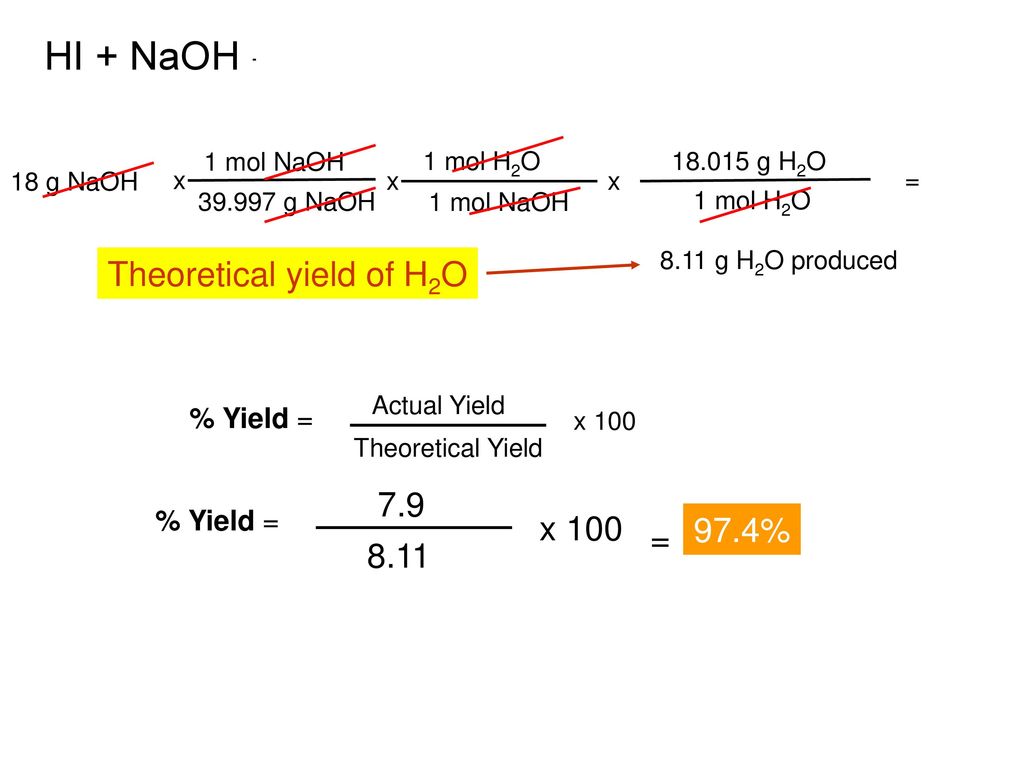
Hi + Naoh

Chemical Reaction Hi Naoh Youtube

Oh 2chi Ome Hcho Naoh C B Hi Acetoned Excess Of Hi 2hcho Naoh

Hi Naoh Solution 0 11n N 9 Reagents Accessories Titration Shop Hanna Instruments Fze
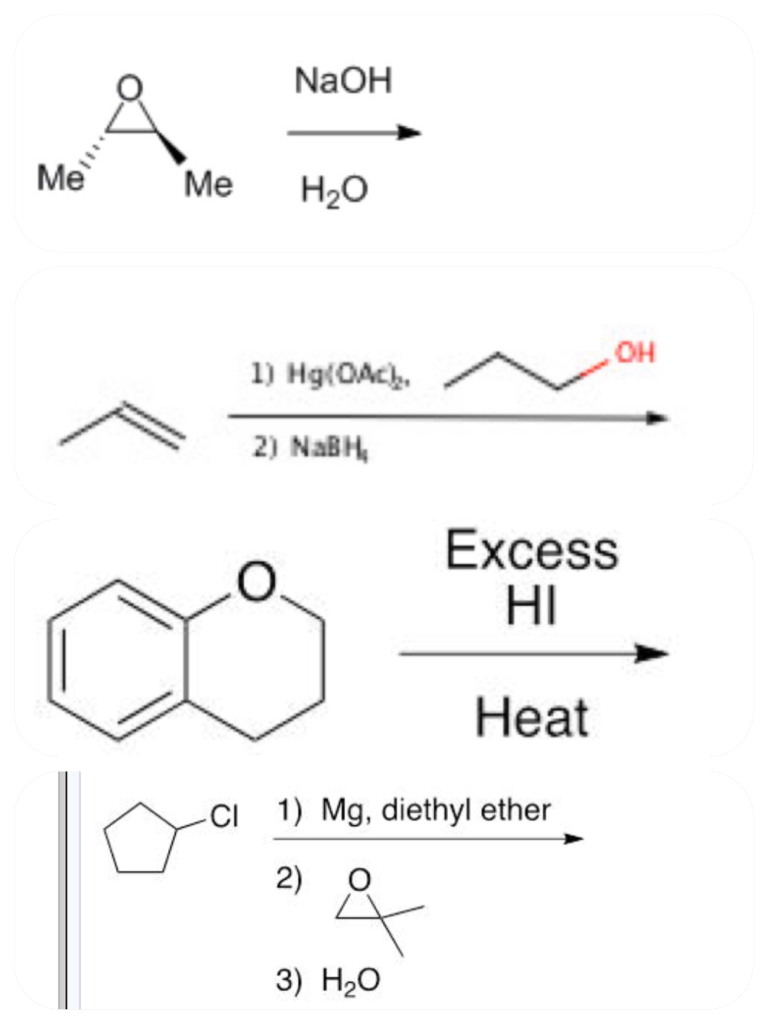
Solved Naoh Me Me H2o Oh 2 Nabh Excess Hi Heat Ci1 Mg Chegg Com

Hi Titration Solution 0 01 N Naoh 1l
2h2 G O2 G 2h2o L Thus H2 Is The Limiting Reagent Ppt Download
HiTech designs and builds wet scrubbers for HF and HCl neutralization (NaOH) or potassium hydroxide (KOH) and convert the halide gas to a salt in the solution H 2 HCl H 2 O NaOH = H 2 NaCl 2H 2 O H 2 HCl H 2 O KOH = H 2 KCl 2H 2 O.
Hi + naoh. HClO 4 NaOH → NaClO 4 H 2 O Check the balance Perchloric acid react with sodium hydroxide to produce sodium perchlorate and water Perchloric acid and sodium hydroxide diluted solutions Find another reaction Our channel Thermodynamic properties of substances The solubility of the substances Periodic table of elements. Hydrobromic acid, #"HBr"#, is a strong acid and sodium hydroxide, #"NaOH"#, is a strong base, so right from the start, you know that you're dealing with a neutralization reaction This should tell you that the net ionic equation will involve the hydrogen cationsor hydronium cations, #"H"_3"O"^()#produced by the strong acid and hydroxide anions produced by the strong base. Сoding to search 3 I2 6 NaOH cnd temp = 5 NaI NaIO3 3 H2O Add / Edited / Evaluation of information 50 out of 5 / number of votes 1 Please register to post comments ChemiDay you always could choose go nuts or keep calm with us or without.
Sodium hydroxide is sometimes called caustic soda or lye It is a common ingrediet in cleaners and soaps At room temperature, sodium hydroxide is a white, odorless solid Liquid sodium hydroxide is colorless and has no odor It can react violently with strong acids and with water Sodium hydroxide is corrosive. Using a pH indicator strip will tell you that NaOH (sodium hydroxide) is a strong alkaline This means it has a pH toward the top end of the pH scale, which ranges from 0 to 14 To calculate the exact pH, work out the molarity of the solution, then apply that to the formula for pH. Sodium hydroxide is sometimes called caustic soda or lye It is a common ingrediet in cleaners and soaps At room temperature, sodium hydroxide is a white, odorless solid Liquid sodium hydroxide is colorless and has no odor It can react violently with strong acids and with water Sodium hydroxide is corrosive.
The absorption peak of the spectra of NaCl and NaOH shifted right and grew larger with the successive increase of the solution concentration As shown in Fig 1 and Fig 2, the wavelength and absorbance at the peaks of NaCl and NaOH both increased as the solution concentration increased Download Download highres image (173KB). Direct link to this balanced equation Instructions on balancing chemical equations. What is sodium hydroxide (NaOH)?.
HI NaOH → NaI H2O Equation is already balanced. (ii) H 2 O 2 in NaOH soln HBr (peroxide initiator) Perbenzoic Acid, C 6 H 5 CO 3 H Select one alkene from the first group, and one reagent from the second group To practice using the drawing program Click Here To review the chemistry of alkenes Click Here When you press the "Show. (0 L) 00 q I 8 A 500 mt sample of vinegar has a concentration of 0800 M What volume of 0150 M NaOH is required to complete the titration?.
The absorption peak of the spectra of NaCl and NaOH shifted right and grew larger with the successive increase of the solution concentration As shown in Fig 1 and Fig 2, the wavelength and absorbance at the peaks of NaCl and NaOH both increased as the solution concentration increased Download Download highres image (173KB). Instructions To balance a chemical equation, enter an equation of a chemical reaction and press the Balance button The balanced equation will appear above. The corrosion behaviour of type 316L stainless steel in aqueous 30–50 wt% NaOH at temperatures up to 90 °C has been elucidated Exposure to room temperature environment showed parabolic weight loss behaviour, with corrosion rates of up to 04 mm/year Higher NaOH concentrations and exposure temperatures resulted in a reduced stability of the electrochemical passivity domain, associated.
Heat of Neutralization HCl (aq) NaOH (aq) Equal volumes, 500 mL, of 30 M hydrochloric acid and 30 M sodium hydroxide solutions having an initial temperature of 0°C react in a calorimeter The resultant solution records a temperature of 400°C The heat gained by the resultant solution can be calculated using. HCl(aq) NaOH(aq) > NaCl(aq) H 2 O(l) Energy Thermochemistry determine the heat exchanged at constant pressure, q = m c ∆T Calculating the limiting reactant, the change in enthalpy of the reaction, ∆H rxn, can be determined since the reaction was conducted under conditions of constant pressure ∆H rxn = q rxn / # moles of limiting reactant This reaction is classified as an. The chemical reaction between strong acid HI and strong base NaOH will be a neutralization reaction, it will lead to the production of a salt along with water So, the balanced chemical equation.
HI and NaOH HI NaOH ( H2O NaI H2CO3 and Sr(OH)2 H2CO3 Sr(OH)2 ( 2 H2O SrCO3 Ca(OH)2 and H3PO4 3 Ca(OH)2 2 H3PO4 ( 6 H2O Ca3(PO4)2 hydrobromic acid and barium hydroxide 2 HBr Ba(OH)2 ( 2 H2O BaBr2 zinc hydroxide and nitric acid Zn(OH)2 2 HNO3 ( 2 H2O Zn(NO3)2 aluminum hydroxide and hydrochloric acid Al(OH)3 3. I assume this is 10% w/v NaOH solution For 1 L of this solution, we have 100 g of NaOH NaOH molar mass = 23 16 1 = 40 g/mole 100/40 = 25 mole NaOH in 1 L = 25 M NaOH dissociates completely in solution, therefore OH = 25 M log(25) =. HI (aq) Phosphorous acid H 3 PO 3 Carbonic acid H 2 CO 3 Sulfuric acid H 2 SO 4 Formic acid HCOOH Name each of the following acids HClO 4 perchloric acid HCOOH formic acid H 3 PO 4 phosphoric acid HCl (aq) hydrochloric acid H 3 BO 3 boric acid H 2 SO 4 sulfuric acid HNO 2 nitrous acid HI (aq) hydroiodic acid CH 3 COOH acetic acid.
A few things to take into account to understand when and how Al₂O₃ reacts with NaOH 1 Pure, crystalline Al₂O₃ does not react with NaOH at room temperature In spite of its amphoteric nature, crystalline Al₂O₃ (as in corundum) requires high tempe. NaOH(aq) HNO3(aq) → NaNO3(aq) H2O(l) Titration 3 In order to use the molar ratio to convert from moles of NaOH to moles of HNO3, we need to convert from volume of NaOH solution to moles of NaOH using the molarity as a conversion factor The following is a sample study sheet for titration problems Study Sheet for AcidBase Titration. HI(ac) NaOH(ac) O HI(ag) NaOH(aq) H(1) Nal(aq) O HI(aq) NaOH(aq) H2O(l) Nalz(ag) OH(aq) 2NaOH(aq) H(1) Na I(ag) O HI(04) NaOH(aq) → H2O(1.
What is sodium hydroxide (NaOH)?. Write a balanced equation for the neutralization of HI(aq) and LiOH(aq) Express your answer as a chemical equation Identify ALL of the phases in your answer This is what I think, 2 HI (aq) LiO (aq) = LiI2 (aq) H2O (l) but masteringchemistrcom keeps saying its wrong, what am i doing wrong?. Hydrofluoric acid, #"HF"#, a weak acid, will react with sodium hydroxide, #"NaOH"#, a strong base, to produce aqueous sodium fluoride, #"NaF"#, and water #"HF"_ ((aq.
NaOH is a strong alkali and HCl acid is a strong acid respectively To identify the equivalence point in the titration, we use titration curves and indicatorsAccording to the concentration of acid and base solutions, we have to choose correct curve and indicator. NAoh Hi is on Facebook Join Facebook to connect with NAoh Hi and others you may know Facebook gives people the power to share and makes the world more. Sodium hydroxide (NaOH), also known as caustic soda or lye, is a highly versatile substance used in a variety of manufacturing processes Sodium hydroxide is a coproduct of chlorine production Share Follow Us on Facebook Twitter YouTube RSS Print this Page Uses & Benefits.
Four solutions of unknown HCl concentrations are titrated with solutions of NaOH The following table lists the volume of each unknown HCl solution, the volume of NaOH solution required to reach the equivalence point, and the concentration of each NaOH solution HCl Volume (mL) NaOH Volume (mL) NaOH (M) 2600 mL 3244 mL M. M NaOH is needed to titrate it?. When HNO3 reacts with NaOH the product that is formed is NaNO3 which is salt and H2O which is water You can write it as HNO3 NaOH = NaNO3 H2O So this is basically a Neutralization reaction between nitric acid(HNO3) and sodium hydroxide (NaOH.
Explanation HI is an acid while, NaOH is a base When an acidreacts with a base to form salt (NaI) and water, the reaction view the full answer Previous question Next question. Balance the reaction of Na H2O = NaOH H2 using this chemical equation balancer!. Mmol NaOH added = (100 mL)( M) = 0500 mmol The net ionic equation and the effect on the amounts of HA and A – are HA OH – 6 A– H 2O Initial 250 0500 0 After rxn 0 0 0500 The total volume at his point is the sum of the initial volume plus the volume of added titrant V = 250 mL 100 mL = 350 mL.
The high concentration of NaNO 3 inhibited the degree of hydration of cement and resulted a looser pore structure, which led to a relatively high leaching rate of Sr 2 However, the addition of NaOH compensated the negative effect on the hydration and pore structure and decreased effectively the leaching rate of Sr 2. Sodium hydroxide NaOH or HNaO CID structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities. NaOH is a strong alkali and HCl acid is a strong acid respectively To identify the equivalence point in the titration, we use titration curves and indicatorsAccording to the concentration of acid and base solutions, we have to choose correct curve and indicator.
Instructions on balancing chemical equations Enter an equation of a chemical reaction and click 'Balance' The answer will appear below;. HI NaOH → NaI H 2 O Check the balance Hydrogen iodide react with sodium hydroxide to produce sodium iodide and water Sodium hydroxide diluted solution. In this video we'll balance the equation NaOH HI = NaI H2O and provide the correct coefficients for each compoundTo balance NaOH HI = NaI H2O you'll.
Hydroiodic HI IHydrobromic HBr BrPerchloric HClO4 ClO4Hydrochloric HCl ClChloric HClO3 ClO3Sulfuric (1) H2SO4 HSO4Nitric HNO3 NO3Strong acids completely dissociate in aq solution (Ka > 1, pKa < 1) Conjugate bases of strong acids are ineffective bases Hydronium ion H3O H2O 1 00 Iodic HIO3 IO316 x 101 080 Oxalic (1) 2H2C2O4 HC2O4. Hydrobromic acid, #"HBr"#, is a strong acid and sodium hydroxide, #"NaOH"#, is a strong base, so right from the start, you know that you're dealing with a neutralization reaction This should tell you that the net ionic equation will involve the hydrogen cationsor hydronium cations, #"H"_3"O"^()#produced by the strong acid and hydroxide anions produced by the strong base. Always use the upper case for the first character in the element name and the lower case for the second character.
L) L 7 A 100 mf sample of an acid is titrated with 455 ml of 00 M Nao What is the concentration of the acid?. HI(aq) 1 NaOH(aq) 1 NaI 1 H 2 O 1 Units molar mass g/mol, weight g Please tell about this free chemistry software to your friends!. Sodium hydroxide solutions can freeze at temperatures beneath 60°F (155°C), depending upon NaOH concentration, which can cause release and buildup of solid NaOH Due to this relatively high freezing point of caustic soda solutions, pipes used in process flow are recommended to be insulated and heat traced.
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH It is a white solid ionic compound consisting of sodium cations Na and hydroxide anions OH − Sodium hydroxide is a highly caustic base and alkali that decomposes proteins at ordinary ambient temperatures and may cause severe chemical burnsIt is highly soluble in water, and readily. If you do get NaOH on your skin, immediately rinse it with a large volume of water Another option is to neutralize any base on the skin with a weak acid, such as vinegar, and then rinse with water Stir the sodium hydroxide, a little at a time, into a large volume of water and then dilute the solution to make one liter. Hydrofluoric acid, #"HF"#, a weak acid, will react with sodium hydroxide, #"NaOH"#, a strong base, to produce aqueous sodium fluoride, #"NaF"#, and water #"HF"_ ((aq.
L) L 7 A 100 mf sample of an acid is titrated with 455 ml of 00 M Nao What is the concentration of the acid?. Sodium hydroxide NaOH or HNaO CID structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities. Hydroiodic HI IHydrobromic HBr BrPerchloric HClO4 ClO4Hydrochloric HCl ClChloric HClO3 ClO3Sulfuric (1) H2SO4 HSO4Nitric HNO3 NO3Strong acids completely dissociate in aq solution (Ka > 1, pKa < 1) Conjugate bases of strong acids are ineffective bases Hydronium ion H3O H2O 1 00 Iodic HIO3 IO316 x 101 080 Oxalic (1) 2H2C2O4 HC2O4.

5 Questions For Exam 1 Fundamentals I Chemistry Chem 271 Docsity

Hi Sodium Hydroxide Solution 0 01n 1l

Hi Hydroiodic Acid Structure Molecular Mass Properties And Uses
A B Pressure Dependent Pl Evolution Of The A C Dotsnaoh And The B Download Scientific Diagram

Answered How Many Ml Of A 0 235 M Naoh Solution Bartleby
Wentzelchem Weebly Com Uploads 2 3 0 7 03 Neutralization Wksht Key Pdf

Organic Chemistry Halogen Derivatives Hi 41 1 Chci Naoh Product Major Oh Ch 0 Oh Ohc Oh 2 1 Oh Ch O Ch 0 Oh 4 3 Oh Kunduz

China Caustic Soda Naoh Flakes And Pearls 99 Min Qingdao Hiseachem Photos Pictures Made In China Com

A Br2 Hv Electrolysis 23 Which Of The Following React Organic Chemistry
2
Why Can T Alcohol React With Aqueous Naoh Physics Forums

How To Balance Naoh Hi Nai H2o Chemical Equation Naoh Hi Nai H2o Reaction Balance Naoh Hi Nai H2o Youtube

How To Balance Naoh Hi Nai H2o Chemical Equation Naoh Hi Nai H2o Reaction Balance Naoh Hi Nai H2o Youtube

Please If You Cant Answer All The Questions Do Not An Swer Any Of Them And Homeworklib
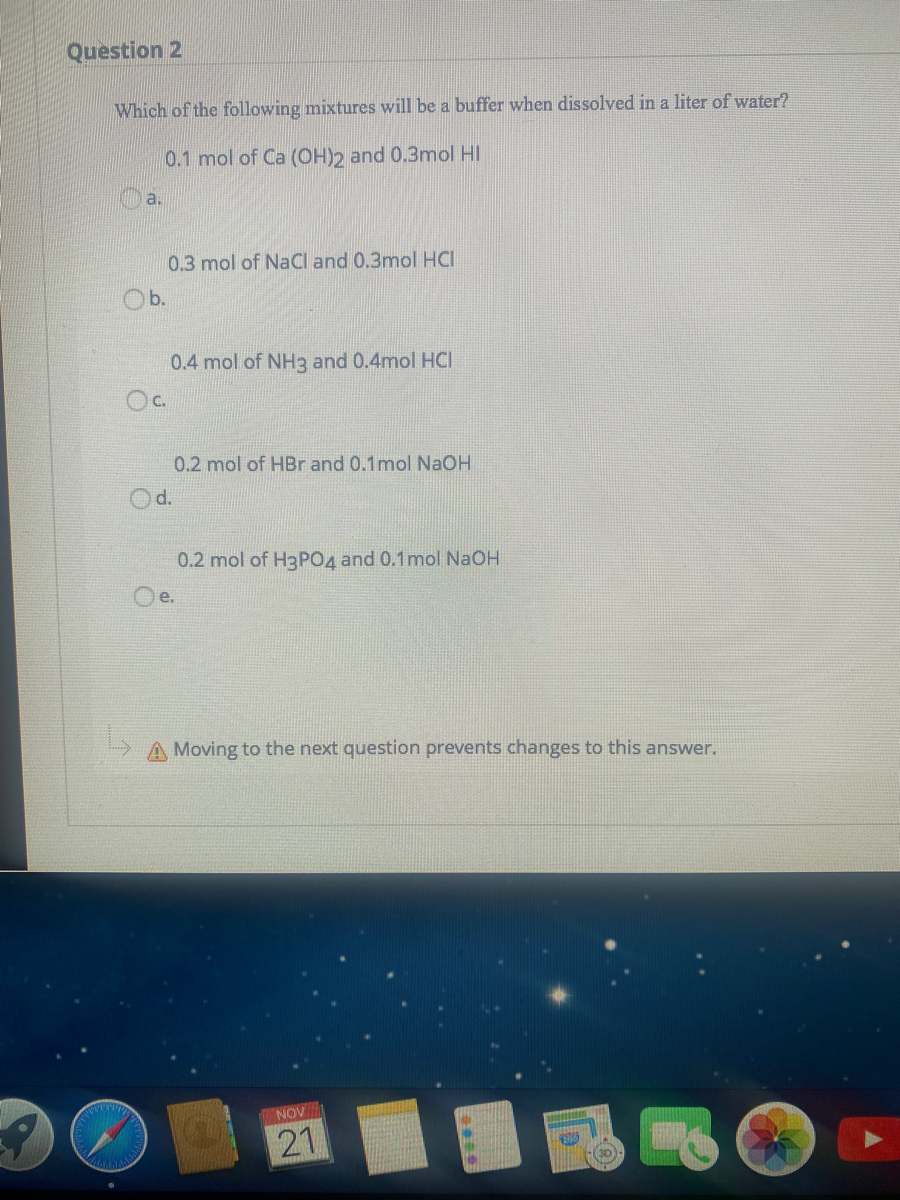
Answered Question 2 Which Of The Following Bartleby

Arrange The Group Of Compounds And Ions In Order Of Increasing Ph 1 Nh4cl H2so4 Naoh Na2co3 2 H2se Hi H2s Nh3 3 Hc Homeworklib
Solved 1 Write The Balanced Chemical Equations For The Neutralization Reactions Between A Hi And Naoh B H2co3 And Sr Oh 2 C Ca Oh 2 And H3po4 Course Hero

Acids Bases
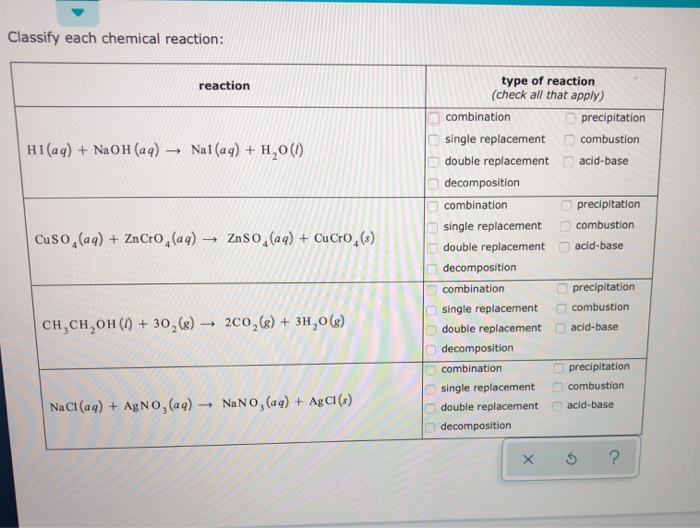
Solved Classify Each Chemical Reaction Reaction Type Of Chegg Com

Solved When Aqueous Solutions Of Hydroiodic Acid And Sodi Chegg Com

Solved Hi Aq Hio Aq Hi Aq Naoh Aq H Aq I Aq H Chegg Com

Classifying Compounds Compounds Can Be Classified By The Number Of Different Elements They Contain Binary Ternary Or Quaternary Hi Naoh Nh 4 Clo Ppt Download

Oneclass Assuming Equal Concentrations Rank These Solutions By Ph Highest Ph Sr Oh 2 Aq Naoh A
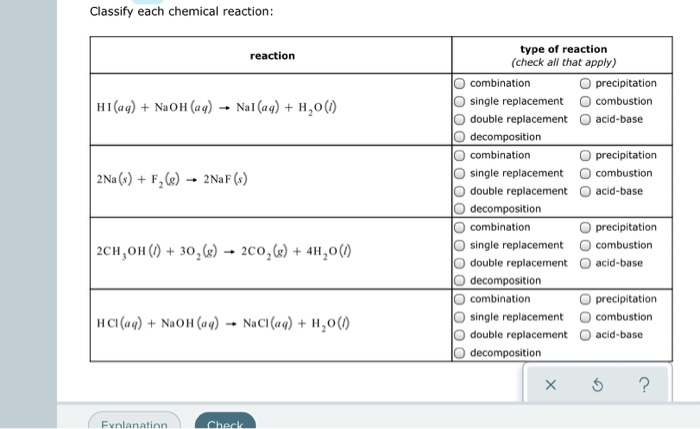
Solved Classify Each Chemical Reaction Reaction Hi Aq Chegg Com
Q Tbn And9gctft Gvmj5jpyo0 Sxmy7rehj 8moxorgrk Vb Ds7jeywroshi Usqp Cau

9krybmqaufadcm

Solved Consider 0 10 M Solutions Of The Following Hno2 Chegg Com

The Products A B And C Of The Following Reaction Are Respectively 1 Ccia 2 Naoh

3 Allyl Alcohol 4 O Cresol In Dow S Process Haloarene Is Converted To Phenol With Fused
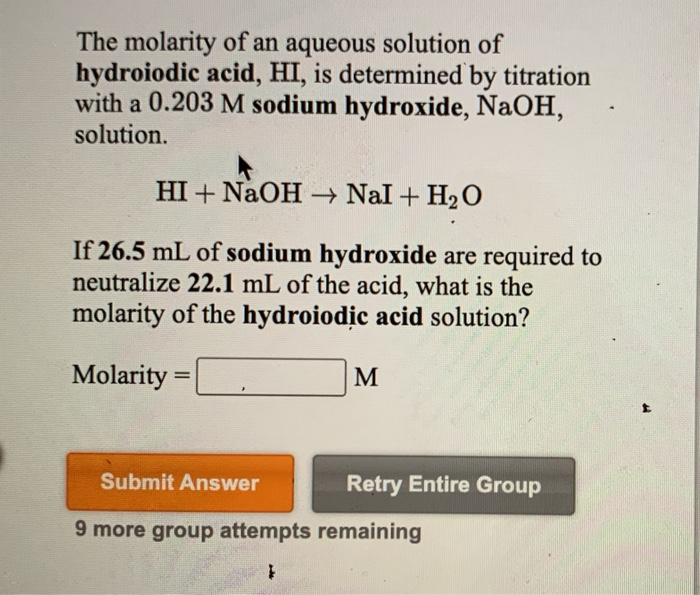
Solved The Molarity Of An Aqueous Solution Of Hydroiodic Chegg Com

Titration Of A Strong Acid With A Strong Base Chemistry Libretexts
Answered 8 What Volume Of 0 0m Hi Is Needed Bartleby

Key Gcc
2
Http Himedialabs Com Td Rm467 Pdf

Compound X One Treatment With Hi Give Y Y On Treatment With Ethan

Neutralization Acids And Bases
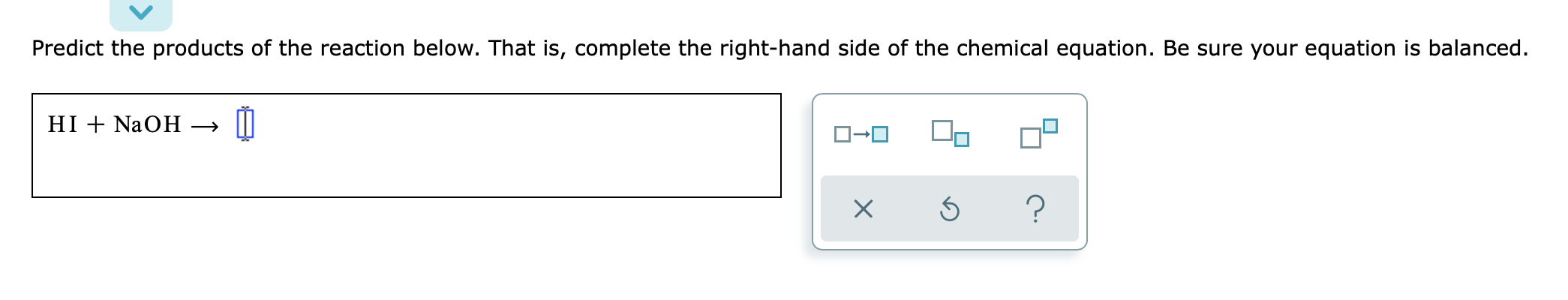
Solved Predict The Products Of The Reaction Below That I Chegg Com
Www Strongnet Org Cms Lib6 Oh Centricity Domain 6 Ch4a Pdf
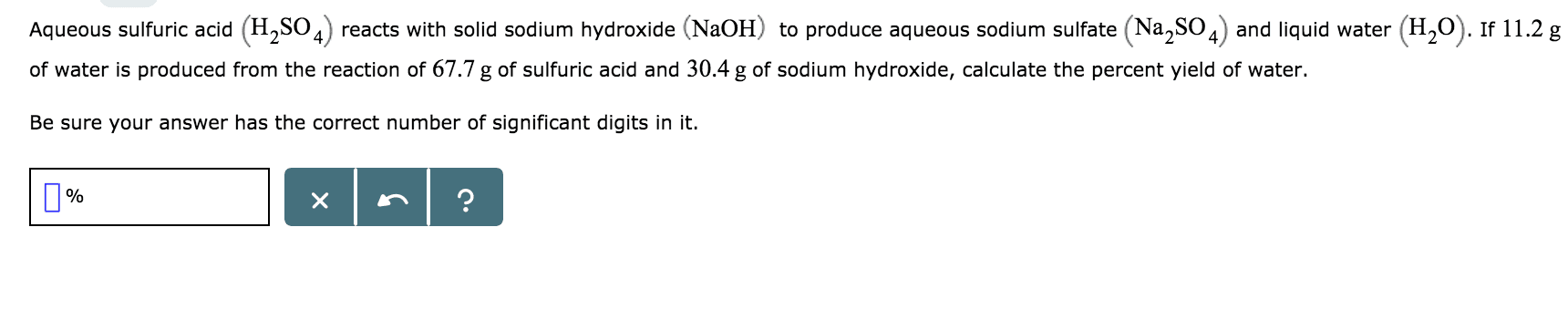
Oneclass Hi Can I Get Help In This Problem Hopefully You Guys Can Help Me Out Thank You Aqueous Su

Darrel Noahj456 Hi Naoh This Is Me Put This Pic In Your Next Stream T Co 06bgf7ig07

Answered The Molarity Of An Aqueous Solution Of Bartleby

Pdf Complex Permittivity Of Naoh Solutions Used In Liquid Metal Circuits

W Arm U P What Is The Ph Of A M Solution Of Hi What Is The Poh Of A M Solution Of Naoh What Is The H Concentration Of A Solution

Hi Temp Level Temperature Sensor For 50 Naoh Solution

Ch A Ch3 C 0 Ch3 Hi Ch B Ch3 Ch2 Ch Ch Cu 573k Oh

Darrel Noahj456 Hi Naoh This Is Me Put This Pic In Your Next Stream T Co 06bgf7ig07

Chg A Ch3 C 0 Ch3 Hi Ch3 B Ch3 Ch2 Ch Ch2 Cu 573k Cu

China Caustic Soda Naoh Flakes And Pearls 99 Min Qingdao Hiseachem China Caustic Soda 99 Industrial Grade

Exam 3 Answer Key

W Arm U P What Is The Ph Of A M Solution Of Hi What Is The Poh Of A M Solution Of Naoh What Is The H Concentration Of A Solution
3

How To Write The Net Ionic Equation For Hi Naoh Nai H2o Youtube

A Ki Hi B Kbr Hbr C Cucl Hcl D Nai Hi E Nach 3 Coo Ch 3 Cooh Ch 10 Acid Base Course Hero

Porcute Su Bar Pressure Concentrated Naoh Solution 600 C Temperature Normal Pressure D 6 8

Darrel Noahj456 Hi Naoh This Is Me Put This Pic In Your Next Stream T Co 06bgf7ig07

Hey Guys Detailed Please Please Ans Must Contain Details Brainly In

Neutralization Reactions Worksheet Hi Naoh H O Nai Hydroxide Acid

Solved Write Balanced Chemical Equations Describing The R Chegg Com

How To Balance Naoh Hi Nai H2o Sodium Hydroxide Hydroiodic Acid Youtube

How To Balance Naoh Hi Nai H2o Sodium Hydroxide Hydroiodic Acid Youtube

Acid Base Neutralization Equation Youtube

12ed Chapter 17 Pearsonbank Q S

A Ch 00 Na No Reaction Hi M Ei B Alcohol Naoh 12
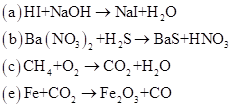
4 17 Balance Each Equation Bartleby
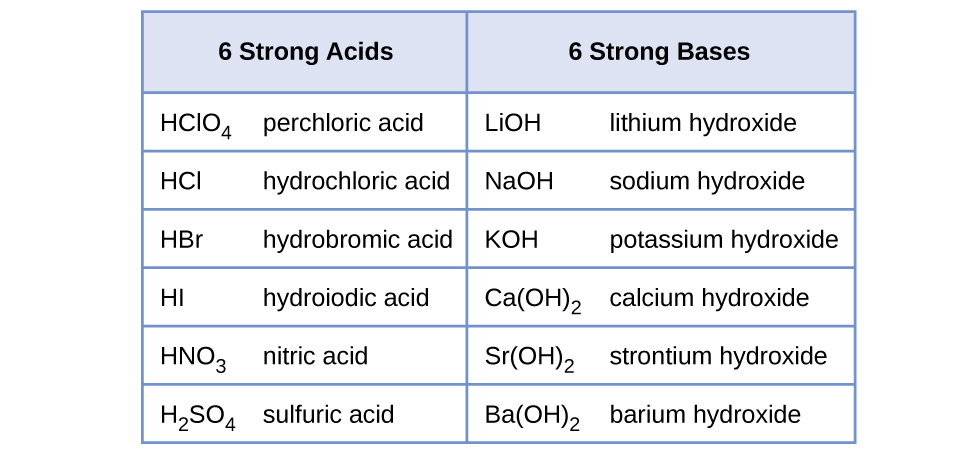
14 3 Relative Strengths Of Acids And Bases Chemistry
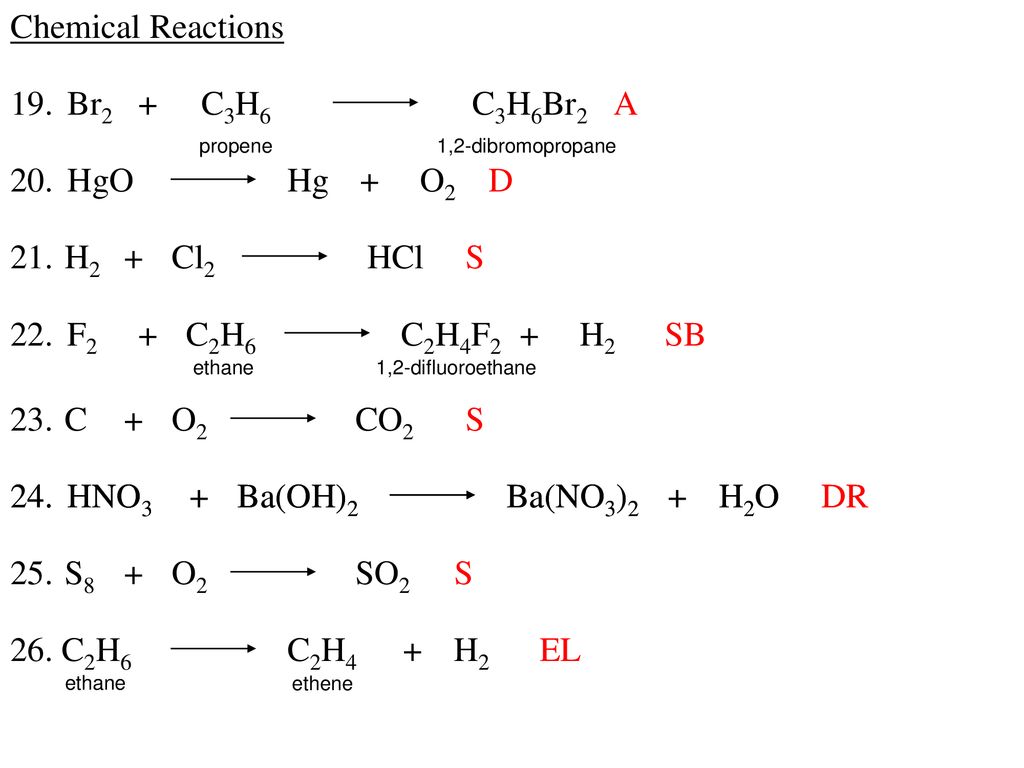
Chemical Reactions 1 H2 I2 Hi S 2 Na H2o Naoh H2 Sr 3 Co O2 Ppt Download

8 Chemical Equations Flames And Sparks Result When Aluminum Foil Ppt Video Online Download

How To Balance Naoh Hi Nai H2o Sodium Hydroxide Hydroiodic Acid Youtube

Which Compound Is Produced When Ethanal Is Heated With Dilute Naoh Solution Brainly In
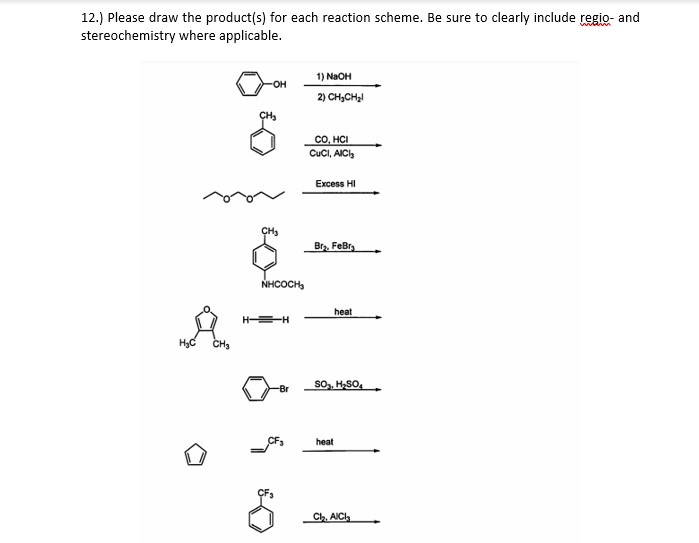
Answered 12 Please Draw The Product S For Bartleby

If A 17 5 Ml Sample Of 1 2 M Solution Of Each Of The Following Acids Is Homeworklib
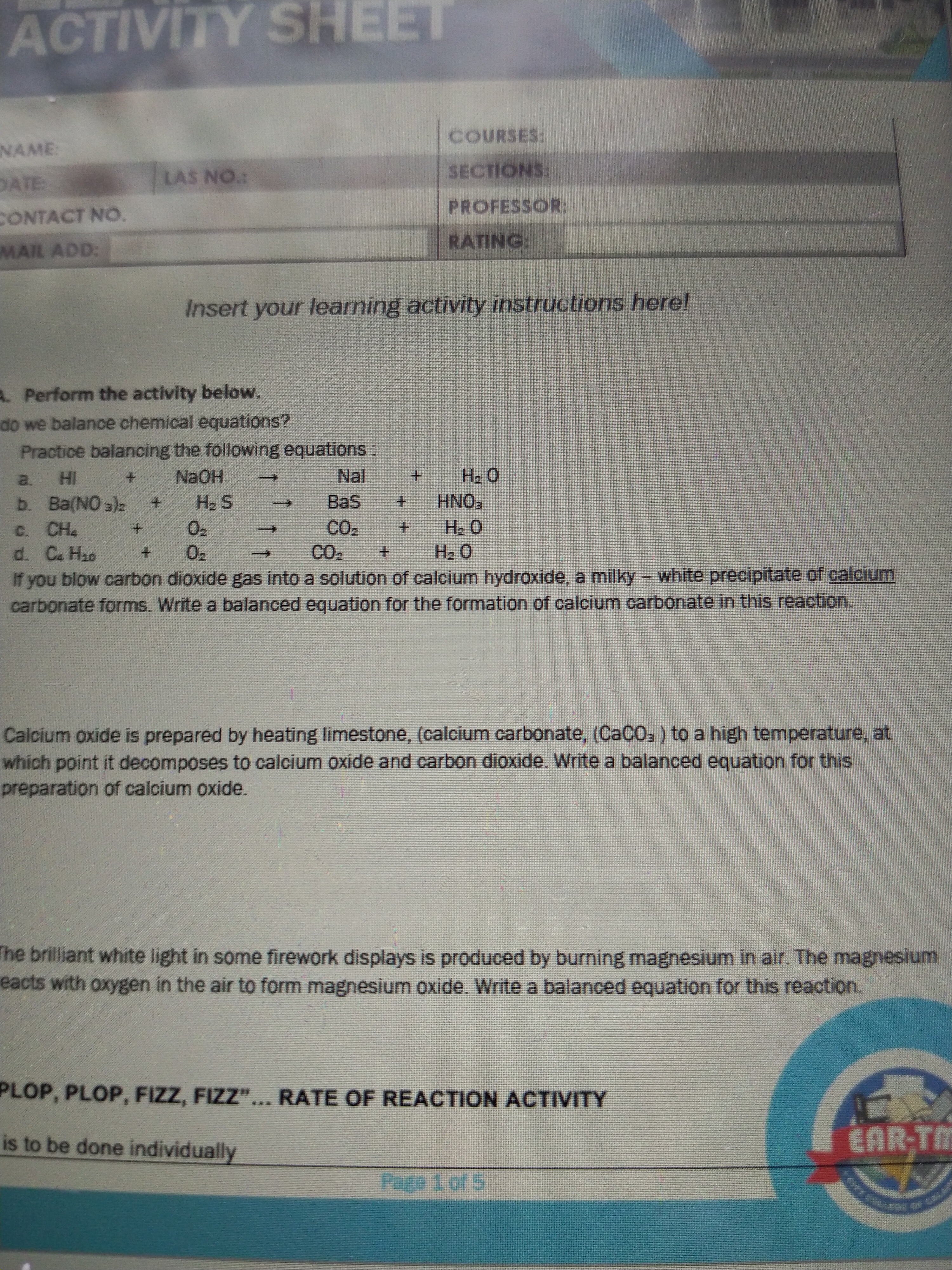
Answered Practice Balancing The Following Bartleby

Please If You Cant Answer All The Questions Do Not An Swer Any Of Them And Homeworklib

Solved What Is The Net Ionic Equation For The Reaction Th Chegg Com
Q Tbn And9gcsqplpzyo0j3jdiklussmkr9yib1jpd8ywwavykgtkpp7urjnug Usqp Cau
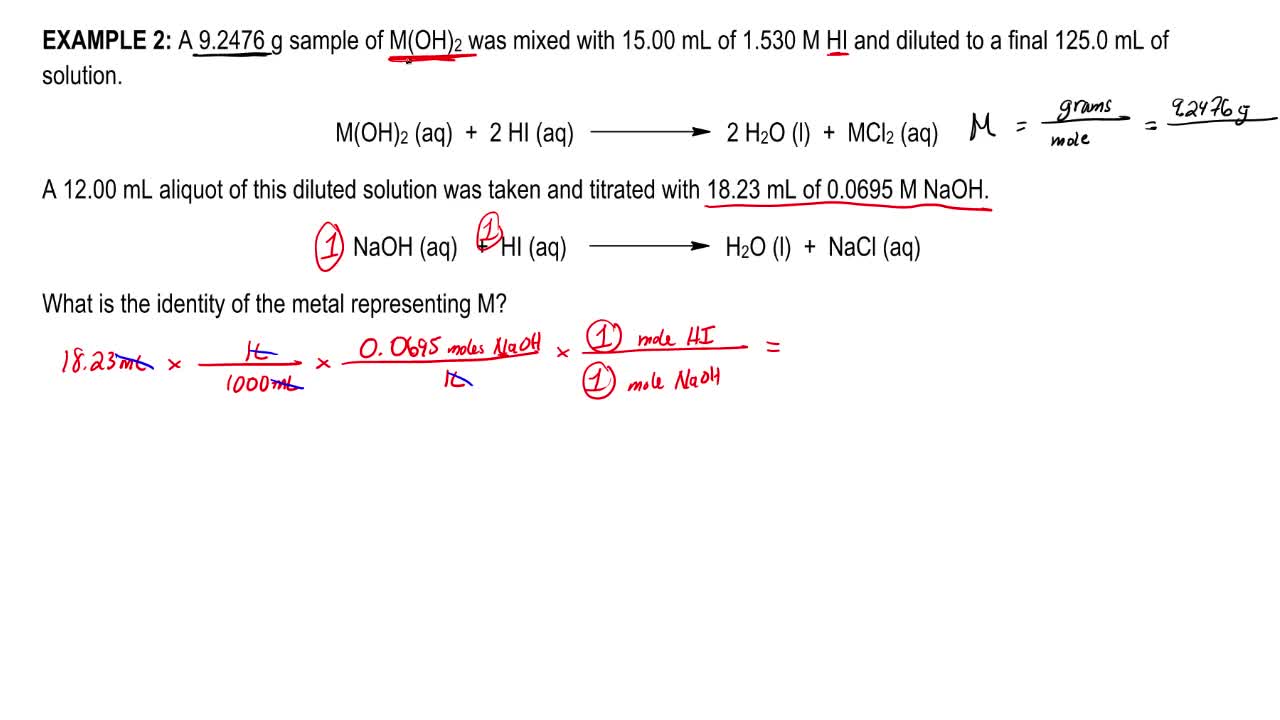
Volumetric Titrations Analytical Chemistry Video Clutch Prep

Neutralization Reactions Worksheet Hi Naoh H O Nai Hydroxide Acid
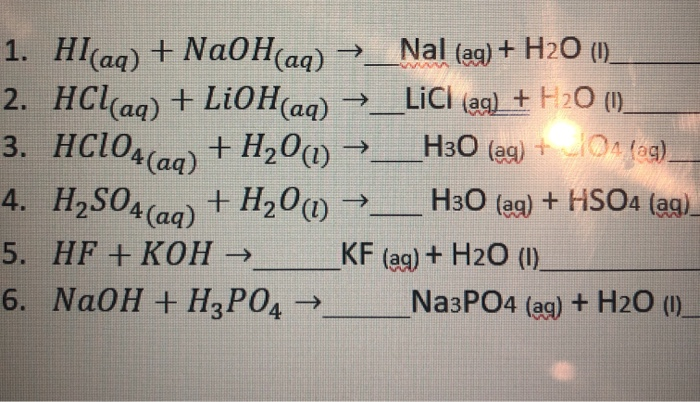
Solved 1 Hi Aq Naoh Aq Nal Ag H 1 2 Hcl Chegg Com

Oh H2 Pt Paul H Ch Cooh Naoh H I 28 Given The Following Cell At

Uses Of Hcl And Naoh In Our Lives Science Acids Bases And Salts Meritnation Com

Classifying Compounds Compounds Can Be Classified By The Number Of Different Elements They Contain Binary Ternary Or Quaternary Hi Naoh Nh 4 Clo Ppt Download

Classifying Compounds Compounds Can Be Classified By The Number Of Different Elements They Contain Binary Ternary Or Quaternary Hi Naoh Nh 4 Clo Ppt Download

The Compound With Formula C6h10o Is
An Ether X With Molecular Formula C5h10o Reacts With Excess Of Hot Aq Hi To Give A Product Which Sarthaks Econnect Largest Online Education Community

Solved Which Of The Following Is Are Strong Electrolyte Chegg Com
Q Tbn And9gcrqjhzd Jtkjc Qozyv8nhkshra6knmd3ajnurszwwumm8i8tpi Usqp Cau

Slides Show

Chemical Reactions 1 H2 I2 Hi S 2 Na H2o Naoh H2 Sr 3 Co O2 Ppt Download

Titration Chemistry For Non Majors

How To Balance Naoh Hi Nai H2o Sodium Hydroxide Hydroiodic Acid Youtube

3 By Which Of The Following Reagent Butanoic Acid Is Converted Into Butane A Red P
Http Campbellchembio Weebly Com Uploads 6 4 9 4 Extra Neutralization Practice With Key Pdf

94 Hi Excess Major Product Ii Chci Naoh Cho Oh Oculoh Loh Cho 4

Ii Chci1 Naoh I Hi Product Major Chemistry Aldehydes Ketones And Carboxylic Acids Meritnation Com

Solved Consider The Chemical Equation Hi Aq Naoh Aq Chegg Com

Classifying Compounds Compounds Can Be Classified By The Number Of Different Elements They Contain Binary Ternary Or Quaternary Hi Naoh Nh 4 Clo Ppt Download

Hi Sodium Hydroxide Solution 1n 1l



