler than the air at the bottom, which steepens the environmental lapse rate and increases instability.
However, water heats up much more slowly than land and so the air above the land will be warmer compared to the air over the ocean The warm air over the land will rise throughout the day, causing low pressure at the surface Over the water, high surface pressure will form because of the colder air To compensate, the air will sink over the ocean. Cold Air Mass A body of air that is cooler than the surrounding air Cold front The front of a cold air mass As it pushes warmer air in front of it, clouds form and there is usually precipitation, wind and cooler temperatures Condensation Water vapor turning into a liquid Makes clouds when moisture in the air condenses on small particles. When water droplets form on the outside of your can or bottle, the surrounding air warms the droplets and makes your drink warmer as a result!.
Fog that forms when warm, moist air from the sea blows onto cooler land, where the air cools and water vapor condenses at ground level Radiation fog Fog that occurs when earth's surface and the air near the surface cool by radiation during the night, and water vapor in the air condenses because it cools below its dew point. Blimps, balloons, and other lighterthanair craft fly because they are lighter than air surrounding them They float upward like a hollow ball floats to the top of water Balance the two containers on the scale Add equal amounts of water and cooking oil to each container The amount is not important One drop of water will always be heavier. Of temperature in a layer of surrounding air will likely be, and usually is different than the dry rate As a result, the parcel’s temperature usually will be different than the surrounding air, and the parcel may be stable or unstable as shown See Figure 2 A key player in convection is latent heat stored in water vapor To create water.
The presence of the ice cubes causes the plastic sheet to become cooler than the surrounding air Since heat travels from a hotter to a cooler region, we would expect the warmer water vapour from the air in the bottle to lose heat to the cooler plastic sheet and condense to form water droplets Thus, the answer for Part (a) would be. Water is unique because _____ It would sink back to the surface because it was more dense than the surrounding air Condensation nuclei, tiny particles of matter in the air, are important because _____ the western coast of the US where the ocean water is usually cooler than the air This fog forms in valleys at night. Cold moist air feels colder than dry moist air (and hot moist air feels hotter than hot dry air) because moist air has a higher specific heat than does dry air Diatomic nitrogen and oxygen molecules form the bulk of the atmosphere Water is a polyatomic molecule that can rotate and translate in 3 dimensions.
As a result, an adiabatically rising air parcel (shown by the red or blue lines) would be cooler than the surrounding environment In this case, the air parcel would cease rising because it is cooler and has a higher density than the surrounding air At any given height, it will more likely sink, a condition known as a stable atmosphere. In the late fall and early winter, the situation is reversed Land surfaces cool off quickly in response to cooler weather, but the same property of water that makes it slow to absorb heat also causes it to cool slowly As a result, continents are usually cooler than the oceans surrounding them during the winter. Specifically, the air should be cooler than the surrounding air and contain a large amount of liquid water or ice This unique blend creates the bubble appearance of these unique clouds Interestingly, these clouds usually indicate that extreme weather conditions may be present 7 Goodness great balls of lighting.
Air released from a tire is cooler than its surroundings because rising air parcels will cool off faster than the surrounding air because rising air will cool off slower (and therefore be warmer as it rises) than the surrounding air The absorptivity of greenhouse gases in the troposphere is enhanced by the.
/male-hand-serving-water-of-a-water-cooler-in-plastic-cup--687403446-5a7b8254ae9ab80036781551.jpg)
What To Know Before Buying A Water Cooler Or Dispenser

Lecture 6 Ideal Gas Law Rising And Sinking Air
/GettyImages-604349952-5aa6e0eea18d9e0038915154.jpg)
Ideal Baby Room Temperature For A Newborn
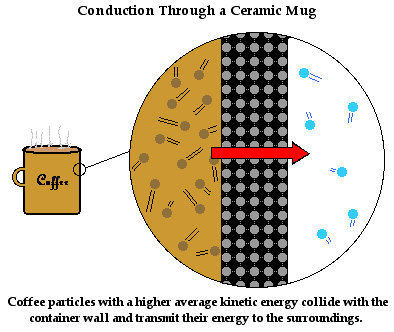
The Physics Classroom Tutorial
Chilling Science Evaporative Cooling With Liquids Scientific American
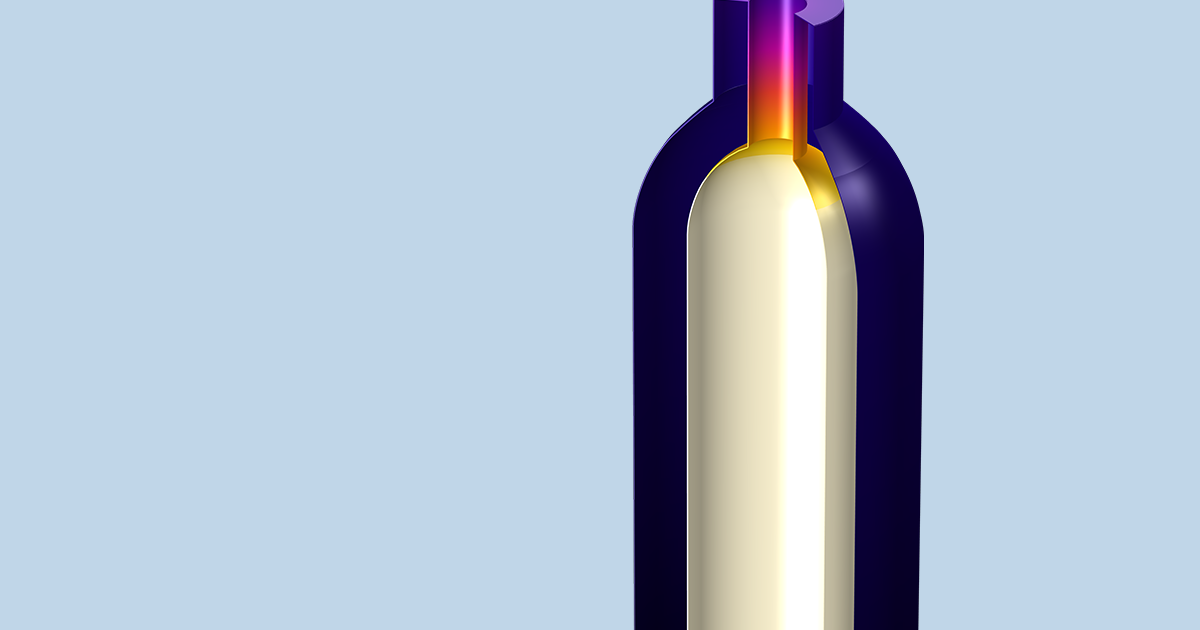
Can A Wine Cooler Actually Keep Your Beverage Cold Comsol Blog

Phase Change And Latent Heat Physics
/509796303-56a131425f9b58b7d0bcebe7.jpg)
Does Blowing On Hot Food Really Make It Cooler
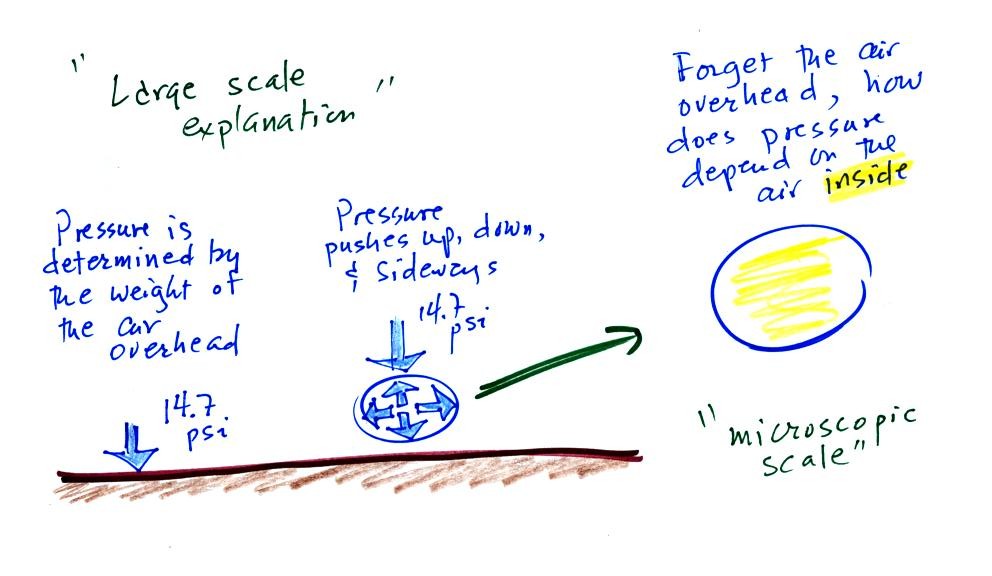
Lecture 6 Ideal Gas Law Rising And Sinking Air

Best Reusable Water Bottles To Give As Gifts In 21 Cnet

Temperature Change And Heat Capacity Physics
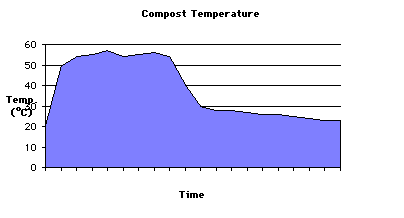
Compost Physics Cornell Composting

Convection Physics

Heat A Simple Introduction To The Science Of Heat Energy

Heating As A Transfer Of Energy Heat Energy Transfer Siyavula

A Homemade Humidifier 10 Ways To Combat Dry Air
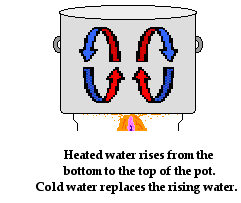
The Physics Classroom Tutorial

Plastic Bottle Manufacturing Process How Plastic Bottles Are Made

Heat Capacity Of Water Vs Heat Capacity Of Oil Science Project Education Com

Heat A Simple Introduction To The Science Of Heat Energy

Vanishing Baking Soda Scientific American
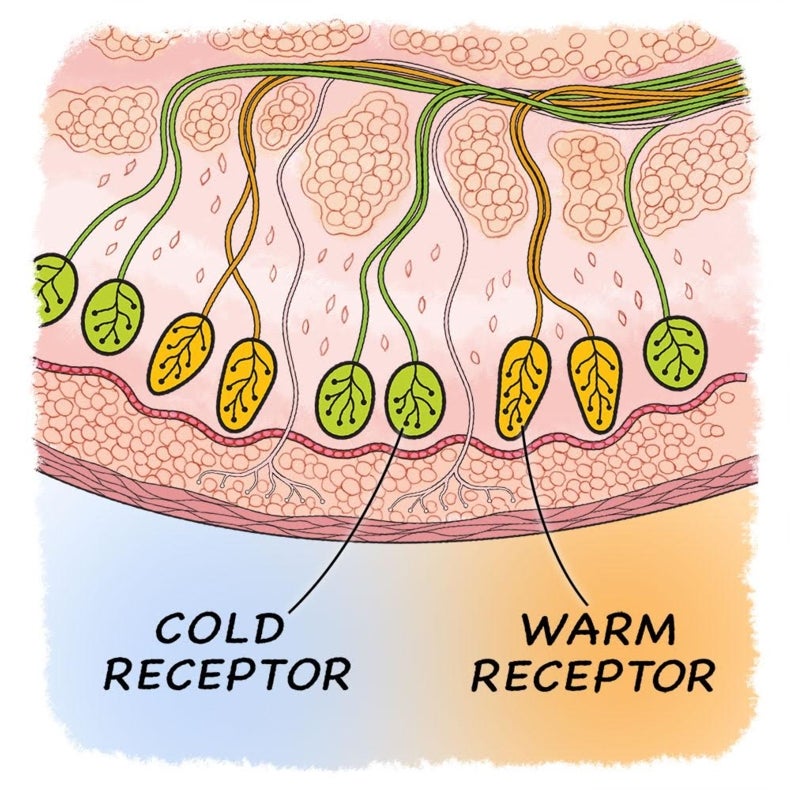
Cold Or Warm Can We Really Tell Scientific American
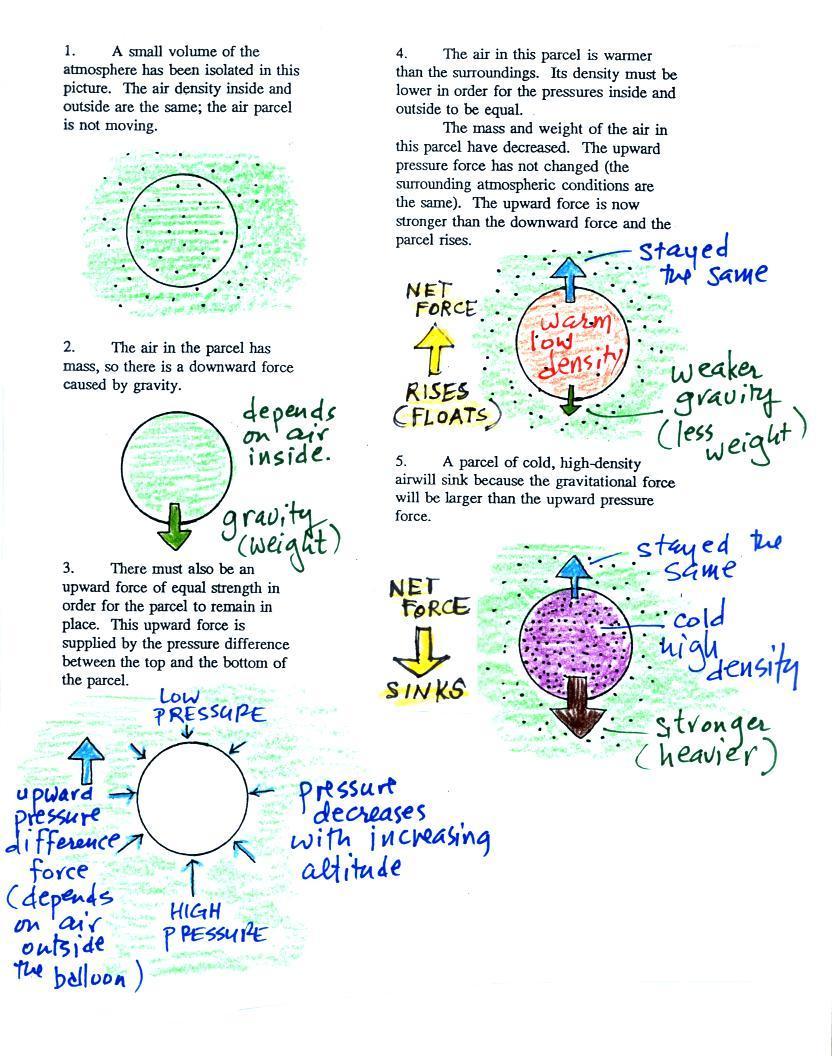
Lecture 6 Ideal Gas Law Rising And Sinking Air

The Burning Candle Rising Water Experiment

How To Stay Cool Without Air Conditioning Prevent Heat Exhaustion Wired

What Is The Best Insulator Air Styrofoam Foil Or Cotton Activity Teachengineering

Lecture 6 Ideal Gas Law Rising And Sinking Air
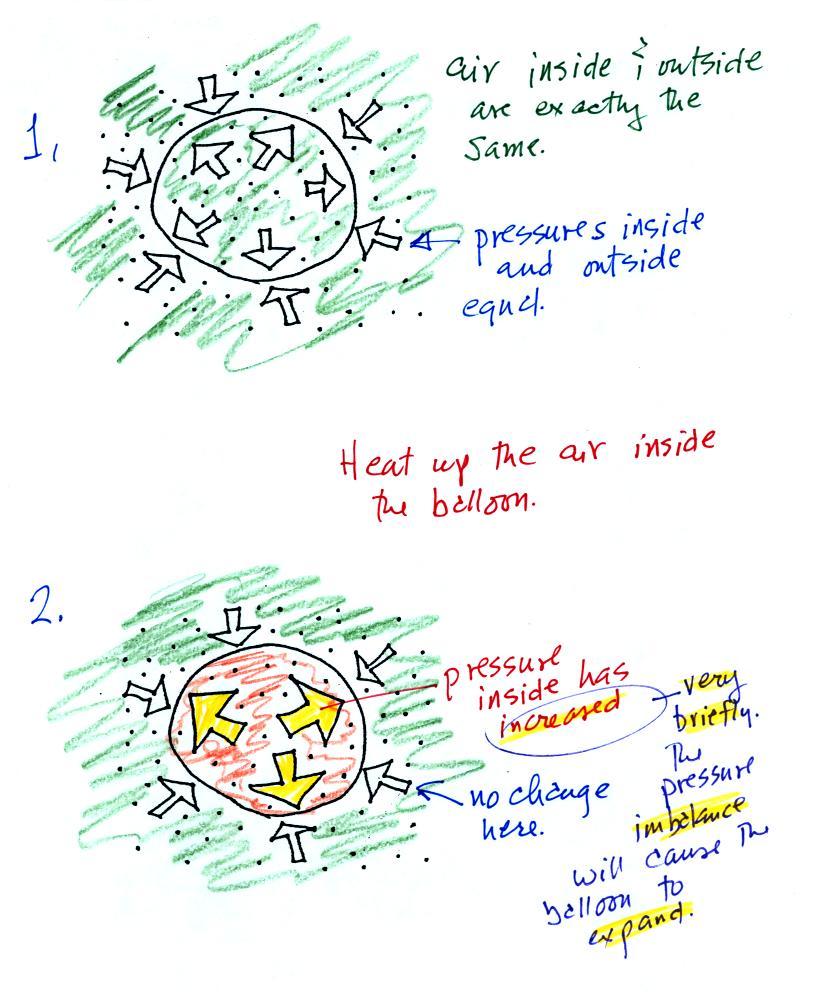
Lecture 6 Ideal Gas Law Rising And Sinking Air
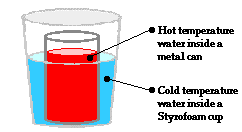
The Physics Classroom Tutorial

Convection Physics

Guide To Keeping Warm With A Hot Water Bottle Hot Water Bottle Shop Hotwaterbottleshop Co Uk

Cooling Tower Wikipedia

Vacuum Flask Wikipedia

11 2 Heat Specific Heat And Heat Transfer Texas Gateway

11 2 Heat Specific Heat And Heat Transfer Texas Gateway
Q Tbn And9gctoq6rpeajywtzzofux4sbox3mdygi5v3q4gd 6hxqvdcheoyko Usqp Cau

Thermal Expansion Of Solids And Liquids Physics
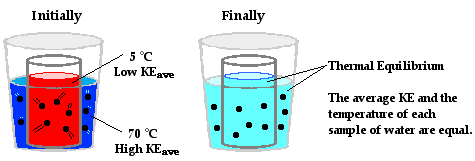
The Physics Classroom Tutorial
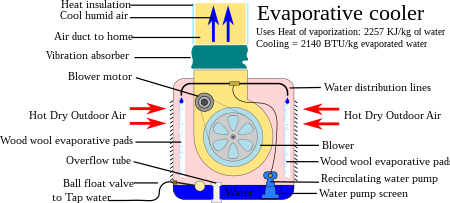
Evaporative Cooler Wikipedia

Mirrors Could Replace Air Conditioning By Beaming Heat Into Space Physics The Guardian
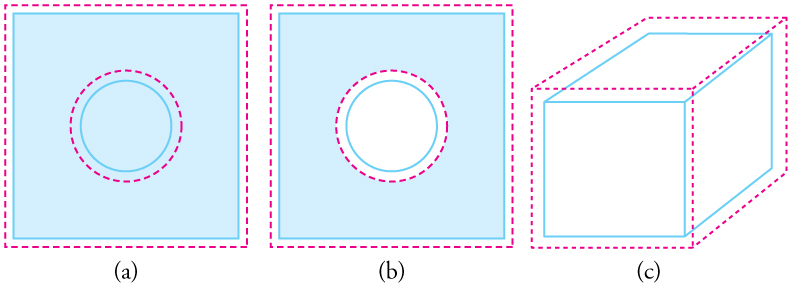
Thermal Expansion Of Solids And Liquids Physics
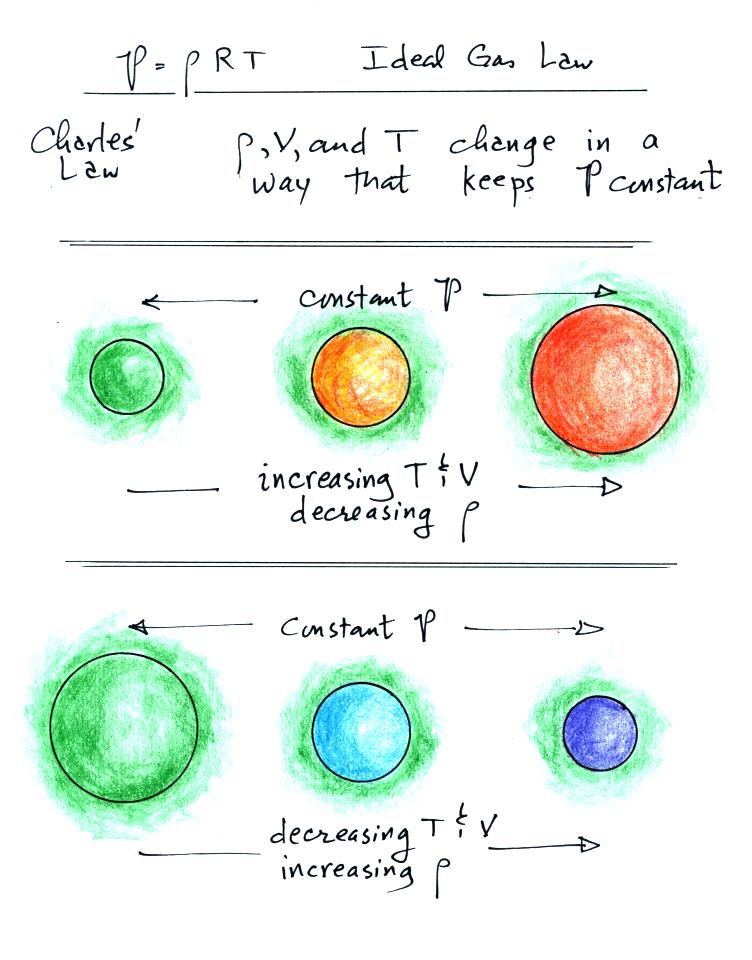
Lecture 6 Ideal Gas Law Rising And Sinking Air

Convection Physics
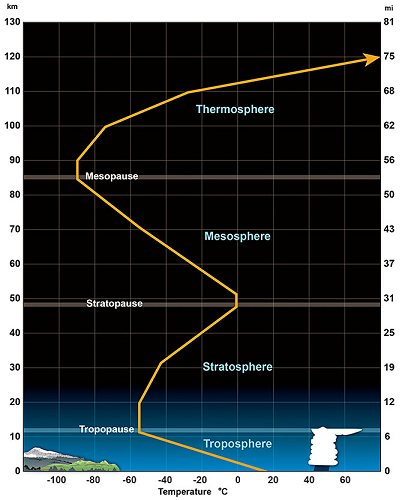
Temperatures Dry Bulb Web Bulb Dew Point
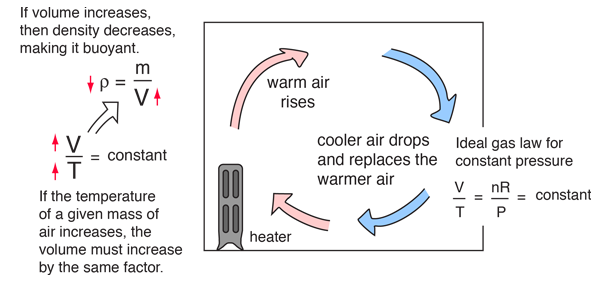
Heat Transfer
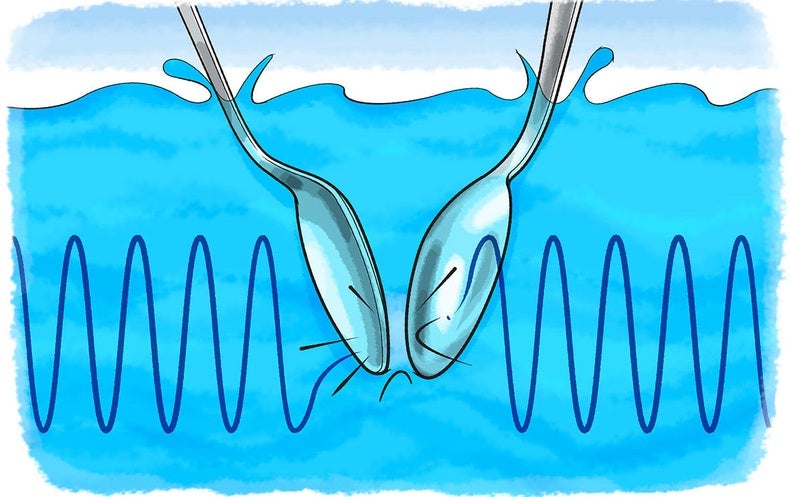
What Do You Hear Underwater Scientific American

Best Reusable Water Bottles To Give As Gifts In 21 Cnet

Guide To Keeping Warm With A Hot Water Bottle Hot Water Bottle Shop Hotwaterbottleshop Co Uk

Lecture 6 Ideal Gas Law Rising And Sinking Air
Q Tbn And9gcqj R9wp7alw3hasjlzrarlnlwqxj8cerbzlszbjg5oi1dk8nu5 Usqp Cau
Temperatures Dry Bulb Web Bulb Dew Point

Thermal Expansion Of Solids And Liquids Physics

c Earth The Creatures That Can Survive Without Water For Years

How Long Can You Survive Without Water c Future
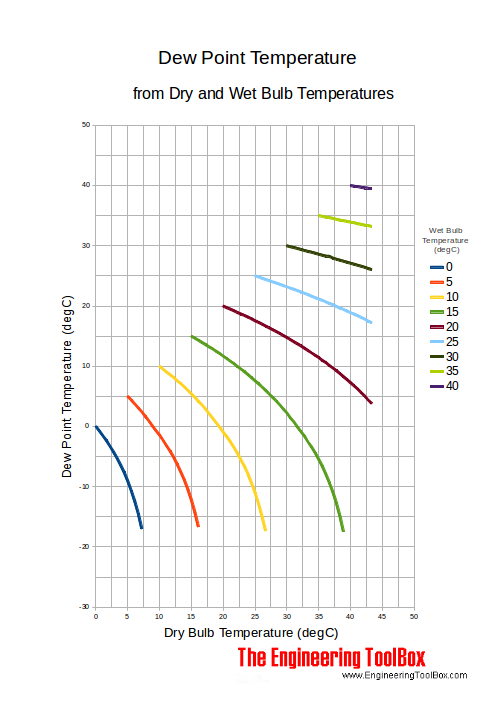
Dry Bulb Wet Bulb And Dew Point Temperatures
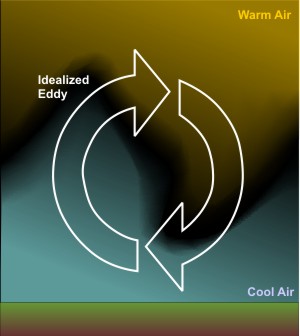
Energy Transfer Convection Meteo 3 Introductory Meteorology
Chilled Drink Calculator How Long To Wait

What Is The Best Insulator Air Styrofoam Foil Or Cotton Activity Teachengineering

Evaporative Cooler Wikipedia
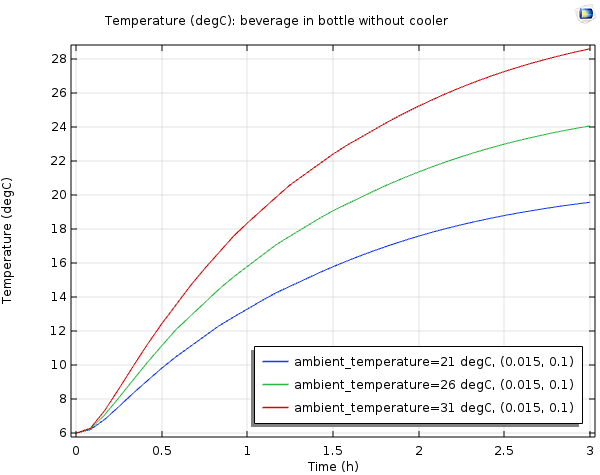
Can A Wine Cooler Actually Keep Your Beverage Cold Comsol Blog

Energy Transfer Convection Meteo 3 Introductory Meteorology

How To Stay Cool Without Air Conditioning Prevent Heat Exhaustion Wired
Q Tbn And9gcqfiaeflhtg5s10smvaefzbz4vaegnxbidpaque8icrgodftrf1 Usqp Cau

Evaporative Cooler Wikipedia

Does Water Expire

Is It True That Hot Water Freezes Faster Than Cold Water Or That Cold Water Boils Faster Than Hot Water Scientific American

Singing Glasses Scientific American

Energy Transfer Convection Meteo 3 Introductory Meteorology

Phase Change And Latent Heat Physics

Mix It Up With Oil And Water Scientific American

Heating As A Transfer Of Energy Heat Energy Transfer Siyavula

Convection Physics

Hot And Cold Extreme Temperature Safety

Heating As A Transfer Of Energy Heat Energy Transfer Siyavula

Heating As A Transfer Of Energy Heat Energy Transfer Siyavula
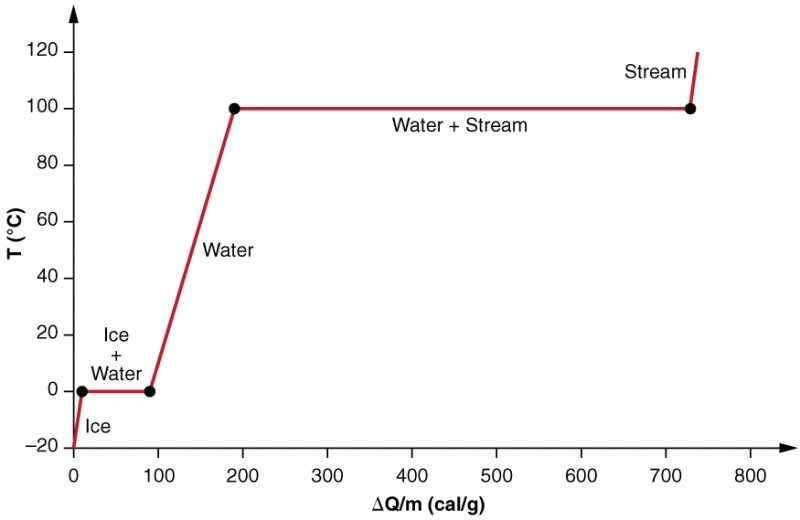
Phase Change And Latent Heat Physics

Can You Cool A House Without Air Conditioning c Future

Yes You Can Boil Water At Room Temperature Here S How Wired

Heating As A Transfer Of Energy Heat Energy Transfer Siyavula
Q Tbn And9gcqj R9wp7alw3hasjlzrarlnlwqxj8cerbzlszbjg5oi1dk8nu5 Usqp Cau
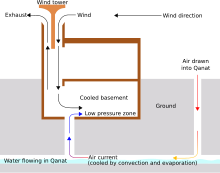
Evaporative Cooler Wikipedia

Boiling It Down To The Bubbles It Is About Heat Transfer A Lab Aloft International Space Station Research

Convection Physics

Water Dispenser Wikipedia
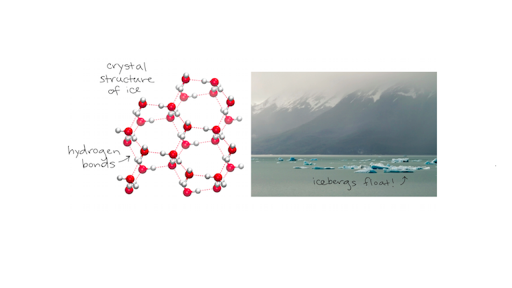
Specific Heat Heat Of Vaporization And Density Of Water Article Khan Academy

Heating As A Transfer Of Energy Heat Energy Transfer Siyavula

Heating As A Transfer Of Energy Heat Energy Transfer Siyavula
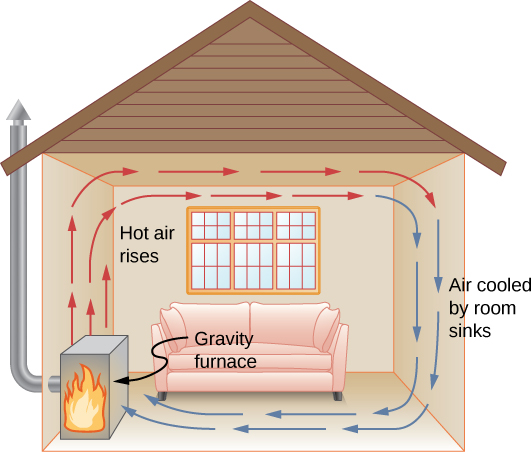
Mechanisms Of Heat Transfer University Physics Volume 2
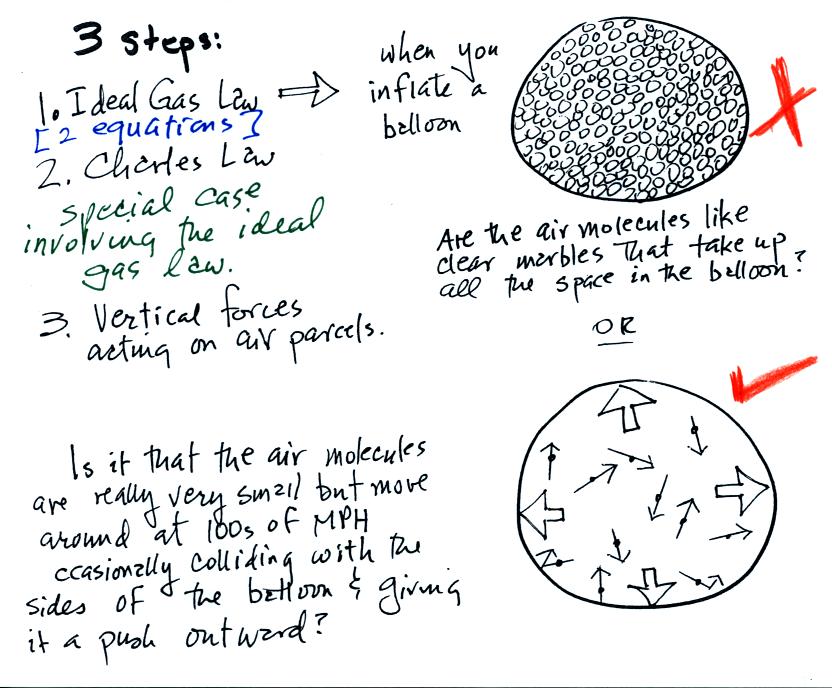
Lecture 6 Ideal Gas Law Rising And Sinking Air
:max_bytes(150000):strip_icc()/GettyImages-1174558875-8372ad85b3aa4e99a0612e043d58fc09.jpg)
What To Know Before Buying A Water Cooler Or Dispenser
/GettyImages-1061851344-670639e17b314169bf6f62ba09bfd259.jpg)
How Condensation And Evaporation Shape Our Weather
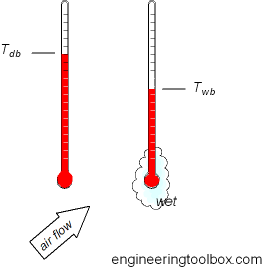
Dry Bulb Wet Bulb And Dew Point Temperatures

Can Water Go Bad And Make You Sick Time
Evaporative Cooler Wikipedia

What Are Convection Currents

Lecture 6 Ideal Gas Law Rising And Sinking Air

The Physics Of Keeping Cool Wired

Energy Transfer Convection Meteo 3 Introductory Meteorology



